Abstract
Background
The Full Outline of UnResponsivness (FOUR) score is a neurological assessment score. Its theoretical benefit over preexisting scores is its evaluation of brainstem reflexes and respiratory pattern which may allow better assessment of patients with severe neurologic impairment.
Objective
Our goal was to perform a scoping systematic review on the available literature for FOUR score and outcome prediction in critically ill patients. The primary outcome of interest was patient global outcome, as assessed by any of: mortality, modified Rankin Score, Glasgow Outcome Score, or any other functional or neuropsychiatric outcome. Information on interobserver reliability was also extracted.
Methods
MEDLINE and five other databases were searched. Inclusion criteria were: humans, adults, and children; prospective randomized controlled trial; prospective cohort, cohort/control, case series, prospective, and retrospective studies. Two reviewers independently screened the results. Full texts for citations passing this initial screen were obtained. Inclusion and exclusion criteria were applied to each article to obtain final articles for review. Results on adult populations are presented here. Data are reported following the preferred reporting items for systematic reviews and meta-analyses guidelines.
Results
The initial search yielded 1709 citations. Of those used, 49 were based on adult and 6 on pediatric populations. All but 8 retrospective adult studies were performed prospectively. Patient categories included traumatic brain injury, intraventricular hemorrhage, intracerebral hemorrhage, subarachnoid hemorrhage, ischemic stroke, general/combined neurology and neurosurgery, post-cardiac arrest, medicine/general critical illness, and patients in the emergency department. A total of 9092 adult patients were studied. Fourteen studies demonstrated good interobserver reliability of the FOUR score. Nine studies demonstrated prognostic value of the FOUR score in predicting mortality and functional outcomes. Thirty-two studies demonstrated equivalency or superiority of the FOUR score compared to Glasgow Coma Score in prediction of mortality and functional outcomes.
Conclusions
The FOUR score has been shown to be a useful outcome predictor in many patients with depressed level of consciousness. It displays good inter-rater reliability among physicians and nurses.
Similar content being viewed by others
Introduction
Clinical assessment of neurological status is a vital element in decision making, outcome prediction, and information sharing among medical professionals. Traditionally, the Glasgow Coma Scale (GCS) has been widely adopted to document and formally assess neurological status. This scale has been praised for its simplicity and ease of use among healthcare workers. However, a shortcoming of the GCS is its inaccuracy in certain patient populations, including those with severe neurological impairment. This population may include intubated patients, which are difficult to assess with the GCS due to their lack of verbal communication. Similarly, alteration of brainstem function and respiratory pattern are important clinical factors reflecting severity of impairment, which the GCS neither addresses nor attempts to quantify.
In 2005, Wijdicks et al. [1] devised a new coma score, the Full Outline of UnResponsiveness (FOUR) score, which addressed the pitfalls of the GCS. The benefit that the FOUR score has over preexisting systems is the inclusion of specific categories for eye movement, motor exam, brainstem reflexes, and respiratory pattern. Thus, the FOUR score provides a structured scoring system for aspects of brainstem function that can be assessed in all patients, including those unable to verbally communicate.
Since its inception, the FOUR score has been studied in a variety of settings and patient populations. Our goal was to perform a scoping systematic review of the existing literature on the application of the FOUR score within critically ill patients and its use in outcome prediction.
Methods
A systematic review using the methodology outlined in the Cochrane Handbook for Systematic Reviewers [2] was conducted. Data are reported following the PRISMA guidelines [3]. The PRISMA checklist is found in the supplementary material as “Appendix A.” The search strategy was decided upon by the primary author (AA) and supervisor (FAZ).
Search Question, Population, Inclusion, and Exclusion Criteria
We aimed to answer the question: What literature is available for FOUR score and outcome prediction in critically ill patients? The primary outcome of interest was patient global outcome, as assessed by any of: mortality, modified Rankin Score (mRS), Glasgow Outcome Score (GOS), or any other functional or neuropsychiatric outcome.
Studies documenting interobserver variability were also included in order to provide context to the reliability of the FOUR score system.
Inclusion criteria were: humans, adults, and children; prospective randomized controlled trial, prospective cohort, cohort/control, case series, prospective and retrospective studies. Non-English studies and those involving animals were excluded. Ultimately, studies on pediatric populations were excluded as these results will be reported in a separate publication. The FOUR score was used as described in the original validation study by Wijdicks et al. [1] (Table 1).
Search Strategy
Six databases were searched from inception to September 2017: MEDLINE, BIOSIS, Scopus, Cochrane Libraries, Globalhealth, and Embase. Published meeting proceedings were included in the search. Following study selection, reference sections of each paper were examined to ensure relevant papers not captured by the initial search were included in the review. “Appendix B” of the supplementary materials highlights the search strategy implemented for each database.
Study Selection
A two-step review was performed. Two reviewers independently screened each resulting title and abstract from the initial search for inclusion. Full texts for citations passing this initial screen were obtained. Inclusion and exclusion criteria were applied to each article to obtain final articles for review. In cases of disagreement between the two reviewers, open discussion was done and a detailed review of the study in question was done to reach a consensus.
Data Collection
Data were extracted from the final list of articles and stored electronically. Data from adult populations were organized into the following categories based on patient pathology and setting by the primary author: patients in the emergency department, medicine and general critical illness patients, traumatic brain injury, intraventricular/intracerebral hemorrhage, subarachnoid hemorrhage, ischemic stroke and general neurology/neurosurgery patients. Data extracted included study country, design, objectives, outcomes, and conclusions made by the study authors. Data on interobserver reliability, if assessed, was also included, as was any information on prognostic ability of the FOUR score.
Quality of Evidence Assessment
Each study was evaluated for quality of evidence using the RTI Item Bank on Risk of Bias and Precision of Observational Studies [4]. This validated item bank is applicable to a variety of observational study designs and evaluates the risk of bias and internal validity of studies using a comprehensive list of itemized questions. “Appendix C” of the supplementary materials provides the tabulated results of the bias assessment for each study included in this scoping review.
Statistical Analysis
A meta-analysis was not performed due to the heterogeneity of data and study design within the studies; thus, a scoping review was performed.
Results
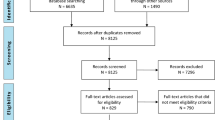
The initial search yielded 1709 citations. Of 55 articles selected for final review, 49 were based on adult populations and will be included in the results of this paper. Sixteen of these articles studied general medical and critical illness populations, 6 articles studied patients in an emergency department setting, 10 articles studied patients with traumatic brain injury, 3 articles studied patients with intracerebral or intraventricular hemorrhage, 1 article studied patients with subarachnoid hemorrhage, 2 articles studied patients with ischemic stroke, and 11 articles studied general neurology and neurosurgery patients. Forty-one of these articles were performed prospectively; the remainder of the articles were performed retrospectively. There were no randomized controlled trials performed in the literature. A total of 9092 adult patients were studied. Figure 1 displays the PRISMA [3] flow diagram of the search results and filtering processes.
Interobserver Reliability
Fourteen studies [1, 5,6,7,8,9,10,11,12,13,14,15,16,17] demonstrated good to excellent interobserver reliability of the FOUR score among raters. In general, a kappa value of 0.4 or less is considered poor, values of 0.4–0.6 are considered fair to moderate, values of 0.6–0.8 are considered good, and values above 0.8 are considered to have excellent inter-rater agreement. The lowest weighted kappa score found in the literature for the FOUR score was 0.68 [10], with the majority being at least 0.80. 3 of the 14 studies were done on patients in the emergency department [5,6,7], 6 on general medical and critical illness patients [8,9,10,11,12,13], and 5 on general neurology and neurosurgical patients [1, 14,15,16,17]. No articles that studied interobserver reliability failed to demonstrate at least good reliability. “Appendix D” of the supplementary materials provides the tabulated results from those studies assessing interobserver reliability.
Prognostic Value When Used Alone
Nine studies [18,19,20,21,22,23,24,25] demonstrated prognostic value of the FOUR score in predicting mortality and functional outcomes. Four were based on general medicine and critical illness populations [18,19,20,21], whereas 5 were based on neurology and neurosurgery populations (including 2 on intracerebral hemorrhage patients [23, 24] and 2 on traumatic brain injury patients [25]).
In a neurological intensive care unit, Akavipat et al. [22] demonstrated the predictive value of the FOUR score in predicting poor outcome at discharge (AUC ROC = 0.88, 95% CI 0.82–0.92) and in-hospital mortality (AUC ROC = 0.92, 95% CI 0.87–0.97). In medicine patients, Rohaut et al. [20] demonstrated the predictive value of the FOUR score in predicting 28-day mortality (c-index of 0.76, 95% CI 0.67–0.84). Other outcomes studied include admission to an intensive care unit [23], overt hepatic encephalopathy [18] and discharge to home or a rehabilitation facility [19]. “Appendix E” of the supplementary materials displays the tabulated results from these studies.
One study [26] examined the use of various weaning parameters (including the FOUR score) in predicting extubation failure in general neurology and neurosurgical patients. The authors found no significant difference in FOUR score between patients who failed extubation and those who were successfully extubated.
Prognostic Value When Compared to the GCS
Thirty-two studies [1, 6, 7, 9, 11, 12, 16, 25, 27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51] demonstrated equivalency or superiority of the FOUR score compared to GCS in the prediction of mortality and functional outcomes. Four of these studied patients in the emergency department [6, 7, 27, 30], 8 studied general medical and critical illness patients [9, 11, 12, 29, 31, 45, 48, 49], 11 studied traumatic brain injury patients [25, 34,35,36, 40,41,42,43,44,45, 47], and 10 studied other neurology/neurosurgery patients (6 studied general neurology and neurosurgical patients [16, 28, 33, 37, 49, 50], 2 studied ischemic stroke patients [38, 39], 1 studied intraventricular hemorrhage patients [32] and 1 studied aneurysmal subarachnoid patients [51]).
Table 2 displays the studies focusing on emergency department populations. Multiple authors demonstrated equal or superior prognostic value of the FOUR score in predicting mortality; for example, Eken et al. [30] showed AUC ROC = 0.788 for FOUR (95% CI 0.722–0.844) and AUC ROC = 0.735 for GCS (95% CI 0.655–0.797) in predicting in-hospital mortality (p = 0.0001). Similarly, Stead et al. [6] demonstrated OR = 0.67 for FOUR (95% CI 0.53–0.84) versus OR = 0.68 for GCS (95% CI 0.56–0.83) in predicting in-hospital mortality (p < 0.001).
Table 3 displays the studies on general medical/critically ill patients. Outcomes studied in this population include those in the intensive care unit (ICU) [49], in-hospital [6, 7, 27, 31] and 28-day mortality [45], successful extubation [45], the ability to become a potential organ donor [29], and other functional outcomes based on the GOS, modified Rankin Scale and Glasgow–Pittsburgh cerebral performance categories [9, 12, 45, 48]. All demonstrated equivalency or superiority of the FOUR score; for example, Wijdicks et al. [49] demonstrated AUC ROC = 0.742 (95% CI 0.694–0.790) for FOUR and AUC ROC = 0.715 for GCS (95% CI 0.663–0.768) in predicting in-ICU mortality (p = 0.001).
Table 4 outlines the studies on traumatic brain injury patients, while Table 5 highlights the other studies on neurology/neurosurgery patient populations. Values for AUC ROC were similar across studies in predicting in-hospital mortality; generally AUC ROC ≥ 0.80 [1, 34, 39, 40, 43, 44, 47]. Mortality was studied at various other time points [35, 46], along with functional outcome based on the GOS, the Karnofsy Performance Score, the Acute Physiology and Chronic Health Evaluation II score and the modified Rankin Scale [34, 36, 40, 41, 43, 44]. Again, all illustrate the equivalent or superior ability of the FOUR score to predict mortality and functional outcomes when compared to GCS.
One study [52] conducted in post-resuscitation encephalopathy patients studied the motor components of both the FOUR score and GCS to predict poor prognosis, and found a lower sensitivity of the FOUR score in outcome prediction (68.7% sensitivity for FOUR, 95% CI 41.4–88.9 vs. 87.5% sensitivity for GCS, 95% CI 61.6–92.6).
Quality of Evidence
Quality of evidence was assessed using the RTI Item Bank on Risk of Bias and Precision of Observational Studies [4]. Based on its itemized list of questions, there was an overall low risk of bias in the studies included in this review.
Discussion
We aimed to perform a scoping review of the FOUR score and its use in outcome prediction. The existing literature around the FOUR score generally demonstrates that it possesses prognostic value alone and in comparison with the GCS, as exemplified through 9 and 32 mainly prospective studies, respectively.
In predicting extubation failure, however, Ko et al. [26] failed to show predictive value for the FOUR score as well as all other weaning parameters they chose to study, including rapid shallow breathing index and spontaneous breathing trial. In neurology and neurosurgical patients, the ability to forcefully cough and actively clear secretions is of importance in successful extubation, and perhaps not specifically assessed by the FOUR score. However, the authors also had missing data regarding etiology of respiratory failure and inaccurate fluid balance, which may have contributed to their negative results. In contrast, Said et al. [45] published a pilot study among a general ICU population, and did show superiority of the FOUR score compared to GCS in predicting successful extubation at 14 days post-intubation.
In comatose patients post cardiopulmonary arrest, Topcuoglu et al. [52] examined the motor parts of the GCS and FOUR score in outcome prediction and showed a lower sensitivity of the FOUR motor component compared to the GCS motor component. When either scores were combined with specific magnetic resonance imaging (MRI) findings, sensitivity improved to 100%. It is important to note that in this study, the authors focus primarily on MRI findings, and specific details regarding how and when the FOUR score is measured, as well as the presence of potentially confounding factors (for example, sedating or paralyzing medications) is unclear.
Taking into consideration the shortcomings of these two studies, overall, the remainder of the present literature around the FOUR score displays its usefulness as a neurological assessment tool. While its accuracy in predicting extubation failure is not clearly established—again, perhaps as a result of the limitations described previously—multiple studies have shown it to hold prognostic significance in predicting mortality and functional outcomes in diverse patient populations, from general medicine patients to those with neurosurgical pathology.
Accurate neurological assessment is made imperfect by the presence of mental alteration caused by sedating medications, endotracheal intubation, and language barriers including patient dysphasia or dysarthria. These conditions make application of the GCS verbal score especially difficult, as it relies heavily on accurate comprehension, orientation and the ability of the patient to verbally respond to the rater. Raters will make adjustments to the GCS to account for the presence of such clouding factors, especially in the presence of endotracheal intubation, but these adjustments are non-standardized across institutions. The FOUR score bypasses this by not including a verbal score based on orientation or the ability of a patient to respond; a degree of comprehension is required to obey simple motor commands only. In patients with receptive aphasia, application of both the FOUR score and GCS may be difficult secondary to inability of the patient to process motor commands; however, the FOUR score subcategories of eye, motor, brainstem and respiratory function can be quantified in a greater number of patients than GCS including those that are intubated or with expressive language impairment only.
In comparison to the GCS, the FOUR score may also be helpful in further subcategorizing patients with severe neurological impairment based on their brainstem function and respiratory pattern, which the GCS is unable to do. This has the potential to better stratify patients with severe neurological injury and provide clinicians with further information regarding overall prognosis. These advantages, combined with its good inter-rater reliability, give the FOUR score the potential to replace conventional scoring systems and allow for precise and consistent neurological assessments among health care providers.
Limitations
Despite the promising results surrounding the application of the FOUR score across these varied patient populations, there are some limitations which deserve highlighting.
First, a main limitation of this review is the heterogeneity of the studies included. Given the assortment of study designs, objectives and patient populations, a meaningful meta-analysis of results was impossible to conduct. Thus, we are left with a purely descriptive analysis of the available literature. With that said, the body of literature which includes almost 10,000 patients provides evidence in support of its use for clinical grading in a variety of situations.
Second, the FOUR score itself is limited by the fact that it requires a more detailed neurological examination and the experience to confidently conduct such an examination. The saving grace of more simplistic systems, such as GCS, is they can be readily employed by various medical and paramedical professionals with fairly consistent reliability. This flexibility of GCS in the face of varied training and backgrounds of the assessor is a major benefit of the system. The FOUR score requires a slightly higher background knowledge of the nervous system, which may limit its application in other settings, such as the pre-hospital environment.
Third, the majority of the literature identified within this review focuses on general medical/ICU or traumatic brain injury patients. Thus, the conclusion regarding the association of the FOUR score with outcome or its performance compared to GCS in other populations, such as subarachnoid hemorrhage or intracerebral hemorrhage, is quite limited at this time. Further work is required in these sub-populations of patients to determine the role of the FOUR score. Studies directly comparing the utility of the FOUR score versus GCS in intubated versus non-intubated patients, and those with brain stem lesions versus those without, are also lacking within these populations. While the advantages of using the FOUR score over GCS in intubated patients is logical, and as previously described relates primarily to its ability to bypass a verbal assessment, this has not been specifically demonstrated in the literature and deserves focused attention in future studies.
Lastly, as with many outcome prediction studies, the presence of observer bias is possible. Only 12 of the 49 studies were transparent about the presence of blinding in their study protocol. Many authors failed to mention whether they blinded their raters and outcome observers, making the presence of observer bias unclear. The numbers of patients in whom life-sustaining therapies were withdrawn in their clinical course is also poorly described in the literature, potentially introducing an element of selection bias as well.
Conclusions
The existing literature favors the FOUR score as a useful outcome predictor in many patients with depressed level of consciousness. It has been studied in a wide variety of critically ill patients, both with and without neurologic pathology in predicting mortality and functional outcomes. It displays good inter-rater reliability among physicians and nurses.
References
Wijdicks EF, Bamlet WR, Maramattom BV, Manno EM, McClelland RL. Validation of a new coma scale: the FOUR score. Ann Neurol. 2005;58(4):585–93.
Higgins J, Green S. Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0. 2011.
Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gotzsche PC, Ioannidis JP, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. PLoS Med. 2009;6(7):e1000100.
Viswanathan M, Berkman N. Development of the RTI item bank on risk of bias and precision of observational studies. In: Rockville, MD, editor. Center RI-UoNCE-bP. Rockville: Agency for Healthcare Research and Quality; 2011.
Kevric J, Jelinek GA, Knott J, Weiland TJ. Validation of the full outline of unresponsiveness (FOUR) scale for conscious state in the emergency department: comparison against the Glasgow coma scale. Emer Med J. 2011;28(6):486–90.
Stead LG, Wijdicks EF, Bhagra A, Kashyap R, Bellolio MF, Nash DL, et al. Validation of a new coma scale, the FOUR score, in the emergency department. Neurocrit Care. 2009;10(1):50–4.
Matheesiriwat N, Kuptniratsaikul S. The FOUR Score and Glasgow Coma Scale to Evaluate the Patients with Intubation at Emergency Room. R Thai Army Med J. 2012;65(3):145–52.
Adcock AK, Kosiorek H, Parich P, Chauncey A, Wu Q, Demaerschalk BM. Reliability of robotic telemedicine for assessing critically ill patients with the Full Outline of Unresponsiveness score and Glasgow Coma Scale. Telemed J E-Health. 2017;23(7):555–60.
Bruno MA, Ledoux D, Lambermont B, Damas F, Schnakers C, Vanhaudenhuyse A, et al. Comparison of the full outline of unresponsiveness and Glasgow Liege Scale/Glasgow Coma Scale in an intensive care unit population. Neurocrit Care. 2011;15:1–7 (in press).
Fischer M, Ruegg S, Czaplinski A, Strohmeier M, Lehmann A, Tschan F, et al. Inter-rater reliability of the Full Outline of UnResponsiveness score and the Glasgow Coma Scale in critically ill patients: a prospective observational study. Crit Care. 2010;14:R64.
Gujjar AR, Jacob PC, Nandhagopal R, Ganguly SS, Obaidy A, Al-Asmi AR. Full Outline of UnResponsiveness score and Glasgow Coma Scale in medical patients with altered sensorium: Interrater reliability and relation to outcome. J Crit Care. 2013;28(3):316.e1–8.
Iyer VN, Mandrekar JN, Danielson RD, Zubkov AY, Elmer JL, Wijdicks EF. Validity of the FOUR score coma scale in the medical intensive care unit. Mayo Clin Proc. 2009;84(8):694–701.
Kramer AA, Wijdicks EF, Snavely VL, Dunivan JR, Naranjo LL, Bible S, et al. A multicenter prospective study of interobserver agreement using the Full Outline of Unresponsiveness score coma scale in the intensive care unit. Crit Care Med. 2012;40(9):2671–6.
Akavipat P. Endorsement of the FOUR score for consciousness assessment in neurosurgical patients. Neurol Med Chir. 2009;49(12):565–71.
Johnson VD, Whitcomb J. Neuro/Trauma intensive care unit nurses’ perception of the use of the full outline of unresponsiveness score versus the Glasgow Coma Scale when assessing the neurological status of intensive care unit patients. Dimens Crit Care Nurs. 2013;32(4):180–3.
Temiz NC, Kose G, Tehli O, Acikel C, Hatipoglu S. A comparison between the effectiveness of Full Outline of Unresponsiveness and Glasgow Coma Score at neurosurgical intensive care unit patients. Turk Neurosurg. 2016;21:21.
Wolf CA, Wijdicks EF, Bamlet WR, McClelland RL. Further validation of the FOUR score coma scale by intensive care nurses. Mayo Clin Proc. 2007;82(4):435–8.
Mouri S, Tripon S, Rudler M, Mallet M, Mayaux J, Thabut D, et al. FOUR score, a reliable score for assessing overt hepatic encephalopathy in cirrhotic patients. Neurocrit Care. 2015;22(2):251–7.
Rittenberger JC, Tisherman SA, Holm MB, Guyette FX, Callaway CW. An early, novel illness severity score to predict outcome after cardiac arrest. Resuscitation. 2011;82(11):1399–404.
Rohaut B, Porcher R, Hissem T, Heming N, Chillet P, Djedaini K, et al. Brainstem response patterns in deeplysedated critically-ill patients predict 28-day mortality. PLoS ONE. 2017;12(4):e0176012.
Young CS, Callaway CW, Rittenberger JC, Post Cardiac Arrest S. Combination of initial neurologic examination and continuous EEG to predict survival after cardiac arrest. Resuscitation. 2015;94:73–9.
Akavipat P, Sookplung P, Kaewsingha P, Maunsaiyat P. Prediction of discharge outcome with the full outline of unresponsiveness (FOUR) score in neurosurgical patients. Acta Med Okayama. 2011;65(3):205–10.
Klaas JP, Braksick S, Mandrekar J, Sedova P, Bellolio MF, Rabinstein AA, et al. Factors associated with the need for intensive care unit admission following supratentorial intracerebral hemorrhage: the triage ICH model. Neurocrit Care. 2017;27(1):75–81.
Mittal MK, Lele A. Predictors of poor outcome at hospital discharge following a spontaneous intracerebral hemorrhage. Int J Neurosci. 2011;121(5):267–70.
Senapathi TGA, Wiryana M, Sinardja K, Nada KW, Sutawan IBKJ, Ryalino C, et al. Jugular bulb oxygen saturation correlates with full outline of responsiveness score in severe traumatic brain injury patients. Open Access Emerg Med. 2017;9:69–72.
Ko R, Ramos L, Chalela JA. Conventional weaning parameters do not predict extubation failure in neurocritical care patients. Neurocrit Care. 2009;10(3):269–73.
Baratloo A, Shokravi M, Safari S, Aziz AK. Predictive value of Glasgow Coma Score and Full Outline of Unresponsiveness score on the outcome of multiple trauma patients. Arch Iran Med. 2016;19(3):215–20.
Chen B, Grothe C, Schaller K. Validation of a new neurological score (FOUR Score) in the assessment of neurosurgical patients with severely impaired consciousness. Acta Neurochir. 2013;155(11):2133–9.
de Groot YJ, Wijdicks EF, van der Jagt M, Bakker J, Lingsma HF, Ijzermans JN, et al. Donor conversion rates depend on the assessment tools used in the evaluation of potential organ donors. Intensive Care Med. 2011;37(4):665–70.
Eken C, Kartal M, Bacanli A, Eray O. Comparison of the Full Outline of Unresponsiveness Score Coma Scale and the Glasgow Coma Scale in an emergency setting population. Eur J Emer Med. 2009;16(1):29–36.
Fugate JE, Rabinstein AA, Claassen DO, White RD, Wijdicks EF. The FOUR score predicts outcome in patients after cardiac arrest. Neurocrit Care. 2010;13(2):205–10.
Giray S, Sen O, Sarica FB, Tufan K, Karatas M, Karakurum Goksel B, et al. Spontaneous Primary Intraventricular Hemorrhage in Adults: clinical data etiology and outcome. Turk Neurosurg. 2009;19(4):338–44.
Gorji MA, Gorji AM, Hosseini SH. Which score should be used in intubated patients’ Glasgow coma scale or full outline of unresponsiveness? Int J Appl Basic Med Res. 2015;5(2):92–5.
Gorji MA, Hoseini SH, Gholipur A, Mohammadpur RA. A comparison of the diagnostic power of the Full Outline of Unresponsiveness scale and the Glasgow coma scale in the discharge outcome prediction of patients with traumatic brain injury admitted to the intensive care unit. Saudi J Anaesth. 2014;8(2):193–7.
Jalali R, Rezaei M. A comparison of the glasgow coma scale score with full outline of unresponsiveness scale to predict patients’ traumatic brain injury outcomes in intensive care units. Crit Care Res Prac. 2014;2014:289803.
Kasprowicz M, Burzynska M, Melcer T, Kubler A. A comparison of the Full Outline of UnResponsiveness (FOUR) score and Glasgow Coma Score (GCS) in predictive modelling in traumatic brain injury. Br J Neurosurg. 2016;30(2):211–20.
Khanal K, Bhandari SS, Shrestha N, Acharya SP, Marhatta MN. Comparison of outcome predictions by the Glasgow coma scale and the Full Outline of UnResponsiveness score in the neurological and neurosurgical patients in the Intensive Care Unit. Indian J Crit Care Med. 2016;20(8):473–6.
Kocak Y, Ozturk S, Ege F, Ekmekci AH. A useful new coma scale in acute stroke patients: Four score. Anaesth Intensive Care. 2012;40(1):131–6.
Mansour OY, Megahed MM, Abd Elghany EHS. Acute ischemic stroke prognostication, comparison between Glasgow Coma Score, NIHS Scale and Full Outline of UnResponsiveness Score in intensive care unit. Alex J of Med. 2015;51(3):247–53.
McNett M, Amato S, Gianakis A, Grimm D, Philippbar SA, Belle J, et al. The FOUR score and GCS as predictors of outcome after traumatic brain injury. Neurocrit Care. 2014;21(1):52–7.
McNett MM, Amato S, Philippbar SA. A comparative study of Glasgow Coma Scale and Full Outline of Unresponsiveness scores for predicting long-term outcome after brain injury. J Neurosci Nursing: J Am Assoc Neurosci Nurses. 2016;48(4):207–14.
Nyam TE, Ao KH, Hung SY, Shen ML, Yu TC, Kuo JR. FOUR score predicts early outcome in patients after traumatic brain injury. Neurocrit Care. 2017;26(2):225–31.
Okasha AS, Fayed AM, Saleh AS. The FOUR score predicts mortality, endotracheal intubation and ICU length of stay after traumatic brain injury. Neurocrit Care. 2014;21(3):496–504.
Sadaka F, Patel D, Lakshmanan R. The FOUR score predicts outcome in patients after traumatic brain injury. Neurocrit Care. 2012;16(1):95–101.
Said T, Chaari A, Hakim KA, Hamama D, Casey WF. Usefulness of full outline of unresponsiveness score to predict extubation failure in intubated critically-ill patients: a pilot study. Int J Crit Illn Inj Sci. 2016;6(4):172–7.
Saika A, Bansal S, Philip M, Devi BI, Shukla DP. Prognostic value of FOUR and GCS scores in determining mortality in patients with traumatic brain injury. Acta Neurochir. 2015;157(8):1323–8.
Sepahvand E, Jalali R, Mirzaei M, Ebrahimzadeh F, Ahmadi M, Amraii E. Glasgow Coma Scale versus Full Outline of UnResponsiveness scale for prediction of outcomes in patients with traumatic brain injury in the intensive care unit. Turk Neurosurg. 2016;26(5):720–4.
Weiss N, Venot M, Verdonk F, Chardon A, Le Guennec L, Llerena MC, et al. Daily FOUR score assessment provides accurate prognosis of long-term outcome in out-of-hospital cardiac arrest. Rev Neurol. 2015;171(5):437–44.
Wijdicks EF, Kramer AA, Rohs T Jr, Hanna S, Sadaka F, O’Brien J, et al. Comparison of the Full Outline of UnResponsiveness score and the Glasgow Coma Scale in predicting mortality in critically ill patients. Crit Care Med. 2015;43(2):439–44.
Wijdicks EF, Rabinstein AA, Bamlet WR, Mandrekar JN. FOUR score and Glasgow Coma Scale in predicting outcome of comatose patients: a pooled analysis. Neurology. 2011;77(1):84–5.
Zeiler FA, Lo BW, Akoth E, Silvaggio J, Kaufmann AM, Teitelbaum J, et al. Predicting outcome in subarachnoid hemorrhage (SAH) utilizing the Full Outline of UnResponsiveness (FOUR) score. Neurocrit Care. 2017;05:05.
Topcuoglu MA, Oguz KK, Buyukserbetci G, Bulut E. Prognostic value of magnetic resonance imaging in post-resuscitation encephalopathy. Intern Med. 2009;48(18):1635–45.
Funding
FAZ has received salary support for dedicated research time, during which this manuscript was completed. Such salary support came from: the Cambridge Commonwealth Trust Scholarship, the Royal College of Surgeons of Canada—Harry S. Morton Travelling Fellowship in Surgery and the University of Manitoba Clinician Investigator Program.
Author information
Authors and Affiliations
Contributions
AA contributed to the conception and design of this work, the acquisition and interpretation of data, the initial drafting of the manuscript and the critical revision of intellectual content. MH contributed to the interpretation of data and the critical revision of intellectual content. FAZ contributed to the conception and design of this work, the interpretation of data and the critical revision of intellectual content. All authors were involved in the final approval of the manuscript to be published.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Almojuela, A., Hasen, M. & Zeiler, F.A. The Full Outline of UnResponsiveness (FOUR) Score and Its Use in Outcome Prediction: A Scoping Systematic Review of the Adult Literature. Neurocrit Care 31, 162–175 (2019). https://doi.org/10.1007/s12028-018-0630-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-018-0630-9