Abstract
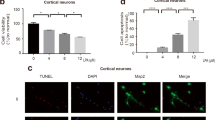
Src family of kinases (SFKs) and focal adhesion kinase (FAK) are two important cellular signaling components known to act cooperatively in the transduction of death and survival signals. We investigated the involvement of these proteins in the mechanism of the injurious response in rat primary neuronal cultures exposed to an insult composed of chemical ischemia (poisoning with iodoacetic acid; 100 μM, for 150 min) followed by 1 h of incubation in the regular medium, an insult shown before to be associated with generation of reactive oxygen species and with the depletion of adenosine triphisphate. The exposure of the neuronal cultures to the insult resulted in cell injury, assessed by the increased release of cytoplasmic lactate dehydrogenase (LDH) into the culture media, which could be attenuated markedly by the presence of the antioxidant LY 231617. The insult resulted in the decreased level of phosphorylation of the SFKs members Src, Fyn, and Yes at the Src Y416-equivalent activation sites and of the FAK Y397 activation site, degradation of FAK to a p85 fragment, and disassembling of the FAK–SFKs complexes. The inhibition of SFKs was found to be responsible for part of the insult-induced cell damage manifested in increased LDH release. Pervanadate, an inhibitor of the phosphotyrosine phosphatases (PTPs), abrogated the inactivation of SFKs and attenuated cell injury, indicating that insult-induced activation of PTPs is involved in SFKs inhibition and the ensued damage. The inhibition of SFKs and FAK is probably the cause of the disassembling of SFKs–FAK complexes, a process known to be associated with apoptosis.








Similar content being viewed by others
References
Abu-Ghazaleh, R., Kabir, J., Jia, H., Lobo, M., & Zachry, I. (2001). Src mediates stimulation by vascular endothelial growth factor of the phosphorylation of focal adhesion kinase at tyrosine 861, and migration and anti-apoptosis in endothelial cells. The Biochemical Journal, 360, 255–264.
Bradford, M. M. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry, 72, 248–254.
Cheung, R. T. (2003). The utility of melatonin in reducing cerebral damage resulting from ischemia and reperfusion. Journal of Pineal Research, 34, 153–160.
Chiang, G. G., & Sefton, B. M. (2000). Phosphorylation of a Src kinase at the autophosphorylation site in the absence of Src kinase activity. The Journal of Biological Chemistry, 275, 6055–6058.
Choi, J. S., Kim, H. Y., Chung, J. W., Chun, M. H., Kim, S. Y., Yoon, S. H., et al. (2005). Activation of Src tyrosine kinase in microglia in the rat hippocampus following transient forebrain ischemia. Neuroscience Letters, 380, 1–5.
Dahmani, S., Rouelle, D., Gressens, P., & Mantz, J. (2005). Effects of Dexmedetomidine on hippocampal focal adhesion kinase tyrosine phosphorylation in physiologic and ischemic conditions. Anesthesiology, 103, 969–977.
Dahmani, S., Tesniere, A., Rouelle, D., Desmonts, J. M., & Mantz, J. (2004). Thiopental and isoflurane attenuate the decrease in hippocampal phosphorylated Focal Adhesion Kinase (pp125 FAK) content induced by oxygen–glucose deprivation. British Journal of Anaesthesia, 93, 270–274.
Dawn, B., Takano, H., Tang, X. L., et al. (2002). Role of Src protein tyrosine kinases in late preconditioning against myocardial infarction. American Journal of Physiology. Heart and Circulatory Physiology, 283, H549–H556.
Droge, W. (2002). Free radicals in the physiological control of cell function. Physiological Reviews, 82, 47–95.
Gervais, F. G., Thornberry, N. A., Ruffolo, S. C., Nicholson, D. W., & Roy, S. (1998). Caspases cleave focal adhesion kinase during apoptosis to generate a FRNK-like polypeptide. The Journal of Biological Chemistry, 273, 17102–17108.
Giannoni, E., Buricchi, F., Raugei, G., Ramponi, G., & Chiarugi, P. (2005). Intracellular reactive oxygen species activate src tyrosine kinase during call adhesion and anchorage-dependent cell growth. Molecular and Cellular Biology, 15, 6391–6403.
Girault, J. A., Costa, A., Derkinderen, P., Studler, J. M., & Toutant, M. (1999). FAK and PYK2/CAKβ in the nervous system: A link between neuronal activity, plasticity and survival? Trends in Neurosciences, 22, 257–263.
Guo, J., Meng, F., Zhang, G., & Zhang, Q. (2003). Free radicals are involved in continuous activation of nonreceptor tyrosine protein kinase c-Src after ischemia/reperfusion in rat hippocampus. Neuroscience Letters, 345, 101–104.
Hashimoto, R., Fujimaki, K., Jeong, M. R., Christ, L., & Chuang, D. (2003). Lithium-induced inhibition of Src tyrosine kinase in rat cerebral cortical neurons: a role in neuroprotection against N-methyl-D-aspartate receptor-mediated excitotoxicity. FEBS Letters, 538, 145–148.
Hattori, R., Otani, H., Uchiyama, T., Imamura, H., Cui, J., Maulik, N., et al. (2001). Src tyrosine kinase is the triger but not the mediator of ischemic preconditioning. American Journal of Physiology. Heart and Circulatory Physiology, 281, H1066–H1074.
Head, B. P., Patel, H. H., Tsutsumi, Y. M., et al. (2008). Caveolin-1 expression is essential for N-methyl-D-aspartate receptor-mediated Src and extracellular signal-regulated kinase 1/2 activation and protection of primary neurons from ischemic cell death. The FASEB Journal, 22, 828–840.
Kelicen, P., Cantuti-Castelvetri, I., Pekiner, C., & Paulson, K. E. (2002). The spin trapping agent PBN stimulates H2O2-induced Erk and Src kinase activity in human neuroblastoma cells. Neurorepor, 13, 1057–1061.
Krieg, T., Qin, Q., McIntosh, E. C., Cohen, M. V., & Downey, J. M. (2002). ACh and adenosine activate PI3-kinase in rabbit hearts through transactivation of receptor tyrosine kinases. American Journal of Physiology. Heart and Circulatory Physiology, 283, H2322–H2330.
Lietha, D., Xinming, C., Derek, F. J. C., Yiqun, L., Schaller, M. D., & Eck, M. J. (2007). Structural basis for the autoinhibition of focal adhesion kinase. Cell, 129, 1177–1187.
Lipton, P. (1999). Ischemic cell death in brain neurons. Physiological Reviews, 79, 1431–1568.
Ludwig, L. M., Weihrauch, D., Kersten, J. R., Pagel, P. S., & Warltier, D. C. (2004). Protein kinase C translocation and Src protein tyrosine kinase activation mediate isoflurane-induced preconditioning in vivo: potential downstream targets of mitochondrial adenosine triphosphate-sensitive potassium channels and reactive oxygen species. Anesthesiology, 100, 532–539.
Mitra, S. K., Hanson, D. A., & Schlaepfer, D. D. (2005). Focal adhesion kinase: In command and control of cell motility. Nature, 6, 56–68.
Mitra, S. K., & Schlaepfer, D. D. (2006). Integrin-regulated FAK–Src signaling in normal and cancer cells. Current Opinion in Cell Biology, 18, 516–523.
Nakka, V. P., Gusain, A., Mehta, S. L., & Raghubir, R. (2008). Molecular mechanisms of apoptosis in cerebral ischemia: Multiple neuroprotective opportunities. Molecular Neurobiology, 37, 7–38.
Park, S. S., Eom, Y. W., Kim, E. H., et al. (2004). Involvement of c-Src kinase in the regulation of TGF-b1 induced apoptosis. Oncogene, 23, 6272–6281.
Ping, P., Song, C., Zhang, J., et al. (2002). Formation of protein kinase C(epsilon)–Lck signaling modules confers cardioprotection. The Journal of Clinical Investigation, 109, 499–507.
Pei, L., Li, Y., Yan, J. Z., Zhang, G. Y., Cui, Z. C., & Zhu, Z. M. (2000). Changes and mechanisms of protein-tyrosine kinase and protein-tyrosine phosphatase activities after brain ischemia/reperfusion. Acta Pharmacologica Sinica, 21, 715–720.
Pumiglia, K. M., Lau, L. F., Huang, C. K., Burroughs, S., & Feinstein, M. B. (1992). Activation of signal transduction in platelets by the tyrosine phosphatase inhibitor pervanadate (vanadyl hydroperoxide). The Biochemical Journal, 286(Pt 2), 441–449.
Reshef, A., Sperling, O., & Zoref-Shani, E. (2000). The adenosine-induced mechanism for the acquisition of ischemic tolerance in primary rat neuronal cultures. Pharmacology & Therapeutics, 87, 151–159.
Roskoski, R. (2004). Src protein-tyrosine kinase structure and regulation. Biochemical and Biophysical Research Communications, 324, 1155–1164.
Roskoski, R. (2005). Src kinase regulation by phosphorylation and dephosphorylation. Biochemical and Biophysical Research Communications, 331, 1–14.
Sperling, O., Bromberg, Y., Oelsner, H., & Zoref-Shani, E. (2003). Reactive oxygen species play an important role in iodoacetate-induced neurotoxicity in primary rat neuronal cultures and in differentiated PC12 cells. Neuroscience Letters, 351, 137–140.
Sun, G., Sharma, A. K., & Budde, R. J. (1998). Autophosphorylation of Src and Yes blocks their inactivation by Csk phosphorylation. Oncogene, 17, 1587–1595.
Wang, S. J. (2003). A role for Src kinase in the regulation of glutamate release from rat cerebrocortical nerve terminals. Neuroreport, 14, 1519–1522.
Wen, L. P., Fahrni, J. A., Troie, S., Guan, J. L., Orth, K., & Rosen, G. D. (1997). Cleavage of focal adhesion kinase by caspases during apoptosis. The Journal of Biological Chemistry, 272, 26056–26061.
Zalewska, T., Makarewicz, D., Janik, B., & Ziemka-Nalecz, M. (2005). Neonatal cerebral hypoxia-ischemia: involvement of FAK-dependent pathway. International Journal of Developmental Neuroscience, 23, 657–662.
Ziemka-Nalecz, M., & Zalewska, T. (2007). Transient forebrain ischemia effects FAK-coupled signaling in gerbil hippocampus. Neurochemistry International, 51, 405–411.
Acknowledgments
This study was supported in part by the Benjamin Turkienicz Estate, the Dr. Ziternick and Haia Silva Ziternick Fund, and by a grant from the Adams Super Center for Brain Research, Tel-Aviv University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shani, V., Bromberg, Y., Sperling, O. et al. Involvement of Src Tyrosine Kinases (SFKs) and of Focal Adhesion Kinase (FAK) in the Injurious Mechanism in Rat Primary Neuronal Cultures Exposed to Chemical Ischemia. J Mol Neurosci 37, 50–59 (2009). https://doi.org/10.1007/s12031-008-9113-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-008-9113-3