Abstract
Background
Fatty acid synthase (FAS) is a valuable lipid enzyme involved in lipid biosynthesis and suggested to contribute in tumor carcinogenesis. Vascular endothelial growth factor (VEGF) is considered a serious angiogenic growth factor in the angiogenic pathway which is a very important in tumor growth and metastasis. Thus, inhibition of lipid biosynthesis and tumor angiogenesis can be new goals for colorectal cancer (CRC) treatment.
Aim of the Work
The assessment of the expression of FAS and VEGF protein and the relationship between them in CRC with the clinicopathological parameters.
Methods
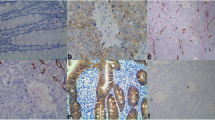
The present retrospective study included 63 paraffin blocks previously diagnosed as primary cases of CRC. The slides were subjected to FAS and VEGF immunohistochemical staining using a streptavidin-biotin-peroxidase. The relationships among FAS and VEGF expression and clinicopathological parameters were statistically analyzed.
Results
The expression rate of FAS was 81% and VEGF was 84.1% in the studied cases. FAS expression was significantly associated with histopathological type (p = 0.02) and grade (p = 0.04), and highly associated with lymph node metastasis and stage (p < 0.001).VEGF was significantly associated with histopathological type (p = 0.01) and tumor depth (p = 0.02); highly associated with grade, lymph node metastasis, and stage (p < 0.001). There was a positive association between FAS and VEGF expression in CRC (p < 0.001).
Conclusion
FAS and VEGF showed a highly significant expression in the studied primary CRC cases. A significant association was observed between their expressions, suggesting the involvement of FAS in tumor angiogenesis. So they constitute potential targets in cancer prevention and treatment and make FAS an attractive antiangiogenic target.


Similar content being viewed by others
References
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65(2):87–108. https://doi.org/10.3322/caac.21262.
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2017. CA Cancer J Clin. 2017;67(1):7–30. https://doi.org/10.3322/caac.21387.
Mokhtar N, Salama A, Badawy O, Khorshed E, Mohamed G, Ibrahim M, Abdelazim H. Gastrointestinal tract tumors. In: Cancer Pathology Registry. A 12-year Registry (2000–2001). 2016a. pp, 32–58.
Mokhtar N, Salama A, Badawy O, Khorshed E, Mohamed G, Ibrahim M, Abdelazim H. General features of total series. In: Cancer Pathology Registry. A 12-year Registry (2000–2001) 2016b. pp, 1–6.
Kuhajda FP. Fatty acid synthase and cancer : new application of an old pathway. Cancer Res. 2006;66(12):5977–81. https://doi.org/10.1158/0008-5472.CAN-05-4673.
Kusakabe T, Nashimoto A, Honma K, Suzuki T. Fatty acid synthase is highly expressed in carcinoma, adenoma and in regenerative epithelium and intestinal metaplasia of the stomach. Histopathol. 2002;40(1):71–9. https://doi.org/10.1046/j.1365-2559.2002.01289.x.
Piyathilake C, Frost A, Manne U, Bell W, Weiss H, Heimburger D, et al. The expression of fatty acid synthase (FASE) is an early event in the development and progression of squamous cell carcinoma of the lung. Hum Pathol. 2000;31(9):1068–73. https://doi.org/10.1053/hupa.2000.9842.
Alò PL, Visca P, Marci A, Mangoni A, Botti C, Di Tondo U. Expression of fatty acid synthase (FAS) as a predictor of recurrence in stage I breast carcinoma patients. Cancer. 1996;77(3):474–82. https://doi.org/10.1002/(SICI)1097-0142(19960201)77:3<474::AID-CNCR8>3.0.CO;2-K.
Epstein JI, Carmichael M, Partin AW. OA-519 (fatty acid synthase) as an independent predictor of pathological stage in adenocarcinoma of the prostate. Urology. 1995;45(1):81–6. https://doi.org/10.1016/S0090-4295(95)96904-7.
Dvorak HF, Weaver VM, Tlsty TD, Bergers G. Tumor microenvironment and progression. J Surg Oncol. 2011;103(6):468–74. https://doi.org/10.1002/jso.21709.
Joyce JA, Pollard JW. Microenvironmental regulation of metastasis. Nat Rev Cancer. 2009;9(4):239–52. https://doi.org/10.1038/nrc2618.
Potente M, Gerhardt H, Carmeliet P. Basic and therapeutic aspects of angiogenesis. Cell. 2011;146(6):873–87. https://doi.org/10.1016/j.cell.2011.08.039.
Nowak DG, Woolard J, Amin EM, Konopatskaya O, Saleem MA, Churchill AJ, et al. Expression of pro- and anti-angiogenic isoforms of VEGF is differentially regulated by splicing and growth factors. J Cell Sci. 2008;121(Pt 20):3487–95. https://doi.org/10.1242/jcs.016410.
Lau T, Roslani A, Lian L, Lee P, Hilmi I, Goh K, et al. Association between EGF and VEGF functional polymorphisms and sporadic colorectal cancer in the Malaysian population. Genet Mol Res. 2014;13(3):5555–61. https://doi.org/10.4238/2014.July.25.9.
Hansen TF, Jakobsen A. Clinical implications of genetic variations in the VEGF system in relation to colorectal cancer. Pharmacogenomics. 2011;12(12):1681–93. https://doi.org/10.2217/pgs.11.118.
Jannuzzi A, Özhan G, Yanar H, Alpertunga B. VEGF gene polymorphisms and susceptibility to colorectal cancer. Genet Test Mol Biomarkers. 2015;19(3):133–7. https://doi.org/10.1089/gtmb.2014.0259.
Hamilton SR, Rubio CA, Vogelstein B, Sobin LH, Kudo S, Fogt F. Carcinoma of the colon and rectum. In: Bosman FT, Carneiro F, Hruban RH, Theise ND, editors. WHO classification of tumours of the digestive system. 4th ed. IARC: Lyon; 2010. p. 134–46.
Wang H, Xi Q, Wu G. Fatty acid synthase regulates invasion and metastasis of colorectal cancer via Wnt signaling pathway. Cancer Med. 2016;5(7):1599–606. https://doi.org/10.1002/cam4.711.
Hafez N, Tahoun N. Expression of cyclooxygenase 2 and vascular endothelial growth factor in gastric carcinoma: relationship with clinicopathological parameters. J Egypt Natl Canc Inst. 2016;28(3):149–56. https://doi.org/10.1016/j.jnci.2016.05.005.
Zaytseva YY, Elliott VA, Rychahou P, Mustain WC, Kim JT, Valentino J, et al. Cancer cell-associated fatty acid synthase activates endothelial cells and promotes angiogenesis in colorectal cancer. Carcinogenesis. 2014;35(6):1341–51. https://doi.org/10.1093/carcin/bgu042.
Zaytseva YY, Rychahou PG, Gulhati P, Elliott VA, Mustain WC, O’Connor K, et al. Inhibition of fatty acid synthase attenuates CD44-associated signaling and reduces metastasis in colorectal cancer. Cancer Res. 2012;72(6):1504–17. https://doi.org/10.1158/0008-5472.CAN-11-4057.
Kearney KE, Pretlow TG, Pretlow TP. Increased expression of fatty acid synthase in human aberrant crypt foci: possible target for colorectal cancer prevention. Int J Cancer. 2009;125:249–52. https://doi.org/10.1002/ijc.24356.
Visca P, Alo PL, Nonno FD, Botti C, Trombetta G, Marandino F, et al. Immunohistochemical expression of fatty acid synthase, apoptotic-regulating genes, proliferating factors, and ras protein product in colorectal adenomas, carcinomas, and adjacent nonneoplastic mucosa. Clin Cancer Res. 1999;5(12):4111–8.
Ogino S, Nosho K, Meyerhardt JA, Kirkner GJ, Chan AT, Kawasaki T, et al. Cohort study of fatty acid synthase expression and patient survival in colon cancer. J Clin Oncol. 2008;26:5713–20. https://doi.org/10.1200/JCO.2008.18.2675.
Visca P, Sebastiani V, Botti C, Diodoro MG, Lasagni RP, Romagnoli F, et al. Fatty acid synthase (FAS) is a marker of increased risk of recurrence in lung carcinoma. Anticancer Res. 2004;24:4169–73.
Uddin S, Hussain AR, Ahmed M, Abubaker J, Al-Sanea N, Abduljabbar A, et al. High prevalence of fatty acid synthase expression in colorectal cancers in Middle Eastern patients and its potential role as a therapeutic target. Am J Gastroenterol. 2009;104(7):1790–801. https://doi.org/10.1038/ajg.2009.230.
Rashid A, Pizer ES, Moga M, Milgraum LZ, Zahurak M, Pasternack GR, et al. Elevated expression of fatty acid synthase and fatty acid synthetic activity in colorectal neoplasia. Am J Pathol. 1997;150:201–8.
Zhan Y, Ginanni N, Tota MR, Wu M, Bays NW, Richon VM, et al. Control of cell growth and survival by enzymes of the fatty acid synthesis pathway in HCT-116 colon cancer cells. Clin Cancer Res. 2008;14(18):5735–42. https://doi.org/10.1158/1078-0432.CCR-07-5074.
Li JN, Gorospe M, Chrest FJ, Kumaravel TS, Evans MK, Han WF, et al. Pharmacological inhibition of fatty acid synthase activity produces both cytostatic and cytotoxic effects modulated by p53. Cancer Res. 2001;61(4):1493–9.
Kridel SJ, Axelrod F, Rozenkrantz N, Smith JW. Orlistat is a novel inhibitor of fatty acid synthase with antitumor activity. Cancer Res. 2004;64(6):2070–5.
Alli PM, Pinn ML, Jaffee EM, McFadden JM, Kuhajda FP. Fatty acid synthase inhibitors are chemopreventive for mammary cancer in neu-N transgenic mice. Oncogene. 2005;24(1):39–46. https://doi.org/10.1038/sj.onc.1208174.
Little JL, Wheeler FB, Fels DR, Koumenis C, Kridel SJ. Inhibition of fatty acid synthase induces endoplasmic reticulum stress in tumor cells. Cancer Res. 2007;67(3):1262–9. https://doi.org/10.1158/0008-5472.CAN-06-1794.
Graner E, Tang D, Rossi S, Baron A, Migita T, Weinstein LJ, et al. The isopeptidasUSP2a regulates the stability of fatty acid synthase in prostate cancer. Cancer Cell. 2004;5(3):253–61.
Menendez JA, Lupu R, Colomer R. Inhibition of tumor-associated fatty acid synthase hyperactivity induces synergistic chemosensitization of HER-2/neu-overexpressing human breast cancer cells to docetaxel (taxotere). Breast Cancer Res Treat. 2004;84:183–95. https://doi.org/10.1023/B:BREA.0000018409.59448.60.
Furuta E, Pai SK, Zhan R, Bandyopadhyay S, Watabe M, MoYY HS, et al. Fatty acid synthase gene is up-regulated by hypoxia via activation of Akt and sterol regulatory element binding protein-1. Cancer Res. 2008;68(4):1003–11. https://doi.org/10.1158/0008-5472.CAN-07-2489.
Zhao HC, Qin R, Chen XX, Sheng X, Wu JF, Wang DB, et al. Microvessel density is a prognostic marker of human gastric cancer. World J Gastroenterol. 2006;12(47):7598–603. https://doi.org/10.3748/wjg.v12.i47.7598.
Zhao ZQ, Yang S, Lu HS. Expression of midkine and vascular endothelial growth factor in gastric cancer and the association of high levels with poor prognosis and survival. Mol Med Rep. 2012;5(2):415–9. https://doi.org/10.3892/mmr.2011.649.
Xu T, Chen D, Chen J. Expression of vascular endothelial growth factor C and its correlation with lymph node metastasis in colorectal carcinoma. J Huazhong Univ Sci Technolog Med Sci. 2004;24(6):596–8.
Vahedi L, Ghasemi M, Yazdani J, Mehrabian-Fard M, Sajadi N. Evaluation of VEGF immunohistochemical expression and correlation with clinicopathologic features in colorectal Cancer. Govaresh. 2015;20(3):199–204.
Soumaoro LT, Uetake H, Takagi Y, Iida S, Higuchi T, Yasuno M, et al. Coexpression of VEGF-C and Cox-2 in human colorectal cancer and its association with lymph node metastasis. Dis Colon Rectum. 2006;49(3):392–8. https://doi.org/10.1007/s10350-005-0247-x.
Xu Q, Chen RX, Wang ZW, Ni QC, Qian JJ, Sheng HX. Clinical research on angiogenesis in colorectal carcinoma and expression of CK20 mRNA in peripheral blood. Zhonghua Yi Xue Za Zhi. 2005;85(17):1205–8. https://doi.org/10.3760/j:issn:0376-2491.2005.17.015.
Mohamed HAD, Abd El All HS, Kamel AA, Yossef WT, Hammam MM. Correlation of vascular endothelial growth factor expression and neovascularization with colorectal carcinoma: a pilot study. J Adenocarcinoma. 2016;1:1–5. https://doi.org/10.21767/2572-309X.10005.
Jia Y, Li Z, He Y, Liang W, Yang H, Ma H. Expression of vascular endothelial growth factor-C and the relationship between lymphangiogenesis and lymphatic metastasis in colorectal cancer. World J Gastroenterol. 2004;10(22):3261–3. https://doi.org/10.3748/wjg.v10.i22.3261.
Zheng S, Han MY, Xiao ZX, Peng JP, Dong Q. Clinical significance of vascular endothelial growth factor expression and neovascularization in colorectal carcinoma. World J Gastroenterol. 2003;9(6):1227–30. https://doi.org/10.3748/wjg.v9.i6.1227.
Wen L, Wang R, Lu X, You C. Expression and clinical significance of vascular endothelial growth factor and fms-related tyrosine kinase 1 in colorectal cancer. Oncol Lett. 2015;9(5):2414–8. https://doi.org/10.3892/ol.2015.3013.
Hedaya M, Helmy A, H E, Hammam O. Cyclo-oxygenase-2 and vascular endothelial growth factor expression in colorectal cancer patients. The Egyptian Journal of Surgery. 2015;34(1):35–40. https://doi.org/10.4103/1110-1121.153369.
van Triest B, Pinedo HM, Blaauwgeers JL, van Diest PJ, Schoenmakers PS, Voorn DA, et al. Prognostic role of thymidylate synthase, thymidine phosphorylase/platelet-derived endothelial cell growth factor and proliferation markers in colorectal cancer. Clin Cancer Res. 2000;6(3):1063–72.
Hashim AF, Al-Janabi AA, Mahdi LH, Al-Toriahi KM, Yasseen AA. Vascular endothelial growth factor (VEGF) receptor expression correlates with histologic grade and stage of colorectal cancer. Libyan J Med. 2010;5(1):5059. https://doi.org/10.3402/ljm.v5i0.5059.
Minagawa N, Nakayama Y, Hirata K, Onitsuka K, Inoue Y, Nagata N, et al. Correlation of plasma level and immunohistochemical expression of vascular endothelial growth factor in patients with advanced colorectal cancer. Anticancer Res. 2002;22(5):2957–63.
White JD, Hewett PW, Kosuge D, McCulloch T, Enholm BC, Carmichael J, et al. Vascular endothelial growth factor-D expression is an independent prognostic marker for survival in colorectal carcinoma. Cancer Res. 2002;62(6):1669–75.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Mohamed, A.H., Said, N.M. Immunohistochemical Expression of Fatty Acid Synthase and Vascular Endothelial Growth Factor in Primary Colorectal Cancer: a Clinicopathological Study. J Gastrointest Canc 50, 485–492 (2019). https://doi.org/10.1007/s12029-018-0104-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12029-018-0104-5