Abstract
Purpose
In Japan, two courses of CDDP+5-FU (CF) therapy followed by surgery are accepted as a standard treatment for stage II/III esophageal cancer (EC) based on the results of the JCOG9907 trial. To gain a better survival, benefit especially for stage III patients in comparison with CF therapy, a three-arm phase III trial (neoadjuvant setting: CF vs. CF + radiation vs. DOC+CF [DCF]) is ongoing. We have aggressively performed DCF therapy for stage III or IV patients since October 2014. We herein review the outcomes of DCF therapy.
Methods
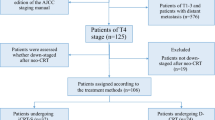
We retrospectively reviewed the cases of 27 patients with stage III or IV EC (male, n = 24; female, n = 3; median age, 70.0 years) who received DCF therapy.
Results
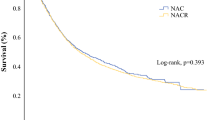
The response rate was 48.1%. Downstaging was achieved over the course of treatment in 14 patients (51.9%). Twenty-six patients transitioned to surgery, with 25 receiving R0 resection. DCF-treated patients who achieved downstaging showed significantly longer relapse-free survival (RFS) than those without downstaging (p = 0.0002). DCF-treated patients with a grade ≥ 1b histological effect showed significantly longer RFS than those with a grade < 1b effect (p = 0.0282). The multivariate analysis showed that downstaging was the only factor significantly associated with RFS in DCF-treated patients.
Conclusions
DCF therapy for stage ≥ III esophageal carcinoma is both feasible and effective. These findings suggest that downstaging and the histological effect might predict the effects of DCF therapy for EC.






Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this published article [and its supplementary information files].
References
Kitagawa Y, Uno T, Oyama T, Kato K, Kato H, Kawakubo H, et al. Esophageal cancer practice guidelines 2017 edited by the Japan esophageal society: part 1. Esophagus. 2018;16:1–24. https://doi.org/10.1007/s10388-018-0641-9.
Kuwano H, Nishimura Y, Oyama T, Kato H, Kitagawa Y, Kusano M, et al. Guidelines for diagnosis and treatment of carcinoma of the esophagus April 2012 edited by the Japan esophageal society. Esophagus. 2015;12:1–30.
Kitagawa Y, Uno T, Oyama T, et al. Esophageal cancer practice guidelines 2017 edited by the Japan esophageal society: part 2. Esophagus. 2018;16:25–43. https://doi.org/10.1007/s10388-018-0642-8.
Ando N, Iizuka T, Ide H, Ishida K, et al. Surgery plus chemotherapy compared with surgery alone for localized squamous cell carcinoma of the thoracic esophagus: a Japan clinical oncology group study-JCOG9204. J Clin Oncol. 2003;2:4592–6.
Ando N, Kato H, Igaki H, Shinoda M, Ozawa S, Shimizu H, et al. A randomized trial comparing postoperative adjuvant chemotherapy with Cisplatin and 5-fluorouracil versus preoperative chemotherapy for localized advanced squamous cell carcinoma of the thoracic esophagus (JCOG9907). Ann Surg Oncol. 2012;19:68–74.
Ando N. Progress in multidisciplinary treatment for esophageal cancer in Japan as reflected in JCOG studies. Esophagus. 2011;8:151–7.
Nakamura K, Kato K, Igaki H, et al. Three-arm phase III trial comparing cisplatin plus 5-FU (CF) versus docetaxel, cisplatin plus 5-FU (DCF) versus radiotherapy with CF (CF-RT) as preoperative therapy for locally advanced esophageal cancer (JCOG1109, NExT Study). Jpn J Clin Oncol. 2013;43:752–5.
Hara H, Tahara M, Daiko H, et al. Phase II feasibility study of preoperative chemotherapy with docetaxel, cisplatin, and fluorouracil for esophageal squamous cell carcinoma. Cancer Sci. 2013;4:1455–60.
Nomura M, Oze I, Abe T, et al. Impact of docetaxel in addition to cisplatin and fluorouracil as neoadjuvant treatment for resectable stage III or T3 esophageal cancer: a propensity score-matched analysis. Cancer Chemother Pharmacol. 2015;76:357–63.
Yamasaki M, Yasuda T, Yano M, et al. Multicenter randomized phase II study of cisplatin and fluorouracil plus docetaxel (DCF) compared with cisplatin and fluorouracil plus Adriamycin (ACF) as preoperative chemotherapy for resectable esophageal squamous cell carcinoma (OGSG1003). Ann Oncol. 2017;28:116–20.
Osaka Y, Shinohara M, Hoshino S, et al. Phase II study of combined chemotherapy with docetaxel, CDDP and 5-FU for highly advanced esophageal cancer. Anticancer Res. 2011;31:633–8.
Hironaka S, Tsubosa H, Mizusawa J, et al. Phase I/II trial of 2-weekly docetaxel combined with cisplatin plus fluorouracil in metastatic esophageal cancer (JCOG0807). Cancer Sci. 2014;105:1189–95.
Kobayashi K, Yamaguchi S, Fujita T, et al. DCF (DOC+CDDP+5-FU) therapy led to curative resection in a patient with advanced esophageal carcinoma after the failure of CF therapy. J Oncol Res Treat. 2018;2:1 (open access).
Kidera Y, Kawakami H, Sakiyama T, et al. Risk factors for cisplatin-induced nephrotoxicity and potential of magnesium supplementation for renal protection. PLoS ONE. 2014;9(7):e101902.
Linot B, Augereau P, Breheret R, et al. Efficacy and safety of early G-CSF administration in patients with head and neck cancer treated by docetaxel-cisplatin and 5-fluorouracil (DCF protocol): a retrospective study. Support Care Cancer. 2014;22:2831–7.
Japan Esophageal Society, editor. Japanese classification of esophageal cancer, 11th edition: part I. Esophagus. 2017;14:1–36.
Japan Esophageal Society, editor. Japanese classification of esophageal cancer, 11th edition: part ii, III. Esophagus. 2017;14:37–65.
Yoshimoto T, Kobayashi S, Kanetaka K, et al. Preoperative chemotherapy with docetaxel, cisplatin, and 5-fluorouracil for locally advanced esophageal carcinosarcoma: a case report and review of the literature. Surgical Case Reports. 2018. https://doi.org/10.1186/s40792-018-0425-4.
Kato K, Chul-Cho B, Takahashi M, Okada M, Lin C-Y, et al. Nivolumab versus chemotherapy in patients with advanced oesophageal squamous cell carcinoma refractory or intolerant to previous chemotherapy (ATTRACTION-3): a multicentre, randomised, open-label, phase 3 trial. Lancet Oncol. 2019;20:1506–17.
Akiyama Y, Sasaki A, Endo F, et al. Outcomes of esophagectomy after chemotherapy with biweekly docetaxel plus cisplatin and fluorouracil for advanced esophageal cancer: a retrospective cohort analysis. World J Surg Oncol. 2018;16:122. https://doi.org/10.1186/s12957-018-1420-8.
Hashimoto H, Abe M, Nakao M. et al., A randomized, double-blind, placebo-controlled phase III trial evaluating olanzapine 5 mg combined with standard antiemetic therapy for the prevention of chemotherapy-induced nausea and vomiting in patients receiving cisplatin-based chemotherapy (J-FORCE study). J Clin Oncol. 2019, 2019; ASCO proceedings abstr 11503.
Acknowledgments
We thank Dr. Brian Quinn of Japan Medical Communication for his assistance with the writing of this manuscript.
Code Availability
Software application (mac OS high Sierra ver. 10.13.6), Word (for text and tables), Power Point (for figures) ver. 16.16.20, and Stat View J ver. 5.0 (for Kaplan Meier curve).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflicts of interest.
Ethical Approval
This study is the part of an observation study on the treatment of esophageal cancer in our institution and has received approval from the internal review board of Nagasaki University Hospital (2019/10/22 IRB No. 19102114).
Consent to Participate
Informed consent are given by all the patients according to this observational study manual, and information of this study is demonstrated in our hospital home page for opt out patients.
Consent for Publication
Additional informed consent was obtained from all individual participants for whom identifying information is included in this article.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kobayashi, K., Kanetaka, K., Yoneda, A. et al. Downstaging and Histological Effects Might Be Reliable Predictors of the Efficacy of DOC+CDDP+5-FU (DCF) as Neoadjuvant Therapy for Stage III or Borderline Resectable Esophageal Cancer: a Single Institute Experience. J Gastrointest Canc 52, 582–592 (2021). https://doi.org/10.1007/s12029-020-00431-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12029-020-00431-x