Abstract
Purpose
Colorectal cancer is the third leading diagnosis accounting for nearly 10% of all new cancers worldwide. The distinct features among BRAF mutant colorectal cancers make these tumor groups hard to treat for oncologists. The median overall survival (OS) of these types of cancers is reported to be 9 to 14 months.
Methods
The study was declared on the Turkish Oncology Study Group Conference and approved. The patients’ data was received from the centers who confirmed to participate. The BRAF-mutated patients were included in the study. The demographic features (age, gender, etc.), type of mutation, tumor localizations, histology, microsatellite instability (MSI) status, metastasis patterns chemotherapeutic agents and progression, and death times were recorded.
Results
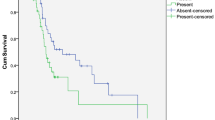
Thirty-nine patients were enrolled in the study. Sixteen patients had concurrent KRAS mutations, while 7 had NRAS mutations. Most of the patients received doublet chemotherapies in combination with anti-VEGF agents in the first and second line of the treatment. There was a significant difference in OS according to the stage which showed a decreased survival in stage IV patients at the time of diagnosis. Concurrent KRAS mutation resulted in increased OS. The median OS was 47 and 24 months favoring the KRAS mutant group. The patients whose primary tumor operated had better survival when compared with other patients. The median OS of the operated group was 47 months, while the non-operated group was 24 months. Liver metastasis was related to worse prognosis at the time of diagnosis in univariate analysis.
Conclusion
In our study we found a high concurrent RAS mutation ratio in a BRAF mutant patient group which was different from prior studies. The concurrent mutations resulted in a favorable outcome in terms of OS which is also different from the current knowledge. More prospective studies are needed especially BRAF-mutated patient population and especially with concurrent RAS mutations.

Similar content being viewed by others
References
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65:87–108.
Schmoll HJ, Van Cutsem E, Stein A, Valentini V, Glimelius B, Haustermans K, et al. Esmo consensus guidelines for management of patients with colon and rectal cancer. A personalized approach to clinical decision making. Ann Oncol. 2012;23:2479–516.
Aykan NF, Yalcin S, Turhal NS, Özdoʇan M, Demir G, Özkan M, et al. Epidemiology of colorectal cancer in Turkey: a cross-sectional disease registry study (a Turkish oncology group trial). Turkish J Gastroenterol. 2015;26:145–53.
Alici S, Aykan NF, Sakar B, Bulutlar G, Kaytan E, Topuz E. Colorectal cancer in young patients: characteristics and outcome. Tohoku J. Exp. Med. 2003. p. 85–93.
Frattini M, Balestra D, Suardi S, Oggionni M, Alberici P, Radice P, et al. Different genetic features associated with colon and rectal carcinogenesis. Clin Cancer Res. 2004;10:4015–21.
Tamas K, Walenkamp AME, de Vries EGE, van Vugt MATM, Beets-Tan RG, van Etten B, et al. Rectal and colon cancer: not just a different anatomic site. Cancer Treat. Rev. 2015. p. 671–9.
Ulanja MB, Rishi M, Beutler BD, Sharma M, Patterson DR, Gullapalli N, et al. Colon cancer sidedness, presentation, and survival at different stages. J Oncol. 2019;2019:1–12.
Dhillon AS, Hagan S, Rath O, Kolch W. MAP kinase signalling pathways in cancer. Oncogene. 2007. p. 3279–90.
Oikonomou E, Makrodouli E, Evagelidou M, Joyce T, Probert L, Pintzas A. BRAFV600E efficient transformation and induction of microsatellite instability versus KRASG12V induction of senescence markers in human colon cancer cells. Neoplasia. 2009;11:1116–31.
Kim IJ, Kang HC, Jang SG, Kim K, Ahn SA, Yoon HJ, et al. Oligonucleotide microarray analysis of distinct gene expression patterns in colorectal cancer tissues harboring BRAF and K-ras mutations. Carcinogenesis. 2006;27:392–404.
Kikuchi H, Pino MS, Min Z, Shirasawa S, Chung DC. Oncogenic KRAS and BRAF differentially regulate hypoxia-inducible factor-1α and -2α in colon cancer. Cancer Res. 2009;69:8499–506.
Sahin IH, Kazmi SMA, Yorio JT, Bhadkamkar NA, Kee BK. Garrett C pher R. Rare though not mutually exclusive: a report of three cases of concomitant kras and braf mutation and a review of the literatuer. J Cancer. 2013;4:320–2.
Cerottini JP, Caplin S, Saraga E, Givel JC, Benhattar J. The type of K-ras mutation determines prognosis in colorectal cancer. Am J Surg. 1998;175:198–202.
Andreyev HJN, Norman AR, Cunningham D, Oates JR, Clarke PA. Kirsten ras mutations in patients with colorectal cancer: the multicenter “RASCAL” study. J Natl Cancer Inst. 1998;90:675–84.
Samowitz WS, Curtin K, Schaffer D, Robertson M, Leppert M, Slattery ML. Relationship of Ki-ras mutations in colon cancers to tumor location, stage, and survival: a population-based study. Cancer Epidemiol Biomarkers Prev. 2000;9:1193–7.
Yoon HH, Tougeron D, Shi Q, Alberts SR, Mahoney MR, Nelson GD, et al. KRAS codon 12 and 13 mutations in relation to disease-free survival in BRAF-wild-type stage III colon cancers from an adjuvant chemotherapy trial (N0147 alliance). Clin Cancer Res. 2014;20:3033–43.
Modest DP, Ricard I, Heinemann V, Hegewisch-Becker S, Schmiegel W, Porschen R, et al. Outcome according to KRAS-, NRAS- and BRAF-mutation as well as KRAS mutation variants: pooled analysis of five randomized trials in metastatic colorectal cancer by the AIO colorectal cancer study group. Ann Oncol. 2016;27:1746–53.
Taieb J, Zaanan A, Le Malicot K, Julié C, Blons H, Mineur L, et al. Prognostic effect of BRAF and KRAS mutations in patients with stage III colon cancer treated with leucovorin, fluorouracil, and oxaliplatin with or without cetuximab. JAMA Oncol. 2016;2:643–53.
Taieb J, Le Malicot K, Shi Q, Lorca FP, Bouché O, Tabernero J, et al. Prognostic value of BRAF and KRAS mutations in MSI and MSS Stage III colon cancer. J Natl Cancer Inst. 2017;109:djw272.
Gonsalves WI, Mahoney MR, Sargent DJ, Nelson GD, Alberts SR, Sinicrope FA, et al. Patient and tumor characteristics and BRAF and KRAS mutations in colon cancer, NCCTG/Alliance N0147. J Natl Cancer Inst. 2014;106.
Taieb J, Tabernero J, Mini E, Subtil F, Folprecht G, Van Laethem J, et al. Adjuvant Folfox4 with or without cetuximab (CTX) in patients (PTS) with resected stage III colon cancer (CC): Dfs and OS results and subgroup analyses of the PETACC8 intergroup phase III trial. Ann Oncol. 2012;23:ixe9.
Roth AD, Tejpar S, Delorenzi M, Yan P, Fiocca R, Klingbiel D, et al. Prognostic role of KRAS and BRAF in stage II and III resected colon cancer: results of the translational study on the PETACC-3, EORTC 40993, SAKK 60-00 trial. J Clin Oncol. 2010;28:466–74.
Wan PTC, Garnett MJ, Roe SM, Lee S, Niculescu-Duvaz D, Good VM, et al. Mechanism of activation of the RAF-ERK signaling pathway by oncogenic mutations of B-RAF. Cell. 2004;116:855–67.
Poulikakos PI, Zhang C, Bollag G, Shokat KM, Rosen N. RAF inhibitors transactivate RAF dimers and ERK signalling in cells with wild-type BRAF. Nature. 2010;464:427–30.
Matallanas D, Birtwistle M, Romano D, Zebisch A, Rauch J, von Kriegsheim A, et al. Raf family kinases: old dogs have learned new tricks. Genes and Cancer. 2011. p. 232–60.
Davies H, Bignell GR, Cox C, Stephens P, Edkins S, Clegg S, et al. Mutations of the BRAF gene in human cancer. Nature. 2002;417:949–54.
Venderbosch S, Nagtegaal ID, Maughan TS, Smith CG, Cheadle JP, Fisher D, et al. Mismatch repair status and BRAF mutation status in metastatic colorectal cancer patients: a pooled analysis of the CAIRO, CAIRO2, COIN, and FOCUS studies. Clin Cancer Res. 2014;20:5322–30.
McCubrey, JA, Steelman LS, Chappell WH, Abrams SL, Wong EWT, Chang F, et al. Roles of the Raf/MEK/ERK pathway in cell growth, malignant transformation and drug resistance. Biochim. Biophys. Acta - Mol. Cell Res. 2007. p. 1263–84.
Jass JR. Classification of colorectal cancer based on correlation of clinical, morphological and molecular features. Histopathology. 2007. p. 113–30.
Guinney J, Dienstmann R, Wang X, De Reyniès A, Schlicker A, Soneson C, et al. The consensus molecular subtypes of colorectal cancer. Nat Med. 2015;21:1350–6.
Bläker H, Alwers E, Arnold A, Herpel E, Tagscherer KE, Roth W, et al. The association between mutations in BRAF and colorectal cancer–specific survival depends on microsatellite status and tumor stage. Clin Gastroenterol Hepatol. 2019;17:455–62 e6.
Atreya CE, Greene C, McWhirter RM, Ikram NS, Elaine Allen I, Van Loon K, et al. Differential radiographic appearance of BRAF v600e-mutant metastatic colorectal cancer in patients matched by primary tumor location. JNCCN J Natl Compr Cancer Netw. 2016;14:1536–43.
Scott RM, Smith ER. The authors reply. N Engl J Med. 2009;361:98.
Souglakos J, Philips J, Wang R, Marwah S, Silver M, Tzardi M, et al. Prognostic and predictive value of common mutations for treatment response and survival in patients with metastatic colorectal cancer. Br J Cancer. 2009;101:465–72.
Van Cutsem E, Köhne CH, Láng I, Folprecht G, Nowacki MP, Cascinu S, et al. Cetuximab plus irinotecan, fluorouracil, and leucovorin as first-line treatment for metastatic colorectal cancer: updated analysis of overall survival according to tumor KRAS and BRAF mutation status. J Clin Oncol. 2011;29:2011–9.
Bokemeyer C, Bondarenko I, Hartmann JT, de Braud F, Schuch G, Zubel A, et al. Efficacy according to biomarker status of cetuximab plus FOLFOX-4 as first-line treatment for metastatic colorectal cancer: the OPUS study. Ann Oncol. 2011;22:1535–46.
Ince WL, Jubb AM, Holden SN, Holmgren EB, Tobin P, Sridhar M, et al. Association of k-ras, b-raf, and p53 status with the treatment effect of bevacizumab. J Natl Cancer Inst. 2005;97:981–9.
Richman SD, Seymour MT, Chambers P, Elliott F, Daly CL, Meade AM, et al. KRAS and BRAF mutations in advanced colorectal cancer are associated with poor prognosis but do not preclude benefit from oxaliplatin or irinotecan: results from the MRC FOCUS trial. J Clin Oncol. 2009;27:5931–7.
Lim DR, Kuk JK, Kim T, Shin EJ. Comparison of oncological outcomes of right-sided colon cancer versus left-sided colon cancer after curative resection. Med (United States). 2017;96.
Petrelli F, Tomasello G, Borgonovo K, Ghidini M, Turati L, Dallera P, et al. Prognostic survival associated with left-sided vs right-sided colon cancer a systematic review and meta-analysis. JAMA Oncol. 2017. p. 211–9.
Popat S, Hubner R, Houlston RS. Systematic review of microsatellite instability and colorectal cancer prognosis. J. Clin. Oncol. 2005. p. 609–18.
Phipps AI, Buchanan DD, Makar KW, Burnett-Hartman AN, Coghill AE, Passarelli MN, et al. BRAF mutation status and survival after colorectal cancer diagnosis according to patient and tumor characteristics. Cancer Epidemiol Biomarkers Prev. 2012;21:1792–8.
Yamauchi M, Morikawa T, Kuchiba A, Imamura Y, Qian ZR, Nishihara R, et al. Assessment of colorectal cancer molecular features along bowel subsites challenges the conception of distinct dichotomy of proximal versus distal colorectum. Gut. 2012;61:847–54.
Consorti F, Lorenzotti A, Midiri G, Di Paola M. Prognostic significance of mucinous carcinoma of colon and rectum: a prospective case-control study. J Surg Oncol. United States. 2000;73:70–4.
Shin US, Yu CS, Kim JH, Kim TW, Lim SB, Yoon SN, et al. Mucinous rectal cancer: effectiveness of preoperative chemoradiotherapy and prognosis. Ann Surg Oncol. United States. 2011;18:2232–9.
Heinemann V, Modest DP, Fischer von Weikersthal L, Decker T, Kiani A, Vehling-Kaiser U, et al. Gender and tumor location as predictors for efficacy: influence on endpoints in first-line treatment with FOLFIRI in combination with cetuximab or bevacizumab in the AIO KRK 0306 (FIRE3) trial. J Clin Oncol [Internet]. American Society of Clinical Oncology; 2014;32:3600–3600. Available from: https://doi.org/10.1200/jco.2014.32.15_suppl.3600
Chapman PB, Hauschild A, Robert C, Haanen JB, Ascierto P, Larkin J, et al. Improved survival with vemurafenib in melanoma with BRAF V600E mutation. N Engl J Med. 2011;364:2507–16.
Corcoran RB, Falchook GS, Infante JR, Hamid O, Messersmith WA, Kwak EL, et al. BRAF V600 mutant colorectal cancer (CRC) expansion cohort from the phase I/II clinical trial of BRAF inhibitor dabrafenib (GSK2118436) plus MEK inhibitor trametinib (GSK1120212). J. Clin. Oncol. 2012. p. 3528–3528.
Cremolini C, Loupakis F, Antoniotti C, Lupi C, Sensi E, Lonardi S, et al. FOLFOXIRI plus bevacizumab versus FOLFIRI plus bevacizumab as first-line treatment of patients with metastatic colorectal cancer: updated overall survival and molecular subgroup analyses of the open-label, phase 3 TRIBE study. Lancet Oncol. 2015;16:1306–15.
Barras D, Missiaglia E, Wirapati P, Sieber OM, Jorissen RN, Love C, et al. BRAF V600E mutant colorectal cancer subtypes based on gene expression. Clin Cancer Res. 2017;23:104–15.
Oliveras-Ferraros C, Vazquez-Martin A, Cufí S, Queralt B, Báez L, Guardeño R, et al. Stem cell property epithelial-to-mesenchymal transition is a core transcriptional network for predicting cetuximab (erbituxTM) efficacy in KRAS wild-type tumor cells. J Cell Biochem. 2011;112:10–29.
Bruera G, Pepe F, Malapelle U, Pisapia P, Dal Mas A, Di Giacomo D, et al. KRAS, NRAS and BRAF mutations detected by next generation sequencing, and differential clinical outcome in metastatic colorectal cancer (MCRC) patients treated with first line FIr-B/ FOx adding bevacizumab (BEV) to triplet chemotherapy. Oncotarget. 2018;9:26279–90.
Malapelle U, Vigliar E, Sgariglia R, Bellevicine C, Colarossi L, Vitale D, et al. Ion Torrent next-generation sequencing for routine identification of clinically relevant mutations in colorectal cancer patients. J Clin Pathol. 2015;68:64–8.
Malapelle U, Pisapia P, Sgariglia R, Vigliar E, Biglietto M, Carlomagno C, et al. Less frequently mutated genes in colorectal cancer: Evidences from next-generation sequencing of 653 routine cases. J Clin Pathol. 2016;69:767–71.
Bruera G, Cannita K, Tessitore A, Russo A, Alesse E, Ficorella C, et al. The prevalent KRAS exon 2 c.35 G > A mutation in metastatic colorectal cancer patients: a biomarker of worse prognosis and potential benefit of bevacizumab-containing intensive regimens? Crit. Rev. Oncol. Hematol. 2015. p. 190–202.
Di Nicolantonio F, Martini M, Molinari F, Sartore-Bianchi A, Arena S, Saletti P, et al. Wild-type BRAF is required for response to panitumumab or cetuximab in metastatic colorectal cancer. J Clin Oncol. 2008
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
The study was approved by the local ethics committee of Afyonkarahisar Health Sciences University
Study Data
Available.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Beypinar, I., Demir, H., Sakin, A. et al. The Real-Life Data of BRAF Mutation on the Treatment of Colorectal Cancer: a TOG Study. J Gastrointest Canc 52, 932–939 (2021). https://doi.org/10.1007/s12029-020-00514-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12029-020-00514-9