Abstract
Background
Optimal pharmacologic thromboprophylaxis dosing is not well described in patients with subarachnoid hemorrhage (SAH) with an external ventricular drain (EVD). Our patients with SAH with an EVD who receive prophylactic enoxaparin are routinely monitored using timed anti-Xa levels. Our primary study goal was to determine the frequency of venous thromboembolism (VTE) and secondary intracranial hemorrhage (ICH) for this population of patients who received pharmacologic prophylaxis with enoxaparin or unfractionated heparin (UFH).
Methods
A retrospective chart review was performed for all patients with SAH admitted to the neurocritical care unit at Emory University Hospital between 2012 and 2017. All patients with SAH who required an EVD were included.
Results
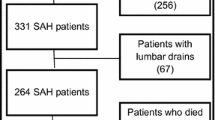
Of 1,351 patients screened, 868 required an EVD. Of these 868 patients, 627 received enoxaparin, 114 received UFH, and 127 did not receive pharmacologic prophylaxis. VTE occurred in 7.5% of patients in the enoxaparin group, 4.4% in the UFH group (p = 0.32), and 3.2% in the no VTE prophylaxis group (p = 0.08). Secondary ICH occurred in 3.83% of patients in the enoxaparin group, 3.51% in the UFH group (p = 1), and 3.94% in the no VTE prophylaxis group (p = 0.53). As steady-state anti-Xa levels increased from 0.1 units/mL to > 0.3 units/mL, there was a trend toward a lower incidence of VTE. However, no correlation was noted between rising anti-Xa levels and an increased incidence of secondary ICH. When compared, neither enoxaparin nor UFH use was associated with a significantly reduced incidence of VTE or an increased incidence of ICH.
Conclusions
In this retrospective study of patients with nontraumatic SAH with an EVD who received enoxaparin or UFH VTE prophylaxis or no VTE prophylaxis, there was no statistically significant difference in the incidence of VTE or secondary ICH. For patients receiving prophylactic enoxaparin, achieving higher steady-state target anti-Xa levels may be associated with a lower incidence of VTE without increasing the risk of secondary ICH.


Similar content being viewed by others
References
Hoh BL, Ko NU, Amin-Hanjani S, Chou SH, Cruz-Flores S, Dangayach NS, et al. 2023 guideline for the management of patients with aneurysmal subarachnoid hemorrhage: a guideline from the American Heart Association/American Stroke Association. Stroke. 2023;54(7):e314–70. Erratum in: Stroke. 2023;54(12):e516.
Zacharia BE, Hickman ZL, Grobelny BT, DeRosa P, Kotchetkov I, Ducruet AF, et al. Epidemiology of aneurysmal subarachnoid hemorrhage. Neurosurg Clin N Am. 2010;21(2):221–33.
Fried HI, Nathan BR, Rowe AS, Zabramski JM, Andaluz N, Bhimraj A, et al. The insertion and management of external ventricular drains: an evidence-based consensus statement: a statement for healthcare professionals from the Neurocritical Care Society. Neurocrit Care. 2016;24(1):61–81.
Tanweer O, Boah A, Huang PP. Risks for hemorrhagic complications after placement of external ventricular drains with early chemical prophylaxis against venous thromboembolisms. J Neurosurg. 2013;119(5):1309–13.
Kshettry VR, Rosenbaum BP, Seicean A, Kelly ML, Schiltz NK, Weil RJ. Incidence and risk factors associated with in-hospital venous thromboembolism after aneurysmal subarachnoid hemorrhage. J Clin Neurosci. 2014;21(2):282–6.
Danish SF, Burnett MG, Ong JG, Sonnad SS, Maloney-Wilensky E, Stein SC. Prophylaxis for deep venous thrombosis in craniotomy patients: a decision analysis. Neurosurgery. 2005;56(6):1286–92.
Agnelli G, Piovella F, Buoncristiani P, Severi P, Pini M, D’Angelo A, et al. Enoxaparin plus compression stockings compared with compression stockings alone in the prevention of venous thromboembolism after elective neurosurgery. N Engl J Med. 1998;339(2):80–5.
Macdonald RL, Amidei C, Baron J, Weir B, Brown F, Erickson RK, et al. Randomized, pilot study of intermittent pneumatic compression devices plus dalteparin versus intermittent pneumatic compression devices plus heparin for prevention of venous thromboembolism in patients undergoing craniotomy. Surg Neurol. 2003;59(5):363–72.
Zachariah J, Snyder KA, Graffeo CS, Khanal DR, Lanzino G, Wijdicks EF, et al. Risk of ventriculostomy-associated hemorrhage in patients with aneurysmal subarachnoid hemorrhage treated with anticoagulant thromboprophylaxis. Neurocrit Care. 2016;25(2):224–9.
Frim DM, Barker FG 2nd, Poletti CE, Hamilton AJ. Postoperative low-dose heparin decreases thromboembolic complications in neurosurgical patients. Neurosurgery. 1992;30(6):830–2.
Bruder M, Schuss P, Konczalla J, El-Fiki A, Lescher S, Vatter H, et al. Ventriculostomy-related hemorrhage after treatment of acutely ruptured aneurysms: the influence of anticoagulation and antiplatelet treatment. World Neurosurg. 2015;84(6):1653–9.
Collen JF, Jackson JL, Shorr AF, Moores LK. Prevention of venous thromboembolism in neurosurgery: a metaanalysis. Chest. 2008;134(2):237–49.
Nyquist P, Jichici D, Bautista C, Burns J, Chhangani S, DeFilippis M, et al. Prophylaxis of venous thrombosis in neurocritical care patients: an executive summary of evidence-based guidelines: a statement for healthcare professionals from the Neurocritical Care Society and Society of Critical Care Medicine. Crit Care Med. 2017;45(3):476–9.
Samuels OB, Sadan O, Feng C, Martin K, Medani K, Mei Y, et al. Aneurysmal subarachnoid hemorrhage: trends, outcomes, and predictions from a 15-year perspective of a single neurocritical care unit. Neurosurgery. 2021;88(3):574–83.
Kothari RU, Brott T, Broderick JP, Barsan WG, Sauerbeck LR, Zuccarello M, et al. The ABCs of measuring intracerebral hemorrhage volumes. Stroke. 1996;27(8):1304–5.
Gard AP, Sayles BD, Robbins JW, Thorell WE, Surdell DL. Hemorrhage rate after external ventricular drain placement in subarachnoid hemorrhage: time to heparin administration. Neurocrit Care. 2017;27(3):350–5.
Wasay M, Khan S, Zaki KS, Khealani BA, Kamal A, Azam I, et al. A non-randomized study of safety and efficacy of heparin for DVT prophylaxis in intracerebral haemorrhage. J Pak Med Assoc. 2008;58(7):362–4.
Orken DN, Kenangil G, Ozkurt H, Guner C, Gundogdu L, Basak M, et al. Prevention of deep venous thrombosis and pulmonary embolism in patients with acute intracerebral hemorrhage. Neurologist. 2009;15(6):329–31.
Dickinson LD, Miller LD, Patel CP, Gupta SK. Enoxaparin increases the incidence of postoperative intracranial hemorrhage when initiated preoperatively for deep venous thrombosis prophylaxis in patients with brain tumors. Neurosurgery. 1998;43(5):1074–81.
Khaldi A, Helo N, Schneck MJ, Origitano TC. Venous thromboembolism: deep venous thrombosis and pulmonary embolism in a neurosurgical population. J Neurosurg. 2011;114(1):40–6.
O’Carroll CB, Capampangan DJ, Aguilar MI, Bravo TP, Hoffman-Snyder CR, Wingerchuk DM, et al. What is the effect of low-molecular weight heparin for venous thromboembolism prophylaxis compared with mechanical methods, on the occurrence of hemorrhagic and venous thromboembolic complications in patients with intracerebral hemorrhage? A critically appraised topic. Neurologist. 2011;17(4):232–5.
Lovenox (enoxaparin) [prescribing information]. Bridgewater, NJ: Sanofi-Aventis; 2017.
Lovenox (enoxaparin) [product monograph]. Laval, Quebec, Canada: Sanofi-Aventis Canada Inc; 2014.
O’Toole RV, Stein DM, O’Hara NN, Frey KP, Taylor TJ, Scharfstein DO, et al. Major Extremity Trauma Research Consortium (METRC). Aspirin or low-molecular-weight heparin for thromboprophylaxis after a fracture. N Engl J Med. 2023;388(3):203–13.
Catapano JS, Koester SW, Parikh PP, Rumalla K, Stonnington HO, Singh R, et al. Association between external ventricular drain removal or replacement and prophylactic anticoagulation in patients with aneurysmal subarachnoid hemorrhage: a propensity-adjusted analysis. Acta Neurochir (Wien). 2023;165(7):1841–6.
Funding
There were no funding sources provided for this study.
Author information
Authors and Affiliations
Contributions
CU: study design, data collection, data analysis, manuscript writing; OS: study design, data analysis, manuscript writing; YS: data analysis, manuscript writing; KNG: study design, manuscript final review; O. Samuels: study design, manuscript final review; SM: data collection, data analysis, manuscript editing; JJ: data collection, manuscript review; YM: study design, study analysis, final manuscript review; WA: study design, data analysis, manuscript writing and final manuscript editing.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflicts of interest.
Ethical Approval/Informed Consent
This article adheres to ethical guidelines and is institutional review board approved. Informed consent was waived.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ukpabi, C., Sadan, O., Shi, Y. et al. Pharmacologic Venous Thromboembolism Prophylaxis in Patients with Nontraumatic Subarachnoid Hemorrhage Requiring an External Ventricular Drain. Neurocrit Care (2024). https://doi.org/10.1007/s12028-024-01993-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12028-024-01993-5