Abstract
Purpose
Long noncoding RNA (lncRNA) has been identified as an important modulator of gene expression and other activities of the cells. This kind of genes also has a significant role in the growth and development of human cancers, including colorectal cancer (CRC). Among lncRNAs, thymopoietin (TMPO)-antisense RNA 1 (TMPO-AS1) is of particular significance and might have a role in CRC regulation. The current study aimed to determine the involvement of TMPO-AS1 in CRC patients.
Methods
In this study, 50 pairs of tumor and tumor marginal samples of CRC patients were investigated to assess the expression level of TMPO-AS1 in this cancer. For this purpose, the total RNA was isolated from the tissues using the TRIzol RNA extraction method, and after synthesis of the complementary DNA, the TMPO-AS1 expression was measured by quantitative real-time PCR (qRT-PCR) technique. In addition, clinicopathological characteristics of the CRC patients were analyzed in the study groups.
Results
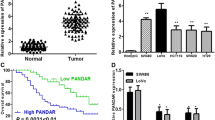
The findings demonstrated the overexpression of TMPO-AS1 in CRC tissues. Interestingly, the expression level of TMPO-AS1 was significantly correlated with clinicopathological features of the patients such as lymph node and distant metastasis.
Conclusion
The overexpression of TMPO-AS1 gene in CRC suggests that this lncRNA and its underlying signaling pathways can be considered a prognostic tumor marker and may pave the way for the future development of novel therapeutic options for CRC.


Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Siegel RL, Miller KD, Fedewa SA, Ahnen DJ, Meester RGS, Barzi A, et al. Colorectal cancer statistics, 2017. CA Cancer J Clin. 2017;67:177–93.
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-tieulent J, Jemal A. Global Cancer statistics, 2012. CA a cancer J Clin. 2015;65:87–108.
Shirafkan N, Mansoori B, Mohammadi A, Shomali N, Ghasbi M, Baradaran B. MicroRNAs as novel biomarkers for colorectal cancer: new outlooks. Biomed Pharmacother Elsevier. 2018;97:1319–30.
Haggar FA, Boushey RP. Colorectal cancer epidemiology: incidence, mortality, survival, and risk factors. Clin Colon Rectal Surg. 2009;22:191–7.
Sadreddini S, Safaralizadeh R, Baradaran B, Aghebati-Maleki L, Hosseinpour-Feizi MA, Shanehbandi D, et al. Chitosan nanoparticles as a dual drug/siRNA delivery system for treatment of colorectal cancer. Immunol Lett. Elsevier B.V.; 2017;181:79–86.
Hallaj-Nezhadi S, Valizadeh H, Dastmalchi S, Baradaran B, Jalali MB, Dobakhti F, et al. Preparation of chitosan-plasmid DNA nanoparticles encoding interleukin-12 and their expression in CT-26 colon carcinoma cells. J Pharm Pharm Sci. 2011;14:181–95.
Xie F, Xiang X, Huang Q, Ran P, Yuan Y, Li Q, et al. Reciprocal control of lncRNA-BCAT1 and β-catenin pathway reveals lncRNA-BCAT1 long non-coding RNA acts as a tumor suppressor in colorectal cancer. Oncotarget. 2017;8:23628–37.
Kazemzadeh M, Safaralizadeh R, Feizi MAH, Somi MH, Shokoohi B. Misregulation of the dependence receptor DCC and its upstream lincRNA, LOC100287225, in colorectal cancer. Tumori J. 2017;103:40–3.
Ponting CP, Oliver PL, Reik W. Evolution and functions of long noncoding RNAs. Cell. Elsevier Inc.; 2009;136:629–41.
Peng WX, Koirala P, Mo YY. LncRNA-mediated regulation of cell signaling in cancer. Oncogene. 2017;36:5661–7.
Jarroux J, Morillon A, Pinskaya M. History, discovery, and classification of lncRNAs. Adv Exp Med Biol. 2017. p. 1–46.
Brosnan CA, Voinnet O. The long and the short of noncoding RNAs. Curr Opin Cell Biol. 2009;21:416–25.
Huang W, Su X, Yan W, Kong Z, Wang D, Huang Y, et al. Overexpression of AR-regulated lncRNA TMPO-AS1 correlates with tumor progression and poor prognosis in prostate cancer. Prostate. 2018;78:1248–61.
Peng F, Wang R, Zhang Y, Zhao Z, Zhou W, Chang Z, et al. Differential expression analysis at the individual level reveals a lncRNA prognostic signature for lung adenocarcinoma. Mol Cancer Molecular Cancer. 2017;16:1–12.
Zhang L, Wang G, Chen S, Ding J, Ju S, Cao H, et al. Depletion of thymopoietin inhibits proliferation and induces cell cycle arrest/apoptosis in glioblastoma cells. World J Surg Oncol World Journal of Surgical Oncology. 2016;14:1–8.
Brachner A, Foisner R. Lamina-associated polypeptide (LAP)2α and other LEM proteins in cancer biology. 2014. p. 143–63.
Siahmansouri H, Somi MH, Babaloo Z, Baradaran B, Jadidi-Niaragh F, Atyabi F, et al. Effects of HMGA2 siRNA and doxorubicin dual delivery by chitosan nanoparticles on cytotoxicity and gene expression of HT-29 colorectal cancer cell line. J Pharm Pharmacol. 2016:1119–30.
Daryabari SS, Safaralizadeh R, Hosseinpourfeizi M, Moaddab Y, Shokouhi B. Overexpression of SSH1 in gastric adenocarcinoma and its correlation with clinicopathological features. J Gastrointest Oncol. 2018;9:728–33.
Djebali S, Davis CA, Merkel A, Dobin A, Lassmann T, Mortazavi A, et al. Landscape of transcription in human cells. Nature. 2012;489:101–8.
Ong MS, Cai W, Tan TZ, Huang RY, Hooi SC, Yap CT, et al. Long non-coding RNA landscape in colorectal cancer. Rna Dis. 2018;5:1–9.
Bhan A, Soleimani M, Mandal SS. Long noncoding RNA and cancer: a new paradigm. Cancer Res. 2017;77:3965–81.
Chen G, Wang Z, Wang D, Qiu C, Liu M, Chen X, et al. LncRNA disease: a database for long-non-coding RNA-associated diseases. Nucleic Acids Res. 2013;41:983–6.
Schmitt AM, Chang HY. Long noncoding rnas in cancer pathways. Cancer Cell. Elsevier Inc.; 2016;29:452–63.
Qin Z, Zheng X, Fang Y. Long noncoding RNA TMPO-AS1 promotes progression of non-small cell lung cancer through regulating its natural antisense transcript TMPO. Biochem Biophys Res Commun Elsevier Ltd. 2019;516:486–93.
Acknowledgments
The authors would like to thank the Immunology Research Center, Tabriz University of Medical Sciences, for providing facilities to carry out this work.
Author information
Authors and Affiliations
Contributions
HM, BB, DS, MA, and SH performed the experiments. BB provided biological materials and reagents. HM and KH wrote the article. HM, DS, MA, and KH performed data analysis. BB and RS participated in the design of the work. RS supervised the study.
Corresponding author
Ethics declarations
Written informed consent was obtained from all participants for their tissues to be utilized in this research. The study was approved by the ethical committee of Tabriz University of Medical Sciences, Tabriz, Iran.
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mohammadrezakhani, H., Baradaran, B., Shanehbandi, D. et al. Overexpression and Clinicopathological Correlation of Long Noncoding RNA TMPO-AS1 in Colorectal Cancer Patients. J Gastrointest Canc 51, 952–956 (2020). https://doi.org/10.1007/s12029-019-00333-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12029-019-00333-7