Abstract
Background
Recent evidence from randomized trials suggests that FOLFOXIRI (fluorouracil, oxaliplatin, and irinotecan) ± bevacizumab is associated with higher response rates, with the potential for conversion of unresectable to resectable disease in metastatic colorectal cancer (mCRC). However, limited evidence is available on the efficacy and safety of this regimen in real-world patients with mCRC. The current study aims to evaluate the conversion rate and safety of FOLFOXIRI ± bevacizumab in real-world patients with unresectable mCRC.
Methods
In this retrospective multicenter population–based cohort study, patients who were diagnosed with unresectable mCRC between January 2015 and December 2018 in Saskatchewan and received FOLFOXIRI ± bevacizumab were assessed. Kaplan–Meier survival methods and the log-rank test were performed.
Results
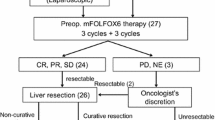
A total of 28 eligible patients with a median age of 51 years (interquartile range 39–60) and a male:female ratio of 11:17 were identified; 39% had rectal cancer, 46% had extrahepatic disease, and 46% had bilobar liver metastases. Overall, 63% of the patients had a positive response to FOLFOXIRI ± bevacizumab and 53% underwent metastasectomy. Of all patients 60% had grade 3/4 toxicity and 32% required hospital admission. No treatment-related mortality was noted. After 4 years, 50% of the patients were alive. Median progression-free survival of patients who underwent surgery was 18 months (95% CI 11.3–24.7) versus 11 months (4–18.1) without surgery (p = 0.28). Median overall survival of patients with surgery was 33 months (17.5–48.5) versus 16 months (8.3–23.7) without surgery (p = 0.03).
Conclusion
The current study suggests that FOLFOXIRI ± bevacizumab therapy in real-world patients with mCRC is associated with a high rate of conversion from unresectable to resectable metastatic disease. Patients with metastasectomy had better survival.


Similar content being viewed by others
References
Arnold M, Sierra MS, Laversanne M, et al. Global patterns and trends in colorectal cancer incidence and mortality. Gut. 2017;66(4):683–91.
Bartlettt DL, Chu E. Can metastatic colorectal cancer be cured? Oncology. 2012;26:266–75.
Cummings LC, Payes JD, Cooper GS. Survival after hepatic resection in metastatic colorectal cancer: a population-based study. Cancer. 2007;109:718.
Morris EJ, Forman D, Thomas JD, et al. Surgical management and outcomes of colorectal cancer liver metastases. Br J Surg. 2010;97:1110.
Rees M, Tekkis PP, Welsh FK, et al. Evaluation of long-term survival after hepatic resection for metastatic colorectal cancer: a multifactorial model of 929 patients. Ann Surg. 2008;247:125.
Simmonds PC, Primrose JN, Colquitt JL, et al. Surgical resection of hepatic metastases from colorectal cancer: a systematic review of published studies. Br J Cancer. 2006;94:982.
Wei AC, Greig PD, Grant D, et al. Survival after hepatic resection for colorectal metastases: a 10-year experience. Ann Surg Oncol. 2006;13:668–92.
Fernandez FG, Drebin JA, Linehan DC, et al. Five-year survival after resection of hepatic metastases from colorectal cancer in patients screened by positron emission tomography with F-18 fluorodeoxyglucose (FDG-PET) Ann Surg. 2004;240:438–447.
Lee WS, Yun SH, Chun HK, et al. Clinical outcomes of hepatic resection and radiofrequency ablation in patients with solitary colorectal liver metastasis. J Clin Gastroenterol. 2008;42(8):945–9.
Lordan JT, Riga A, Worthington TR, Karanjia ND. Early and long-term outcomes of patients undergoing liver resection and diaphragm excision for advanced colorectal liver metastases. Ann R Coll Surg Engl. 2009;91(6):483–8.
Nikfarjam M, Shereef S, Kimchi ET, et al. Survival outcomes of patients with colorectal liver metastases following hepatic resection or ablation in the era of effective chemotherapy. Ann Surg Oncol. 2009;16(7):1860–7.
Choti MA, Sitzmann JV, Tiburi MF, et al. Trends in long-term survival following liver resection for hepatic colorectal metastases. Ann Surg. 2002;235(6):759–66.
Ahmed S, Johnson K, Ahmed O, Iqbal N. Advances in the management of colorectal cancer: from biology to treatment. Int J Colorectal Dis. 2014;29:1031–42.
Kanthan R, Senger JL, Ahmed S, Kanthan SC. Recent advances in the management of stage IV colon cancer. J Cancer Ther. 2012;3:1104–18.
Loupakis F, Cremolini C, Masi G, et al. Initial therapy with FOLFOXIRI and bevacizumab for metastatic colorectal cancer. N Engl J Med. 2014;371:1609–18.
Gruenberger T, Bridgewater J, Chau I, et al. Bevacizumab plus mFOLFOX-6 or FOLFOXIRI in patients with initially unresectable liver metastases from colorectal cancer: the OLIVIA multinational randomised phase II trial. Ann Oncol. 2015;26:702–8.
Falcone A, Ricci S, Brunetti I, et al. Phase III trial of infusional fluorouracil, leucovorin, oxaliplatin, and irinotecan (FOLFOXIRI) compared with infusional fluorouracil, leucovorin, and irinotecan (FOLFIRI) as first-line treatment for metastatic colorectal cancer: the Gruppo Oncologico Nord Ovest. J Clin Oncol. 2007;25:1670.
Masi G, Vasile E, Loupakis F, et al. Randomized trial of two induction chemotherapy regimens in metastatic colorectal cancer: an updated analysis. J Natl Cancer Inst. 2011;103:21.
Tomasello G, Petrelli F, Ghidini M, et al. FOLFOXIRI plus bevacizumab as conversion therapy for patients with initially unresectable metastatic colorectal cancer: a systematic review and pooled analysis. JAMA Oncol. 2017;3:e170278.
Ahmed S, Leis A, Kanthan S, et al. Surgical management of the primary tumor in stage IV colorectal cancer: a confirmatory retrospective cohort study. J Cancer. 2016;7:837–45.
Caputo F, Santini C, Bardasi C, et al. BRAF-mutated colorectal cancer: clinical and molecular insights. Int J Mol Sci. 2019;20(21):5369.
Conversion From Unresectable To Resectable Metastatic Colorectal Cancer. (CONVERSION) https://clinicaltrials.gov/ct2/show/NCT03401294
Ahmed S, Anderson D, Chalchal H, et al. Conversion from unresectable to resectable liver metastases in real-world patients with liver-only metastatic colorectal cancer (mCRC) treated with FOLFOXIRI plus bevacizumab: The Conversion trial. J Clin Onc 2021;39:3suppl.
Acknowledgements
We acknowledge Saskatchewan Cancer Agency and its Data Access Committee for providing support for this research.
Funding
This study was funded by the 2019 Dean’s Summer Student Research Project grant at University of Saskatchewan, College of Medicine.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics
This study was approved by the University of Saskatchewan’s Biomedical Research Ethic Board.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bhatti, T., Moser, M., Tan, K.T. et al. Rate of Curative Surgery in Real-world Patients with Unresectable Metastatic Colorectal Cancer Treated with FOLFOXIRI ± Bevacizumab: A Western Canadian Province Experience. J Gastrointest Canc 53, 427–433 (2022). https://doi.org/10.1007/s12029-021-00634-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12029-021-00634-w