Abstract
Introduction
The Clot Lysis: Evaluating Accelerated Resolution of IVH trial examined whether irrigating the ventricular system with alteplase improved functional outcomes in patients with small intracerebral hemorrhage (ICH) and large intraventricular hemorrhage (IVH). Thalamic ICH location was common and was associated with poor outcome. One possible explanation is thalamic ICH-associated mass effect obstructing the third ventricle. We hypothesized that patients with thalamic ICH obstructing the third ventricle would have worse functional outcomes compared to patients without obstructing lesions.
Methods
ICH obstruction of third ventricle was defined as third ventricle compression on 1 or more axial computed tomography slices visually impeding cerebral spinal fluid flow. If the third ventricle was casted with IVH, it was scored as such. Multivariable logistic regression analyses were used to determine whether obstruction of the third ventricle predicts poor functional outcomes defined as modified Rankin score (mRS) 4–6, higher mRS, and mortality at 180 days. Models were adjusted for thalamic ICH location, ICH volume, IVH volume, age, hydrocephalus, baseline Glasgow coma scale, and percentage of low cerebral perfusion pressures during treatment.
Results
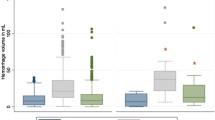
Among saline-treated patients, obstruction of the third ventricle by IVH was a significant predictor of higher mRS at 180 days (OR 1.87, CI 1.01–3.47) and mortality at 180 days (OR 2.73, CI 1.27–5.87) while obstruction by ICH was not. In contrast, among alteplase-treated patients, obstruction by ICH was a significant predictor of mRS 4–6 (OR 3.20, CI 1.30–7.88) and higher mRS at 180 days (OR 2.33, CI 1.24–4.35), while obstruction by IVH was not.
Conclusions
Poor outcomes were associated with mass-related obstruction of the third ventricle from thalamic ICH in alteplase-treated patients and from IVH in saline-treated patients. Once the ventricular system is cleared with alteplase, obstruction of cerebral spinal fluid flow from thalamic ICH might become important in functional recovery.

Similar content being viewed by others
References
Broderick JP, Brott T, Tomsick T, Miller R, Huster G. Intracerebral hemorrhage more than twice as common as subarachnoid hemorrhage. J Neurosurg. 1993;78(2):188–91.
Hallevi H, Albright KC, Aronowski J, Barreto AD, Martin-Schild S, Khaja AM, et al. Intraventricular hemorrhage: anatomic relationships and clinical implications. Neurology. 2008;70(11):848–52.
Moradiya Y, Murthy SB, Newman-Toker DE, Hanley DF, Ziai WC. Intraventricular thrombolysis in intracerebral hemorrhage requiring ventriculostomy: a decade-long real-world experience. Stroke. 2014;45(9):2629–35.
Gaberel T, Magheru C, Parienti JJ, Huttner HB, Vivien D, Emery E. Intraventricular fibrinolysis versus external ventricular drainage alone in intraventricular hemorrhage: a meta-analysis. Stroke. 2011;42(10):2776–81.
Nieuwkamp DJ, de Gans K, Rinkel GJ, Algra A. Treatment and outcome of severe intraventricular extension in patients with subarachnoid or intracerebral hemorrhage: a systematic review of the literature. J Neurol. 2000;247(2):117–21.
Naff N, Williams MA, Keyl PM, Tuhrim S, Bullock MR, Mayer SA, et al. Low-dose recombinant tissue-type plasminogen activator enhances clot resolution in brain hemorrhage: the intraventricular hemorrhage thrombolysis trial. Stroke. 2011;42(11):3009–16.
Hanley DF, Lane K, McBee N, Ziai W, Tuhrim S, Lees KR, et al. Thrombolytic removal of intraventricular haemorrhage in treatment of severe stroke: results of the randomised, multicentre, multiregion, placebo-controlled CLEAR III trial. Lancet. 2017;389(10069):603–11.
Ziai WC, Tuhrim S, Lane K, McBee N, Lees K, Dawson J, et al. A multicenter, randomized, double-blinded, placebo-controlled phase III study of Clot Lysis Evaluation of Accelerated Resolution of Intraventricular Hemorrhage (CLEAR III). Int J Stroke. 2014;9(4):536–42.
Ziai WC, Melnychuk E, Thompson CB, Awad I, Lane K, Hanley DF. Occurrence and impact of intracranial pressure elevation during treatment of severe intraventricular hemorrhage. Crit Care Med. 2012;40(5):1601–8.
Delcourt C, Sato S, Zhang S, Sandset EC, Zheng D, Chen X, et al. Intracerebral hemorrhage location and outcome among INTERACT2 participants. Neurology. 2017;88(15):1408–14.
Eslami V, Nekoobakht-Tak S, Tahsili-Fahadan P, Dlugash R, McBee N, Lane K, et al. Influence of intracerebral hemorrhage location in patients with spontaneous intraventricular hemorrhage results from the clot lysis: evaluating accelerated resolution of intraventricular hemorrhage (CLEAR) III trial. In: Annual MEETING of the Neurocritical Care Society 2016, National Harbor, MD.
Zacharia BE, Vaughan KA, Hickman ZL, Bruce SS, Carpenter AM, Petersen NH, et al. Predictors of long-term shunt-dependent hydrocephalus in patients with intracerebral hemorrhage requiring emergency cerebrospinal fluid diversion. Neurosurg Focus. 2012;32(4):E5.
Miller C, Tsivgoulis G, Nakaji P. Predictors of ventriculoperitoneal shunting after spontaneous intraparenchymal hemorrhage. Neurocrit Care. 2008;8(2):235–40.
Ng LK, Schwarz G, Mishkin MM. Hematoma from arteriovenous malformation producing hydrocephalus and simulating a thalamic tumor. Report of two cases. J Neurosurg. 1971;34(2 Pt 1):229–35.
Tokumitsu N, Sako K, Kunimoto M, Nakai H, Yonemasu Y. Glial cyst in the thalamus with intracystic hemorrhage—case report. Neurol Med Chir (Tokyo). 1997;37(3):284–7.
Mahta A, Katz PM, Kamel H, Azizi SA. Intracerebral hemorrhage with intraventricular extension and no hydrocephalus may not increase mortality or severe disability. J Clin Neurosci. 2016;30:56–9.
Diringer MN, Edwards DF, Zazulia AR. Hydrocephalus: a previously unrecognized predictor of poor outcome from supratentorial intracerebral hemorrhage. Stroke. 1998;29(7):1352–7.
Hansen BM, Ullman N, Norrving B, Hanley DF, Lindgren A. Applicability of clinical trials in an unselected cohort of patients with intracerebral hemorrhage. Stroke. 2016;47(10):2634–7.
Acknowledgments
We wish to thank the CLEAR III patients and their families and the CLEAR III investigators and coordinators who provided and cared for them. The CLEAR III study was funded by the National Institute of Neurological Disorders and Stroke.
Funding
Dr. Daniel F. Hanley was awarded significant research support of Grants R01NS046309, 5U01 NS062851-05, and 5U01 NS080824-02.
Author information
Authors and Affiliations
Contributions
NLU contributed to study conception and design, acquisition of data, analysis and interpretation of data, and drafting of the manuscript. PTF contributed to study conception and design, acquisition of data, and critical revision to the manuscript. CBT contributed to analysis and interpretation of data, and critical revisions to the manuscript. WCZ contributed to acquisition of data, analysis and interpretation of data, and critical revision to the manuscript. DFH contributed to study conception and design and critical revision to the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Ms. Thompson, Dr. Hanley, Dr. Ullman, and Dr. Ziai report grants from the National Institutes of Health (NINDS) during the conduct of the study. Dr. Tahsili-Fahadan has nothing to disclose.
Clinical trial registration
ClinicalTrials.gov NCT00784134.
Rights and permissions
About this article
Cite this article
Ullman, N.L., Tahsili-Fahadan, P., Thompson, C.B. et al. Third Ventricle Obstruction by Thalamic Intracerebral Hemorrhage Predicts Poor Functional Outcome Among Patients Treated with Alteplase in the CLEAR III Trial. Neurocrit Care 30, 380–386 (2019). https://doi.org/10.1007/s12028-018-0610-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-018-0610-0