Abstract
Introduction
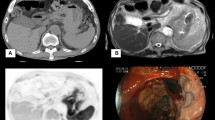
Pretreatment diagnosis by diffusion-weighted magnetic resonance imaging (DW-MRI) is useful to determine the effect of chemotherapy for gastric cancer. Here, we investigated the relationship among DW-MRI, endoscopy, and tumor markers.
Patients
Eight patients underwent hemostatic radiotherapy (RT) for gastric cancer in this prospective study from 2019 to 2021. The patients completed MRI, endoscopy, and blood tests before RT; MRI, endoscopy, and blood tests 1 month after RT; and MRI and blood tests 3 months after RT. Correlations between changes in apparent diffusion coefficient (ADC) derived from DW-MRI and the tumor marker carcinoembryonic antigen (CEA) were investigated.
Results
Univariate analysis of overall survival showed that sex and chemotherapy treatment were statistically significant factors. The CEA values before and 1 month after RT decreased significantly. There was no statistical difference between the CEA value 1 and 3 months after RT. The ADC value before and 1 month after RT increased significantly but not between 1 and 3 months after RT. Comparing the ratio of ADC before RT to 1 (or 3) month(s) after RT with that of CEA before RT to 1 (or 3) month(s) after RT, we found an inverse relationship between the two ratios.
Conclusions
Therefore, changes in ADC and CEA are correlated. Additionally, 3 months after RT, the decrease in ADC appeared earlier than the decrease in CEA. ADC may indicate a biological change earlier than CEA, and the ratios of ADC and CEA may be important factors. These aspects warrant further confirmation in a larger sample population.






Similar content being viewed by others
Data Availability
Research data are stored in an institutional repository and will be shared upon request to the corresponding author.
References
Tanaka O, Sugiyama A, Omatsu T, et al. Hemostatic radiotherapy for inoperable gastric cancer: a pilot study. Br J Radiol. 2020;93:20190958.
Tey J, Zheng H, Soon YY, et al. Palliative radiotherapy in symptomatic locally advanced gastric cancer: a phase II trial. Cancer Med. 2019;8:1447–58. https://doi.org/10.1002/cam4.2021.
Tanaka O, Matsuura K, Sugiyama A, et al. Hemostatic radiotherapy used twice for inoperable progressive gastric cancer with bleeding. J Gastrointest Cancer. 2019;50:151–5.
Tanaka O, Yokoi R, Mukai T, et al. Radiotherapy for gastric bleeding from tumor invasion of recurrent colon cancer with liver metastasis after resection. J Gastrointest Cancer. 2019;50:349–52.
Tanaka O, Yamada M, Kato T, et al. Two sessions of radiotherapy were successful in treating gastric cancer with bleeding. J Gastrointest Cancer. 2019;50:955–9.
Lee YH, Lee JW, Jang HS. Palliative external beam radiotherapy for the treatment of tumor bleeding in inoperable advanced gastric cancer. BMC Cancer. 2017;17:541. https://doi.org/10.1186/s12885-017-3508-x.
Digklia A, Wagner AD. Advanced gastric cancer: current treatment landscape and future perspectives. World J Gastroenterol. 2016;22:2403–14. https://doi.org/10.3748/wjg.v22.i8.2403.
Zhang Y, Yu J. The role of MRI in the diagnosis and treatment of gastric cancer. Diagn Interv Radiol. 2020;26:176–82. https://doi.org/10.5152/dir.2019.19375.
Borggreve AS, Goense L, Brenkman HJF, et al. Imaging strategies in the management of gastric cancer: current role and future potential of MRI. Br J Radiol. 2019;92:20181044. https://doi.org/10.1259/bjr.20181044.
Liu S, Zhang Y, Chen L, et al. Whole-lesion apparent diffusion coefficient histogram analysis: significance in T and N staging of gastric cancers. BMC Cancer. 2017;17:665. https://doi.org/10.1186/s12885-017-3622-9.
Onal Y, Samanci C. The role of diffusion-weighted imaging in patients with gastric wall thickening. Curr Med Imaging Rev. 2019;15:965–71. https://doi.org/10.2174/1573405614666181115120109.
Tang L, Wang XJ, Baba H, et al. Gastric cancer and image-derived quantitative parameters: part 2-a critical review of DCE-MRI and 18F-FDG PET/CT findings. Eur Radiol. 2020;30:247–60. https://doi.org/10.1007/s00330-019-06370-x.
Soydan L, Demir AA, Torun M, et al. Use of diffusion-weighted magnetic resonance imaging and apparent diffusion coefficient in gastric cancer staging. Curr Med Imaging. 2020;16:1278–89. https://doi.org/10.2174/1573405616666200218124926.
Liu S, Wang H, Guan W, et al. Preoperative apparent diffusion coefficient value of gastric cancer by diffusion-weighted imaging: correlations with postoperative TNM staging. J Magn Reson Imaging. 2015;42:837–43. https://doi.org/10.1002/jmri.24841.
Joo I, Lee JM, Kim JH, et al. Prospective comparison of 3T MRI with diffusion-weighted imaging and MDCT for the preoperative TNM staging of gastric cancer. J Magn Reson Imaging. 2015;41:814–21. https://doi.org/10.1002/jmri.24586.
Javadinia SA, Shahidsales S, Fanipakdel A, et al. The esophageal cancer and the PI3K/AKT/mTOR signaling regulatory microRNAs: a novel marker for prognosis, and a possible target for immunotherapy. Curr Pharm Des. 2018;24:4646–51. https://doi.org/10.2174/1381612825666190110143258 (PMID: 30636576).
Javadinia SA, Shahidsales S, Fanipakdel A, et al. Therapeutic potential of targeting the Wnt/β-catenin pathway in the treatment of pancreatic cancer. J Cell Biochem. 2018. https://doi.org/10.1002/jcb.27835 (PMID: 30368889).
Fanipakdel A, Seilanian Toussi M, Rezazadeh F, et al. Overexpression of cancer-testis antigen melanoma-associated antigen A1 in lung cancer: a novel biomarker for prognosis, and a possible target for immunotherapy. J Cell Physiol. 2019;234:12080–6. https://doi.org/10.1002/jcp.27884 (PMID: 30569450).
Javadinia SA, Gholami A, Joudi Mashhad M, et al. Anti-tumoral effects of low molecular weight heparins: a focus on the treatment of esophageal cancer. J Cell Physiol. 2018;233:6523–9. https://doi.org/10.1002/jcp.26613 (PMID: 29741755).
Fazilat-Panah D, Vakili Ahrari Roudi S, Keramati A, et al. Changes in cytokeratin 18 during neoadjuvant chemotherapy of breast cancer: a prospective study. Iran J Pathol 2020;15:117–126. https://doi.org/10.30699/ijp.2020.116238.2261. (PMID: 32215027; PMCID: PMC7081760).
Cardoso R, Coburn N, Seevaratnam R, et al. A systematic review and meta-analysis of the utility of EUS for preoperative staging for gastric cancer. Gastric Cancer. 2012;15(Suppl 1):S19–26. https://doi.org/10.1007/s10120-011-0115-4.
Saito T, Kurokawa Y, Takiguchi S, et al. Accuracy of multidetector-row CT in diagnosing lymph node metastasis in patients with gastric cancer. Eur Radiol. 2015;25:368–74. https://doi.org/10.1007/s00330-014-3373-9.
Saito T, Kosugi T, Nakamura N, et al. Treatment response after palliative radiotherapy for bleeding gastric cancer: a multicenter prospective observational study (JROSG 17–3). Gastric Cancer (in press). https://doi.org/10.1007/s10120-021-01254-w.
Tanaka O, Omatsu T, Kariya S, et al. Usefulness of diffusion-weighted magnetic resonance imaging for evaluating the effect of hemostatic radiotherapy for unresectable gastric cancer. Clin J Gastroenterol. 2019;12:269–73.
Lutz ST, Jones J, Chow E. Role of radiation therapy in palliative care of the patient with cancer. J Clin Oncol 2014;32:2913–2919. https://doi.org/10.1200/JCO.2014.55.1143.
Rueth NM1, Shaw D, D'Cunha J, et al. Esophageal stenting and radiotherapy: a multimodal approach for the palliation of symptomatic malignant dysphagia. Ann Surg Oncol 2012;19:4223–4228. https://doi.org/10.1245/s10434-012-2459-3.
Picardi V, Deodato F, Guido A. Palliative short-course radiation therapy in rectal cancer: a phase 2 study. Int J Radiat Oncol Biol Phys. 2016;95:1184–90. https://doi.org/10.1016/j.ijrobp.2016.03.010.
Lacarrière E, Smaali C, Benyoucef A, et al. http://www.scielo.br/pdf/ibju/v39n6/1677-5538-ibju-39-06-808.pdf. Int Braz J Urol. 2013;39:808–816.
Eleje GU, Eke AC, Igberase GO, et al. Palliative interventions for controlling vaginal bleeding in advanced cervical cancer. Cochrane Database Syst Rev 2019;3:CD011000. https://doi.org/10.1002/14651858.CD011000.pub3. (PMID: 30888060).
Author information
Authors and Affiliations
Contributions
Statistical analysis: Osamu Tanaka.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tanaka, O., Yagi, N., Tawada, M. et al. Hemostatic Radiotherapy for Gastric Cancer: MRI as an Alternative to Endoscopy for Post-Treatment Evaluation. J Gastrointest Canc 54, 554–563 (2023). https://doi.org/10.1007/s12029-022-00837-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12029-022-00837-9