Abstract
Purpose
One study found higher leukocytes 8-hydroxy-2′-deoxyguanosine (8-OHdG) levels in patients with spontaneous intracerebral hemorrhage (ICH) than in healthy subjects due to the oxidation of guanosine from deoxyribonucleic acid (DNA). The objective of this study was to determine whether there is an association between oxidative damage of serum DNA and ribonucleic acid (RNA) and mortality in patients with ICH.
Methods
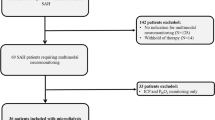
In this observational and prospective study, patients with severe supratentorial ICH (defined as Glasgow Coma Scale < 9) were included from six Intensive Care Units of Spanish hospitals. At the time of severe ICH diagnosis, concentrations in serum of malondialdehyde (as lipid peroxidation biomarker) and of the three oxidized guanine species (OGS) (8-hydroxyguanosine from RNA, 8-hydroxyguanine from DNA or RNA, and 8-OHdG from DNA) were determined. Thirty-day mortality was considered the end-point study.
Results
Serum levels of OGS (p < 0.001) and malondialdehyde (p = 0.002) were higher in non-surviving (n = 46) than in surviving patients (n = 54). There was an association of serum OGS levels with serum malondialdehyde levels (rho = 0.36; p = 0.001) and 30-day mortality (OR = 1.568; 95% CI 1.183–2.078; p = 0.002).
Conclusions
The novel and most important finding of our study was that serum OGS levels in ICH patients are associated with mortality.




Similar content being viewed by others
Abbreviations
- APACHE-II:
-
Acute physiology and chronic health evaluation
- aPTT:
-
Activated partial thromboplastin time
- CT:
-
Computed tomography
- FIO2 :
-
Pressure of arterial oxygen/fraction inspired oxygen
- GCS:
-
Glasgow Coma Scale
- ICH:
-
Spontaneous intracerebral hemorrhage
- INR:
-
International normalized ratio
- OGS:
-
Oxidized guanine species
- PaO2 :
-
Pressure of arterial oxygen
References
Broderick J, Connolly S, Feldmann E, Hanley D, Kase C, Krieger D, Mayberg M, Morgenstern L, Ogilvy CS, Vespa P, Zuccarello, American Heart Association; American Stroke Association Stroke Council; High Blood Pressure Research Council; Quality of Care and Outcomes in Research Interdisciplinary Working Group. Guidelines for the management of spontaneous intracerebral hemorrhage in adults: 2007 update: a guideline from the American Heart Association/American Stroke Association Stroke Council, High Blood Pressure Research Council, and the Quality of Care and Outcomes in Research Interdisciplinary Working Group. Stroke. 2007;38:2001–23.
Morgenstern LB, Hemphill JC 3rd, Anderson C, Becker K, Broderick JP, Connolly ES Jr, Greenberg SM, Huang JN, MacDonald RL, Messé SR, Mitchell PH, Selim M, Tamargo RJ. American Heart Association Stroke Council and Council on Cardiovascular Nursing Guidelines for the management of spontaneous intracerebral hemorrhage: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2010;41:2108–29.
Hemphill JC 3rd, Greenberg SM, Anderson CS, Becker K, Bendok BR, Cushman M, Fung GL, Goldstein JN, Macdonald RL, Mitchell PH, Scott PA, Selim MH, Woo D, American Heart Association Stroke Council; Council on Cardiovascular and Stroke Nursing; Council on Clinical Cardiology. Guidelines for the Management of Spontaneous Intracerebral Hemorrhage: a Guideline for Healthcare Professionals From the American Heart Association/American Stroke Association. Stroke. 2015;46:2032–60.
Aronowski J, Zhao X. Molecular pathophysiology of cerebral hemorrhage: secondary brain injury. Stroke. 2011;42:1781–6.
Aronowski J, Hall CE. New horizons for primary intracerebral hemorrhage treatment: experience from preclinical studies. Neurol Res. 2005;27:268–79.
Katsuki H. Exploring neuroprotective drug therapies for intracerebral hemorrhage. J Pharmacol Sci. 2010;114:366.
Hu X, Tao C, Gan Q, Zheng J, Li H, You C. Oxidative stress in intracerebral hemorrhage: sources, mechanisms, and therapeutic targets. Oxid Med Cell Longev. 2016;2016:3215391.
Li Z, Wu J, Deleo CJ. RNA damage and surveillance under oxidative stress. IUBMB Life. 2006;58:581–8.
Nunomura A, Moreira PI, Takeda A, Smith MA, Perry G. Oxidative RNA damage and neurodegeneration. Curr Med Chem. 2007;14:2968–75.
Ba X, Boldogh I. 8-Oxoguanine DNA glycosylase 1: beyond repair of the oxidatively modified base lesions. Redox Biol. 2018;14:669–78.
Markkanen E. Not breathing is not an option: how to deal with oxidative DNA damage. DNA Repair (Amst). 2017;59:82–105.
Kino K, Hirao-Suzuki M, Morikawa M, Sakaga A, Miyazawa H. Generation, repair and replication of guanine oxidation products. Genes Environ. 2017;39:21.
AbdulSalam SF, Thowfeik FS, Merino EJ. Excessive reactive oxygen species and exotic DNA lesions as an exploitable liability. Biochemistry. 2016;55:5341–52.
Che Y, Wang JF, Shao L, Young T. Oxidative damage to RNA but not DNA in the hippocampus of patients with major mental illness. J Psychiatry Neurosci. 2010;35:296–302.
Kumagai S, Jikimoto T, Saegusa J. Pathological roles of oxidative stress in autoimmune diseases. Rinsho Byori. 2003;51:126–32.
Kikuchi A, Takeda A, Onodera H, Kimpara T, Hisanaga K, Sato N, Nunomura A, Castellani RJ, Perry G, Smith MA, Itoyama Y. Systemic increase of oxidative nucleic acid damage in Parkinson’s disease and multiple system atrophy. Neurobiol Dis. 2002;9:244–8.
Aschbacher K, O’Donovan A, Wolkowitz OM, Dhabhar FS, Su Y, Epel E. Good stress, bad stress and oxidative stress: insights from anticipatory cortisol reactivity. Psychoneuroendocrinology. 2013;38:1698–708.
Malins DC, Haimanot R. Major alterations in the nucleotide structure of DNA in cancer of the female breast. Cancer Res. 1991;51:5430–2.
Shin CS, Moon BS, Park KS, Kim SY, Park SJ, Chung MH, Lee HK. Serum 8-hydroxy-guanine levels are increased in diabetic patients. Diabetes Care. 2001;24:733–7.
Di Minno A, Turnu L, Porro B, Squellerio I, Cavalca V, Tremoli E, Di Minno MN. 8-Hydroxy-2-deoxyguanosine levels and cardiovascular disease: a systematic review and meta-analysis of the literature. Antioxid Redox Signal. 2016;24:548–55.
Di Minno A, Turnu L, Porro B, Squellerio I, Cavalca V, Tremoli E, Di Minno MN. 8-Hydroxy-2-deoxyguanosine levels and heart failure: a systematic review and meta-analysis of the literature. Nutr Metab Cardiovasc Dis. 2017;27:201–8.
Paredes-Sánchez E, Montiel-Company JM, Iranzo-Cortés JE, Almerich-Torres T, Bellot-Arcís C, Almerich-Silla JM. Meta-analysis of the use of 8-OHdG in Saliva as a marker of periodontal disease. Dis Markers. 2018;2018:7916578.
Chen YC, Chen CM, Liu JL, Chen ST, Cheng ML, Chiu DT. Oxidative markers in spontaneous intracerebral hemorrhage: leukocyte 8-hydroxy-2′-deoxyguanosine as an independent predictor of the 30-day outcome. J Neurosurg. 2011;115:1184–90.
Teasdale G, Jennett B. Assessment of coma and impaired consciousness. A practical scale. Lancet. 1974;2:81–4.
Lorente L, Martín MM, Abreu-González P, Sabatel R, Ramos L, Argueso M, Solé-Violán J, Riaño-Ruiz M, Jiménez A, García-Marín V. Serum Malondialdehyde Levels and Mortality in Patients with Spontaneous Intracerebral Hemorrhage. World Neurosurg. 2018;113:e542–7.
Knaus WA, Draper EA, Wagner DP, Zimmerman JE. APACHE II: a severity of disease classification system. Crit Care Med. 1985;13:818–29.
Hemphill JC 3rd, Bonovich DC, Besmertis L, Manley GT, Johnston SC. The ICH score: a simple, reliable grading scale for intracerebral hemorrhage. Stroke. 2001;32:891–7.
Kothari RU, Brott T, Broderick JP, Barsan WG, Sauerbeck LR, Zuccarello M, Khoury J. The ABCs of measuring intracerebral hemorrhage volumes. Stroke. 1996;27:1304–5.
Draper HH, Hadley M. Malondialdehyde determination as index of lipid peroxidation. Methods Enzymol. 1990;186:421–31.
Dalle-Donne I, Rossi R, Colombo R, Giustarini D, Milzani A. Biomarkers of oxidative damage in human disease. Clin Chem. 2006;52:601–23.
Kikugawa K, Kojima T, Yamaki S, Kosugi H. Interpretation of the thiobarbituric acid reactivity of rat liver and brain homogenates in the presence of ferric ion and ethylediaminotetraacetic acid. Anal Biochem. 1992;202:249–55.
Nakamura T, Keep RF, Hua Y, Schallert T, Hoff JT, Xi G. Deferoxamine-induced attenuation of brain edema and neurological deficits in a rat model of intracerebral hemorrhage. J Neurosurg. 2004;100:672–8.
Nakamura T, Kuroda Y, Hosomi N, Okabe N, Kawai N, Tamiya T, Xi G, Keep RF, Itano T. Serine protease inhibitor attenuates intracerebral hemorrhage-induced brain injury and edema formation in rat. Acta Neurochir Suppl. 2010;106:307–10.
Lu F, Nakamura T, Okabe N, Himi N, Nakamura-Maruyama E, Shiromoto T, Narita K, Tsukamoto I, Xi G, Keep RF, Miyamoto O. COA-Cl, a novel synthesized nucleoside analog, exerts neuroprotective effects in the acute phase of intracerebral hemorrhage. J Stroke Cerebrovasc Dis. 2016;25:2637–43.
Acknowledgements
This study was supported by a Grant (OA18/011) from Fundación DISA a la Investigación Médica 2017 (Santa Cruz de Tenerife. Spain). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
LLo conceived, designed and coordinated the study, participated in acquisition and interpretation of data, and drafted the manuscript. MMM, RS, LR, MA, JJC, JSV, and VGM participated in acquisition of data. AGC and APC carried out the analyses of DNA and RNA oxidative damage. PAG carried out the analyses of malondialdehyde concentrations. AJ participated in the interpretation of data. All authors revised the manuscript critically for important intellectual content and made the final approval of the version to be published.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lorente, L., Martín, M.M., González-Rivero, A.F. et al. High Serum DNA and RNA Oxidative Damage in Non-surviving Patients with Spontaneous Intracerebral Hemorrhage. Neurocrit Care 33, 90–96 (2020). https://doi.org/10.1007/s12028-019-00864-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-019-00864-8