Abstract
Background
Lateral periodic discharges (LPDs) have been recognized as a common electroencephalographic (EEG) pattern in critically ill patients. However, management decisions in these patients are still a challenge for clinicians. This study investigates hemodynamic changes associated with LPDs and evaluates if this pattern is likely to represent an ictal, interictal, or ictal–interictal continuum phenomenon via non-invasive near infra-red spectroscopy (NIRS) with concurrent with continuous EEG.
Methods
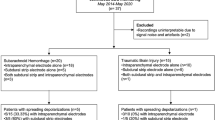
Seventeen patients admitted to the intensive care unit with LPDs on continuous electroencephalogram (EEG) were included. Participants engaged in NIRS recording—with scalp probes on right and left frontal regions simultaneously. Associations between LPDs laterality, primary frequency, NIRS a of cerebral oxygen saturation (SO2), total hemoglobin concentration (tHb), oxygenated hemoglobin concentration (O2Hb), de-oxygenated hemoglobin concentration (HHb), and variables in participant medical history were studied.
Results
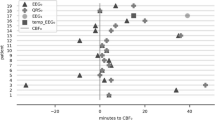
Hemispheres with LPDs showed higher overall SO2 when compared to non-LPDs hemispheres (57% vs 52%, p = 0.03). Additionally, mildly increased tHb, O2Hb, and mildly decreased HHb concentrations were detected in the hemisphere showing LPDs, but changes were not statistically significant. A higher primary frequency of LPDs was associated with lower cerebral SO2 (Pearson correlation r = − 0.55, p = 0.022) and O2Hb (Pearson correlation r = − 0.52, p = 0.033). In patients with seizure during their EEG recording (64.7%), lower tHb (28.2 μmol/L vs 37.8 μmol/L, p = 0.049) and O2Hb (15.5 μmol/L vs 24.2 μmol/L, p = 0.033) were recorded in the LPDs hemisphere.
Conclusions
This study demonstrates an increased cerebral SO2 in the hemisphere with LPDs, and decreased SO2 and O2Hb when the frequency of LPDs increases. The findings indicate that LPDs increase oxygen demand on the ipsilateral hemisphere. We infer that a threshold of LPDs frequency might exit, when the cerebral oxygen demand begins to supersede the ability of delivery, and saturation decreases.



Similar content being viewed by others
References
Hirsch LJ, LaRoche SM, Gaspard N, et al. American Clinical Neurophysiology Society’s standardized critical care EEG terminology: 2012 version. J Clin Neurophysiol. 2013;30:1–27.
Lin L, Drislane FW. Lateralized periodic discharges: a literature review. J Clin Neurophysiol. 2018;35:189–97.
Snodgrass SM, Tsuburaya K, Ajmone-Marsan C. Clinical significance of periodic lateralized epileptiform discharges: relationship with status epilepticus. J Clin Neurophysiol. 1989;6:159–72.
Herlopian A, Struck AF, Rosenthal E, et al. Neuroimaging correlates of periodic discharges. J Clin Neurophysiol. 2018;35:279–94.
Ruiz AR, Vlachy J, Lee JW, et al. Association of periodic and rhythmic electroencephalographic patterns with seizures in critically ill patients. JAMA Neurol. 2017;74:181–8.
Kalamangalam GP, Pohlmann-Eden B. Ictal-interictal continumm. J Clin Neurophysiol. 2018;35:274–8.
Kalamangalam GP, Slater JD. Periodic lateralized epileptiform discharges and afterdischarges: common dynamic mechanisms. J Clin Neurophysiol. 2015;32:331–40.
Chong DJ, Hirsch LJ. Which EEG patterns warrant treatment in the critically ill? Reviewing the evidence for treatment of periodic epileptiform discharges and related patterns. J Clin Neurophysiol. 2005;22:79–91.
Koren JP, Herta J, Pirker S, et al. Rhythmic and periodic EEG patterns of ‘ictal–interictal uncertainty’ in critically ill neurological patients. Clin Neurophysiol. 2016;127:1176–81.
Shvarts V, Zoltsay G, Bowyer SM, et al. Periodic discharges: insight from magnetoencephalography. J Clin Neurophysiol. 2017;34:196–206.
Maciel CB, Hirsch LJ. Definition and classification of periodic and rhythmic patterns. J Clin Neurophysiol. 2018;35:179–88.
Folbergrova J, Ingvar M, Siesjo BK. Metabolic changes in cerebral cortex, hippocampus, and cerebellum during sustained bicuculline-induced seizures. J Neurochem. 1981;37:1228–38.
Schwartz TH. Neurovascular coupling and epilepsy: hemodynamic markers for localizing and predicting seizure onset. Epilepsy Curr. 2007;7:91–4.
Tenney JR, Duong TQ, King JA, et al. fMRI of the brain activation in a genetic rate model of absence seizures. Epilepsia. 2004;45:576–82.
Seyal M. Frontal hemodynamic changes precede EEG onset of temporal lobe seizures. Clin Neurophysiol. 2014;125:442–8.
Peng B, Dai J, Li J, et al. Ictal cerebral haemodynamic characteristics during typical absence seizures, compared to focal seizures with brief alteration of awareness. Epileptic Disord. 2019;21:244–51.
Venkatraman A, Khawaja A, Bag A, et al. Perfusion MRI can impact treatment decision in ictal-interictal continuum. J Clin Neurophysiol. 2017;34:e15–8.
Witsch J, Frey H-P, Schmidt JM, et al. Electroencephalographic periodic discharges and frequency-dependent brain tissue hypoxia in acute brain injury. JAMA Neurol. 2017;74:301–9.
Subramaniam T, Jain A, Hall LT, et al. Lateralized period discharges frequency correlates with glucose metabolism. Neurology. 2019;92:e670–4.
Wolf M, von Siebenthal K, Keel M, et al. Tissue oxygen saturation measured by near infrared spectrophotometry correlates with arterial oxygen saturation during induced oxygenation changes in neonates. Physiol Meas. 2000;21:481–91.
Toronov V, Franceschini MA, Filiaci M, et al. Near-infrared study of fluctuations in cerebral hemodynamics during rest and motor stimulation: temporal analysis and spatial mapping. Med Phys. 2000;27:801–15.
Michalos A, Safonova LP, Olopade CS, et al. Near-infrared spectroscopy for the assessment of vascular responsiveness of the brain: a screening method for cerebrovascular morbidity in obstructive sleep apnea. Sleep. 2003;26(suppl):A390-391.
Kim YH, Kin BJ, Bae JS. Application of near-infrared spectroscopy in clinical neurology. Ann Clin Neurophysiol. 2018;20:57–65.
Fantini S, Franceschini MA, Gratton E. Semi-infinite-geometry boundary problem for light migration in highly scattering media: a frequency-domain study in the diffusion approximation. JOSA B. 1994;11:2128–38.
Gratton E, Fantini S, Franceschini MA, et al. Measurements of scattering and absorption changes in muscle and brain. Philos Trans R Soc Lond B Biol Sci. 1997;352:727–35.
Takane Y, Shibata K, Nishimura Y, et al. Crossed cerebellar and contralateral thalamic hyperfusion in epilepsy. Int Med. 2017;56:1121–2.
Ergun EL, Salanci BV, Erbas B, et al. SPECT in periodic lateralized epileptiform discharges (PLEDs): a case report on PLEDs. Ann Nucl Med. 2006;20:227–31.
Ali II, Pirzada NA, Vaughn BV. Periodic lateralized epileptiform discharges after complex partial status epilepticus associated with increased focal blood flow. J Clin Neurophysiol. 2001;18:565–9.
Aye SMM, Lim KS, Ramli NM, et al. Periodic lateralized epileptiform discharges (PLEDs) in cerebral lupus correlated with white-matter lesions in brain MRI and reduced cerebral blood flow in SPECT. Lupus. 2013;22:510–4.
Vespa P, Tubi M, Claassen J, et al. Metabolic crisis occurs with seizures and periodic discharges after brain trauma. Ann Neurol. 2016;79:579–90.
Nguyen DK, Tremblay J, Pouliot P, et al. Non-invasive continuous EEG-fNIRS recording of temporal lobe seizures. Epilepsy Res. 2012;99:112–26.
Acknowledgements
We thank Stanca Iacob MD, Ph. D. for useful suggestions and editing of the manuscript. This research is supported by the OSF Illinois Neurological Institute research fund, Spring 2017.
Funding
This project was completed with funding from the OSF Healthcare Illinois Neurological Institute research fund.
Author information
Authors and Affiliations
Contributions
MX, principal investigator, designed and conceptualized study, analyzed and interpreted EEG and NIRS data, participated in drafting, and edited manuscript. NW, co-investigator, participated in designing and conceptualizing study, collected/analyzed EEG and NIRS data, and drafted manuscript. AM, co-investigator, provided equipment and advice on NIRS data, and edited manuscript. EE, co-investigator, analyzed and interpreted EEG, and edited manuscript. MG, co-investigator, provides technical guidance on NIRS recording. HW, co-investigator, performed statistical analysis.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no financial, consultant, or institutional conflicts of interest related to this study to declare.
Ethical Approval
This study was approved by the University of Illinois College of Medicine at Peoria IRB, Peoria IL. The manuscript complies with all instruction to authors and was approved all authors. It has not been published elsewhere ad is not under consideration by another journal.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Winslow, N., George, M., Michalos, A. et al. Hemodynamic Changes Associated with Lateralized Periodic Discharges: A Near-Infrared Spectroscopy and Continuous EEG Study. Neurocrit Care 35, 153–161 (2021). https://doi.org/10.1007/s12028-020-01154-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-020-01154-4