Abstract
Purpose
Colon cancer is predominantly a disease of older adults. Studies determining the influence of age on outcomes of colon cancer have conflicting results. We aim to determine the long-term outcomes and utilisation of adjuvant chemotherapy of older adults compared with younger adults who had had a resection of a primary colon cancer.
Methods
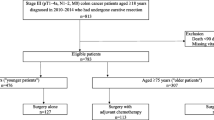
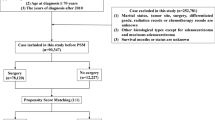
Consecutive patients who had resection of a primary colon cancer between January 1, 2000 and December 31, 2010 were identified from a prospective database and stratified into three age groups: ≤ 69 years, 70 to 79 years, and ≥ 80 years. Age-related differences in patients, cancer, and treatment characteristics were determined by chi-square tests. Five-year overall survival and cancer-specific survival were determined by Kaplan-Meier method and by multivariable Cox regression analysis to adjust for potential confounding factors.
Results
Of 1135 included patients, 469 (41%) patients were aged ≤ 69 years, 382 (34%) were 70–79 years, and 284 (25%) were ≥ 80 years. Increasing age group predicted more comorbidity (p < 0.001), cardiac comorbidity (p < 0.001), right-sided cancers (p < 0.001), and less adjuvant chemotherapy (stage III only; p < 0.001). Increasing age group was associated with worse overall survival by stage (p < 0.001) but not cancer-specific survival by stage (p = 0.83). Adjuvant chemotherapy in patients with stage III colon cancer independently predicted improved overall survival (p < 0.001) and cancer-specific survival (p = 0.01).
Conclusions
Compared with younger adults, older adults with colon cancer had worse survival outcomes and received less adjuvant chemotherapy.






Similar content being viewed by others
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424. https://doi.org/10.3322/caac.21492.
Australian Institute of Health and Welfare. Colon Cancer ACIM. https://www.aihw.gov.au/reports/cancer/cancer-data-in-australia/acim-books. 2018 Accessed 16 May 2019
Audisio RA, Veronesi P, Ferrario L, Cipolla C, Andreoni B, Aapro M. Elective surgery for gastrointestinal tumours in the elderly. Ann Oncol. 1997;8(4):317–26.
Patel SS, Nelson R, Sanchez J, Lee W, Uyeno L, Garcia-Aguilar J, et al. Elderly patients with colon cancer have unique tumor characteristics and poor survival. Cancer. 2013;119(4):739–47. https://doi.org/10.1002/cncr.27753.
Kamran SC, Clark JW, Zheng H, Borger DR, Blaszkowsky LS, Allen JN, et al. Primary tumor sidedness is an independent prognostic marker for survival in metastatic colorectal cancer: results from a large retrospective cohort with mutational analysis. Cancer Med. 2018;7(7):2934–42. https://doi.org/10.1002/cam4.1558.
Loupakis F, Yang D, Yau L, Feng S, Cremolini C, Zhang W, et al. Primary tumor location as a prognostic factor in metastatic colorectal cancer. J Natl Cancer Inst. 2015;107(3). https://doi.org/10.1093/jnci/dju427.
Marusch F, Koch A, Schmidt U, Zippel R, Gastmeier J, Ludwig K, et al. Impact of age on the short-term postoperative outcome of patients undergoing surgery for colorectal carcinoma. Int J Color Dis. 2002;17(3):177–84.
Kotake K, Asano M, Ozawa H, Kobayashi H, Sugihara K. Tumour characteristics, treatment patterns and survival of patients aged 80 years or older with colorectal cancer. Color Dis. 2015;17(3):205–15. https://doi.org/10.1111/codi.12826.
Baquero GA, Rich MW. Perioperative care in older adults. J Geriatr Cardiol. 2015;12(5):465–9. https://doi.org/10.11909/j.issn.1671-5411.2015.05.018.
McNicol L, Story DA, Leslie K, Myles PS, Fink M, Shelton AC, et al. Postoperative complications and mortality in older patients having non-cardiac surgery at three Melbourne teaching hospitals. Med J Aust. 2007;186(9):447–52.
Sieber FE, Barnett SR. Preventing postoperative complications in the elderly. Anesthesiol Clin. 2011;29(1):83–97. https://doi.org/10.1016/j.anclin.2010.11.011.
Crombag MR, Joerger M, Thurlimann B, Schellens JH, Beijnen JH, Huitema AD. Pharmacokinetics of selected anticancer drugs in elderly cancer patients: focus on breast cancer. Cancers (Basel). 2016;8(1). https://doi.org/10.3390/cancers8010006.
Hurria A, Dale W, Mooney M, Rowland JH, Ballman KV, Cohen HJ, et al. Designing therapeutic clinical trials for older and frail adults with cancer: U13 conference recommendations. J Clin Oncol. 2014;32(24):2587–94. https://doi.org/10.1200/jco.2013.55.0418.
Moth EB, Vardy J, Blinman P. Decision-making in geriatric oncology: systemic treatment considerations for older adults with colon cancer. Expert Rev Gastroenterol Hepatol. 2016;10(12):1321–40. https://doi.org/10.1080/17474124.2016.1244003.
Kune GA, Kune S, Field B, White R, Brough W, Schellenberger R, et al. Survival in patients with large-bowel cancer. A population-based investigation from the Melbourne Colorectal Cancer Study. Dis Colon Rectum. 1990;33(11):938–46.
McMillan DC, McArdle CS. The impact of young age on cancer-specific and non-cancer-related survival after surgery for colorectal cancer: 10-year follow-up. Br J Cancer. 2009;101:557–60. https://doi.org/10.1038/sj.bjc.6605222.
Devon KM, Vergara-Fernandez O, Victor JC, McLeod RS. Colorectal cancer surgery in elderly patients: presentation, treatment, and outcomes. Dis Colon Rectum. 2009;52(7):1272–7. https://doi.org/10.1007/DCR.0b013e3181a74d2e.
Chandrasinghe PC, Ediriweera DS, Nazar T, Kumarage S, Hewavisenthi J, Deen KI. Overall survival of elderly patients having surgery for colorectal cancer is comparable to younger patients: results from a South Asian population. Gastroenterol Res Pract. 2017;2017:9670512. https://doi.org/10.1155/2017/9670512.
Dekker JW, van den Broek CB, Bastiaannet E, van de Geest LG, Tollenaar RA, Liefers GJ. Importance of the first postoperative year in the prognosis of elderly colorectal cancer patients. Ann Surg Oncol. 2011;18(6):1533–9. https://doi.org/10.1245/s10434-011-1671-x.
Jessup JM, McGinnis LS, Steele GD Jr, Menck HR, Winchester DP. The National Cancer Data Base. Report on colon cancer. Cancer. 1996;78(4):918–26. https://doi.org/10.1002/(sici)1097-0142(19960815)78:4<918::aid-cncr32>3.0.co;2-w.
Holt PR, Kozuch P, Mewar S. Colon cancer and the elderly: from screening to treatment in management of GI disease in the elderly. Best Pract Res Clin Gastroenterol. 2009;23(6):889–907. https://doi.org/10.1016/j.bpg.2009.10.010.
Wilkinson GR. Drug metabolism and variability among patients in drug response. N Engl J Med. 2005;352(21):2211–21. https://doi.org/10.1056/NEJMra032424.
Widdison AL, Barnett SW, Betambeau N. The impact of age on outcome after surgery for colorectal adenocarcinoma. Ann R Coll Surg Engl. 2011;93(6):445–50. https://doi.org/10.1308/003588411x587154.
Kennecke HF, Yin Y, Davies JM, Speers CH, Cheung WY, Lee-Ying R. Prognostic effect of sidedness in early stage versus advanced colon cancer. Health Science Reports. 2018;1(8):e54. https://doi.org/10.1002/hsr2.54.
Petrelli F, Tomasello G, Borgonovo K, et al. Prognostic survival associated with left-sided vs right-sided colon cancer: a systematic review and meta-analysis. JAMA Oncology. 2017;3(2):211–9. https://doi.org/10.1001/jamaoncol.2016.4227.
Iacopetta B, Grieu F, Amanuel B. Microsatellite instability in colorectal cancer. Asia Pac J Clin Oncol. 2010;6(4):260–9. https://doi.org/10.1111/j.1743-7563.2010.01335.x.
Sarfati D, Hill S, Blakely T, Robson B, Purdie G, Dennett E, et al. The effect of comorbidity on the use of adjuvant chemotherapy and survival from colon cancer: a retrospective cohort study. BMC Cancer. 2009;9:116. https://doi.org/10.1186/1471-2407-9-116.
Sarfati D, Tan L, Blakely T, Pearce N. Comorbidity among patients with colon cancer in New Zealand. N Z Med J. 2011;124(1338):76–88.
Tooth L, Hockey R, Byles J, Dobson A. Weighted multimorbidity indexes predicted mortality, health service use, and health-related quality of life in older women. J Clin Epidemiol. 2008;61(2):151–9. https://doi.org/10.1016/j.jclinepi.2007.05.015.
He X, Clarke SJ, McLachlan AJ. Clinical pharmacology of chemotherapy agents in older people with cancer. Curr Gerontol Geriatr Res. 2011;2011:628670. https://doi.org/10.1155/2011/628670.
Rieker RJ, Hammer E, Eisele R, Schmid E, Hogel J. The impact of comorbidity on the overall survival and the cause of death in patients after colorectal cancer resection. Langenbeck's Arch Surg. 2002;387(2):72–6. https://doi.org/10.1007/s00423-002-0291-0.
Søgaard M, Thomsen RW, Bossen KS, Sørensen HT, Nørgaard M. The impact of comorbidity on cancer survival: a review. Clin Epidemiol. 2013;5(Suppl 1):3–29. https://doi.org/10.2147/clep.s47150.
Nitsche U, Stöss C, Friess H. Effect of adjuvant chemotherapy on elderly colorectal cancer patients: lack of evidence. Gastrointest Tumors. 2017;4(1-2):11–9. https://doi.org/10.1159/000479318.
Burdette-Radoux S, Muss HB. Adjuvant chemotherapy in the elderly: whom to treat, what regimen? Oncologist. 2006;11(3):234–42. https://doi.org/10.1634/theoncologist.11-3-234.
Iwashyna TJ, Lamont EB. Effectiveness of adjuvant fluorouracil in clinical practice: a population-based cohort study of elderly patients with stage III colon cancer. J Clin Oncol. 2002;20(19):3992–8. https://doi.org/10.1200/jco.2002.03.083.
Sargent DJ, Goldberg RM, Jacobson SD, Macdonald JS, Labianca R, Haller DG, et al. A pooled analysis of adjuvant chemotherapy for resected colon cancer in elderly patients. N Engl J Med. 2001;345(15):1091–7. https://doi.org/10.1056/NEJMoa010957.
Hurria A, Siccion EP. Assessing the 'fit' older patient for chemotherapy. Oncology (Williston Park). 2014;28(7):594, 596, 598–9.
Lichtman SM, Villani G. Chemotherapy in the elderly: pharmacologic considerations. Cancer Control. 2000;7(6):548–56.
Millan M, Merino S, Caro A, Feliu F, Escuder J, Francesch T. Treatment of colorectal cancer in the elderly. World J Gastrointest Oncol. 2015;7(10):204–20. https://doi.org/10.4251/wjgo.v7.i10.204.
Field KM, Kosmider S, Jefford M, Michael M, Jennens R, Green M, et al. Chemotherapy dosing strategies in the obese, elderly, and thin patient: results of a nationwide survey. Journal of Oncology Practice. 2008;4(3):108–13. https://doi.org/10.1200/jop.0832001.
Acknowledgments
Authors would like to thank Professor Pierre Chapuis, Dr Owen Dent, and Gael Sinclair who assisted with the provision of the data from the Concord Repatriation General Hospital database on resected colorectal cancers.
Funding
MS received a Sydney Catalyst Postgraduate Research PhD Scholarship in 2018.
Author information
Authors and Affiliations
Contributions
Mohsen Shafiei contributed in data collection, statistical analysis, and writing of the manuscript. He is the corresponding author. Philip Beale and Prunella Blinman (lead author) contributed in study design, data validation, final writing, and editing of the manuscript.
Corresponding author
Ethics declarations
Authors’ Disclosures
Authors of this manuscript have no relevant financial or other relationships to disclose.
Conflicts of Interest
The authors declare that they have no conflict(s) of interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shafiei, M., Beale, P. & Blinman, P. Utilisation of Adjuvant Chemotherapy and 5-Year Survival Analysis of Prospectively Recorded Cohort Data for Older Adults Versus Younger Adults with Resected Primary Colon Cancer. J Gastrointest Canc 51, 988–997 (2020). https://doi.org/10.1007/s12029-019-00343-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12029-019-00343-5