Abstract
Background
Recurrent spreading depolarizations (SDs) occur in patients after aneurysmal subarachnoid hemorrhage (aSAH), resulting in metabolic stress to brain. These events are closely associated with delayed cerebral ischemia. Preclinical data suggest that the beneficial effect of nimodipine demonstrated in clinical trials may be related to inhibition of SD rather than limitation of large artery vasospasm.
Methods
Subjects enrolled in a phase 3 trial of intraventricularly delivered, sustained-release nimodipine (EG-1962) versus standard of care oral nimodipine (NEWTON 2) who required surgical clipping had subdural strip electrodes implanted for monitoring of SD. SD was then scored blinded to NEWTON 2 allocation.
Results
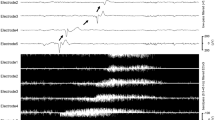
Five subjects underwent electrocorticography monitoring of SD. Three of five patients had SD. There were fewer SDs, a lower rate of SD, and shorter depression durations in subjects treated with EG-1962 compared to standard of care. Outcomes were worse in the standard of care group, though there were baseline imbalances.
Conclusions
These results are consistent with a beneficial effect of locally delivered nimodipine (EG-1962) on SD after aSAH in more severely injured patients who are at risk of delayed cerebral ischemia related to SD. Larger studies are warranted to test this effect.


Similar content being viewed by others
References
Woitzik J, Dreier JP, Hecht N, et al. Delayed cerebral ischemia and spreading depolarization in absence of angiographic vasospasm after subarachnoid hemorrhage. J Cereb Blood Flow. 2012;32:203–12.
Dorhout Mees SM, Rinkel GJ, Feigin VL, et al. Calcium antagonists for aneurysmal subarachnoid haemorrhage. Cochrane Database Syst Rev. 2007. https://doi.org/10.1002/14651858.CD000277.pub3.
Macdonald RL, Higashida RT, Keller E, et al. Randomized trial of clazosentan in patients with aneurysmal subarachnoid hemorrhage undergoing endovascular coiling. Stroke. 2012;43:1463–9.
Dreier JP, Woitzik J, Fabricius M, et al. Delayed ischaemic neurological deficits after subarachnoid haemorrhage are associated with clusters of spreading depolarizations. Brain. 2006;129:3224–37.
Dreier JP, Windmuller O, Petzold G, Lindauer U, Einhaupl KM, Dirnagl U. Ischemia triggered by red blood cell products in the subarachnoid space is inhibited by nimodipine administration or moderate volume expansion/hemodilution in rats. Neurosurgery. 2002;51:1457–65.
Dietz RM, Weiss JH, Shuttleworth CW. Zn2+ influx is critical for some forms of spreading depression in brain slices. J Neurosci. 2008;28:8014–24.
Szabo I, Tóth OM, Torok Z, et al. The impact of dihydropyridine derivatives on the cerebral blood flow response to somatosensory stimulation and spreading depolarization. Br J Pharmacol. 2019;176:1222–34.
Carlson AP, Hanggi D, Macdonald RL, Shuttleworth CW. Nimodipine reappraised: an old drug with a future. Curr Neuropharmacol. 2020;18:65–82.
Macdonald RL, Hanggi D, Strange P, et al. Nimodipine pharmacokinetics after intraventricular injection of sustained-release nimodipine for subarachnoid hemorrhage. J Neurosurg. 2019;1:1–7.
Hanggi D, Etminan N, Aldrich F, et al. Randomized, open-label, phase 1/2a study to determine the maximum tolerated dose of intraventricular sustained release nimodipine for subarachnoid hemorrhage (NEWTON [nimodipine microparticles to enhance recovery while reducing toxicity after subarachnoid hemorrhage]). Stroke. 2017;48:145–51.
Dreier JP, Fabricius M, Ayata C, et al. Recording, analysis, and interpretation of spreading depolarizations in neurointensive care: review and recommendations of the COSBID research group. J Cereb Blood Flow Metab. 2017;37:1595–625.
Hartings JA, Li C, Hinzman JM, et al. Direct current electrocorticography for clinical neuromonitoring of spreading depolarizations. J Cereb Blood Flow Metab. 2017;37:1857–70.
Hanggi D, Etminan N, Mayer SA, et al. Clinical trial protocol: phase 3, multicenter, randomized, double-blind, placebo-controlled, parallel-group, efficacy, and safety study comparing EG-1962 to standard of care oral nimodipine in adults with aneurysmal subarachnoid hemorrhage [NEWTON-2 (nimodipine microparticles to enhance recovery while reducing toxicity after subarachnoid hemorrhage)]. Neurocrit Care. 2019;30:88–97.
Barth M, Capelle H-H, Weidauer S, et al. Effect of nicardipine prolonged-release implants on cerebral vasospasm and clinical outcome after severe aneurysmal subarachnoid hemorrhage. Stroke. 2007;38:330–6.
Lu N, Jackson D, Luke S, Festic E, Hanel RA, Freeman WD. Intraventricular nicardipine for aneurysmal subarachnoid hemorrhage related vasospasm: assessment of 90 days outcome. Neurocrit Care. 2012;16:368–75.
Janovak L, Turcsanyi A, Bozo E, et al. Preparation of novel tissue acidosis-responsive chitosan drug nanoparticles: characterization and in vitro release properties of Ca2+ channel blocker nimodipine drug molecules. Eur J Pharm Sci. 2018;123:79–88.
Acknowledgements
The authors thank the NEWTON 2 steering committee and investigators as well as the COSBID (co-operative studies of brain injury depolarization) consortium for support with the project.
Funding
NEWTON 2 was funded fully by Edge Therapeutics, other funding from the department of neurosurgery at the University of New Mexico and an unrestricted educational Grant from Edge therapeutics.
Author information
Authors and Affiliations
Contributions
Design, manuscript drafting and revisions were performed by APC. Conception of the study was performed by APC and RLM. Data analysis was performed by APC, AA and CWS. Review of the manuscript was performed by AA, DH and CWS. Conception of the parent study was performed by DH and RLM. Critical revisions of the manuscript was performed by RLM and CWS.
Corresponding author
Ethics declarations
Conflict of interest
Carlson—unrestricted educational Grant from Edge Therapeutics. Hänggi—consulting fees for serving on the steering committee for Edge Therapeutics. Macdonald—Grant support from the Brain Aneurysm Foundation and the Canadian Institutes for Health Research; previously Chief Scientific Officer of Edge Therapeutics, Inc., Stock ownership of PDS Biotechnology, Shuttleworth—NINDS.
Informed Consent
All appropriate ethical guidelines were rigorously followed. Informed consent was prospectively used for both studies. Both the monitoring study and the parent RCT were approved by the local IRB (UNM-HRPO-10-159 and UNM-HRPO-16-074).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Carlson, A.P., Alchbli, A., Hänggi, D. et al. Effect of Locally Delivered Nimodipine Microparticles on Spreading Depolarization in Aneurysmal Subarachnoid Hemorrhage. Neurocrit Care 34, 345–349 (2021). https://doi.org/10.1007/s12028-020-00935-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-020-00935-1