Abstract
Purpose
Defects in DNA repair pathways have been linked with colorectal cancer (CRC). Adjuvant radiotherapy has become commonplace in the treatment of rectal cancer however it is associated with a higher rate of second cancer formation. It is known that radiation results in DNA damage directly or indirectly by radiation-induced bystander effect (RIBE) by causing double-strand breaks (DSBs). The majority of work in RIBE has been performed in cell lines and limited studies have been in or ex vivo.
Methods
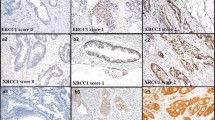
The first study aim was to examine by immunohistochemistry, levels of DSB (expression of the protein MRE11) in normal colonic tissue outside the irradiated field post neo-adjuvant radiotherapy (group 1). These levels were compared to (a) irradiated tumour tissue post neo-adjuvant radiation within the same group, (b) a CRC patient group (group 2) who had not undergone neo-adjuvant radiotherapy and (c) a non-cancer patient group (group 3). The second aim was to determine if MRE11 expression levels were related to survival or radio-sensitivity post neo-adjuvant radiotherapy.
Results
There was a highly significant increase in MRE 11 expression in group 1 versus groups 2 and 3 (p < 0.001). There was no association between MRE11 levels and survival or radio-sensitivity.
Conclusion
Our findings show radiotherapy causes DSBs at significantly higher levels in normal colonic mucosa of patients post neo-adjuvant treatment which may represent RIBE. If this damage remains unrepaired, increased levels of genomic instability may contribute to the higher occurrence of second cancers in patients treated post neo-adjuvant radiotherapy.


Similar content being viewed by others
References
Nagasawa H, Little JB. Induction of sister chromatid exchanges by extremely low doses of alpha-particles. Cancer Res. 1992;52(22):6394–6.
Han W et al. Nitric oxide mediated DNA double strand breaks induced in proliferating bystander cells after alpha-particle irradiation. Mutat Res. 684(1–2): 81–9.
Yang H, Asaad N, Held KD. Medium-mediated intercellular communication is involved in bystander responses of X-ray-irradiated normal human fibroblasts. Oncogene. 2005;24(12):2096–103.
Ryan R, et al. Pathological response following long-course neoadjuvant chemoradiotherapy for locally advanced rectal cancer. Histopathology. 2005;47(2):141–6.
Cancer survivors: living longer, and now, better. Lancet. 2004; 364(9452): 2153–4.
Gerard A, et al. Preoperative radiotherapy as adjuvant treatment in rectal cancer. Final results of a randomized study of the European Organization for Research and Treatment of Cancer (EORTC). Ann Surg. 1988;208(5):606–14.
Adjuvant radiotherapy for rectal cancer: a systematic overview of 8,507 patients from 22 randomised trials. Lancet. 2001; 358(9290): 1291–304.
Birgisson H, et al. Late adverse effects of radiation therapy for rectal cancer — a systematic overview. Acta Oncol. 2007;46(4):504–16.
Birgisson H, et al. Occurrence of second cancers in patients treated with radiotherapy for rectal cancer. J Clin Oncol. 2005;23(25):6126–31.
Valerie K, Povirk LF. Regulation and mechanisms of mammalian double-strand break repair. Oncogene. 2003;22(37):5792–812.
Lavin MF. ATM and the Mre11 complex combine to recognize and signal DNA double-strand breaks. Oncogene. 2007;26(56):7749–58.
Ebi H, et al. Novel NBS1 heterozygous germ line mutation causing MRE11-binding domain loss predisposes to common types of cancer. Cancer Res. 2007;67(23):11158–65.
Giannini G, et al. Mutations of an intronic repeat induce impaired MRE11 expression in primary human cancer with microsatellite instability. Oncogene. 2004;23(15):2640–7.
Samowitz WS, et al. Microsatellite instability and survival in rectal cancer. Cancer Causes Control. 2009;20(9):1763–8.
Franchitto A, et al. The mammalian mismatch repair protein MSH2 is required for correct MRE11 and RAD51 relocalization and for efficient cell cycle arrest induced by ionizing radiation in G2 phase. Oncogene. 2003;22(14):2110–20.
Barwell J, et al. Biallelic mutation of MSH2 in primary human cells is associated with sensitivity to irradiation and altered RAD51 foci kinetics. J Med Genet. 2007;44(8):516–20.
Tokairin Y, et al. Accelerated growth of intestinal tumours after radiation exposure in Mlh1-knockout mice: evaluation of the late effect of radiation on a mouse model of HNPCC. Int J Exp Pathol. 2006;87(2):89–99.
Choudhury A et al. MRE11 expression is predictive of cause-specific survival following radical radiotherapy for muscle-invasive bladder cancer. Cancer Res. 70(18): 7017–26.
Soderlund K, et al. Intact Mre11/Rad50/Nbs1 complex predicts good response to radiotherapy in early breast cancer. Int J Radiat Oncol Biol Phys. 2007;68(1):50–8.
Agarwal S, Tafel AA, Kanaar R. DNA double-strand break repair and chromosome translocations. DNA Repair (Amst). 2006;5(9–10):1075–81.
van Gent DC, Hoeijmakers JH, Kanaar R. Chromosomal stability and the DNA double-stranded break connection. Nat Rev Genet. 2001;2(3):196–206.
Hoeijmakers JH. Genome maintenance mechanisms for preventing cancer. Nature. 2001;411(6835):366–74.
Conflict of interest
The authors declare they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sheridan, J., Tosetto, M., Gorman, J. et al. Effects of Radiation on Levels of DNA Damage in Normal Non-adjacent Mucosa from Colorectal Cancer Cases. J Gastrointest Canc 44, 41–45 (2013). https://doi.org/10.1007/s12029-012-9442-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12029-012-9442-x