Abstract
Background
Improved prognostication during the acute phase of intracerebral hemorrhage (ICH) could influence goals of care. We investigated the utility of diffusion tensor imaging (DTI)-derived data obtained during the acute phase of ICH in predicting outcome, compared with the ICH score.
Methods
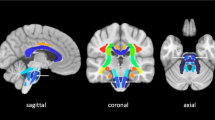
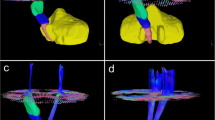
We measured fractional anisotropy (FA) values in 5 slices below the level of the lesion on the affected and unaffected corticospinal tracts (CST) and in the cerebral peduncles (CPs) in 32 patients with supratentorial ICH who had DTI MRI within 4 days after ictus. We calculated the FA ratio (rFA = FAaffected side/FAunaffected side), and examined the value of the ICH score and rFA in predicting functional outcome assessed by modified Ranking Scale (mRS) at follow-up, using ROC analyses.
Results
The rFA values at the CPs level, but not the 5 slices below the lesion, were significantly lower in the group with poor functional outcome (defined as mRS > 2) than in those with good functional outcome (0.96 ± 0.14 vs. 0.99 ± 0.08, p = 0.025). The ICH score had greater areas under ROC curve in predicting functional outcome compared to the mean rFA (AUC 0.74 vs. 0.44; p = 0.01 for mRS > 2; and 0.84 vs. 0.40; p < 0.001 for mRS > 3).
Conclusions
The prognostic value of the ICH score surpassed that of DTI-derived data during the acute phase of ICH in this cohort of patients. Prospective and larger studies are needed to validate our findings and to assess the prognostic role of various DTI-derived measures at different times following ICH.

Similar content being viewed by others
References
Saver JL, Johnston KC, Homer D, et al. Infarct volume as a surrogate or auxiliary outcome measure in ischemic stroke clinical trials. The RANTTAS investigators. Stroke. 1999;30:293–8.
Crafton KR, Mark AN, Cramer SC. Improved understanding of cortical injury by incorporating measures of functional anatomy. Brain. 2003;126:1650–9.
Shelton FN, Reding MJ. Effect of lesion location on upper limb motor recovery after stroke. Stroke. 2001;32:107–12.
Broderick JP, Brott TG, Duldner JE, Tomsick T, Huster G. Volume of intracerebral hemorrhage. A powerful and easy-to-use predictor of 30-day mortality. Stroke. 1993;24:987–93.
Helweg-Larsen S, Sommer W, Strange P, Lester J, Boysen G. Prognosis for patients treated conservatively for spontaneous intracerebral hematomas. Stroke. 1984;15:1045–8.
Luo GG, Ramchurn A, Chen YF, et al. Early predictors of hematoma resorption rate in medically treated patients with spontaneous supratentorial hemorrhage. J Neurol Sci. 2013;327:55–60.
Brouwers HB, Greenberg SM. Hematoma expansion following acute intracerebral hemorrhage. Cerebrovasc Dis. 2013;35:195–201.
Juvela S. Risk factors for impaired outcome after spontaneous intracerebral hemorrhage. Arch Neurol. 1995;52:1193–200.
Hemphill JC III, Bonovich DC, Besmertis L, Manley GT, Johnston SC. The ICH score: a simple, reliable grading scale for intracerebral hemorrhage. Stroke. 2001;32:891–7.
Becker KJ, Baxter AB, Cohen WA, et al. Withdrawal of support in intracerebral hemorrhage may lead to self-fulfilling prophecies. Neurology. 2001;56:766–72.
Mori S, Zhang J. Principles of diffusion tensor imaging and its applications to basic neuroscience research. Neuron. 2006;51:527–39.
Pierpaoli C, Jezzard P, Basser PJ, Barnett A, Di Chiro G. Diffusion tensor mr imaging of the human brain. Radiology. 1996;201:637–48.
Cho SH, Kim SH, Choi BY, et al. Motor outcome according to diffusion tensor tractography findings in the early stage of intracerebral hemorrhage. Neurosci Lett. 2007;421:142–6.
Yu C, Zhu C, Zhang Y, et al. A longitudinal diffusion tensor imaging study on wallerian degeneration of corticospinal tract after motor pathway stroke. Neuroimage. 2009;47:451–8.
Kim DG, Ahn YH, Byun WM, et al. Degeneration speed of corticospinal tract in patients with cerebral infarct. NeuroRehabilitation. 2007;22:273–7.
Koyama T, Tsuji M, Miyake H, Ohmura T, Domen K. Motor outcome for patients with acute intracerebral hemorrhage predicted using diffusion tensor imaging: an application of ordinal logistic modeling. J Stroke Cerebrovasc Dis. 2012;21:704–11.
Puig J, Pedraza S, Blasco G, et al. Wallerian degeneration in the corticospinal tract evaluated by diffusion tensor imaging correlates with motor deficit 30 days after middle cerebral artery ischemic stroke. AJNR Am J Neuroradiol. 2010;31:1324–30.
Moller M, Frandsen J, Andersen G, Gjedde A, Vestergaard-Poulsen P, Ostergaard L. Dynamic changes in corticospinal tracts after stroke detected by fibretracking. J Neurol Neurosurg Psychiatry. 2007;78:587–92.
Radlinska B, Ghinani S, Leppert IR, Minuk J, Pike GB, Thiel A. Diffusion tensor imaging, permanent pyramidal tract damage, and outcome in subcortical stroke. Neurology. 2010;75:1048–54.
Lindenberg R, Zhu LL, Ruber T, Schlaug G. Predicting functional motor potential in chronic stroke patients using diffusion tensor imaging. Hum Brain Mapp. 2012;33:1040–51.
Zhu LL, Lindenberg R, Alexander MP, Schlaug G. Lesion load of the corticospinal tract predicts motor impairment in chronic stroke. Stroke. 2010;41:910–5.
Kuzu Y, Inoue T, Kanbara Y, et al. Prediction of motor function outcome after intracerebral hemorrhage using fractional anisotropy calculated from diffusion tensor imaging. Cerebrovasc Dis. 2012;33:566–73.
Kusano Y, Seguchi T, Horiuchi T, et al. Prediction of functional outcome in acute cerebral hemorrhage using diffusion tensor imaging at 3t: a prospective study. AJNR Am J Neuroradiol. 2009;30:1561–5.
Wang DM, Li J, Liu JR, Hu HY. Diffusion tensor imaging predicts long-term motor functional outcome in patients with acute supratentorial intracranial hemorrhage. Cerebrovasc Dis. 2012;34:199–205.
Koyama T, Tsuji M, Nishimura H, Miyake H, Ohmura T, Domen K. Diffusion tensor imaging for intracerebral hemorrhage outcome prediction: comparison using data from the corona radiata/internal capsule and the cerebral peduncle. J Stroke Cerebrovasc Dis. 2013;22:72–9.
Kothari RU, Brott T, Broderick JP, et al. The ABCs of measuring intracerebral hemorrhage volumes. Stroke. 1996;27:1304–5.
Patel N, Rao VA, Heilman-Espinoza ER, Lai R, Quesada RA, Flint AC. Simple and reliable determination of the modified ranking scale score in neurosurgical and neurological patients: The mrs-9q. Neurosurgery. 2012;71:971–5 discussion 975.
Jung YJ, Jang SH. The fate of injured corticospinal tracts in patients with intracerebral hemorrhage: diffusion tensor imaging study. AJNR Am J Neuroradiol. 2012;33(9):1775–8.
Kwak SY, Son SM, Choi BY, et al. Prognostic factors for motor outcome in patients with compressed corticospinal tract by intracerebral hematoma. NeuroRehabilitation. 2011;29:85–90.
Yoshioka H, Horikoshi T, Aoki S, et al. Diffusion tensor tractography predicts motor functional outcome in patients with spontaneous intracerebral hemorrhage. Neurosurgery. 2008;62:97–103 discussion 103.
Haris M, Gupta RK, Husain N, Hasan KM, Husain M, Narayana PA. Measurement of DTI metrics in hemorrhagic brain lesions: possible implication in MRI interpretation. J Magn Reson Imaging. 2006;24:1259–68.
Clarke JL, Johnston SC, Farrant M, Bernstein R, Tong D, Hemphill JC III. External validation of the ICH score. Neurocrit Care. 2004;1:53–60.
Cheung RT, Zou LY. Use of the original, modified, or new intracerebral hemorrhage score to predict mortality and morbidity after intracerebral hemorrhage. Stroke. 2003;34:1717–22.
Funding
Drs. Wen-Dan Tao and Ming Liu are supported in part by the research grants from the National Natural Science Foundation of P.R. China (Grant No. 81371282) and Construct Program of Key Clinical Disciplines of the Ministry of Health of P.R. China (Project 010202). Dr. Selim receives grant support (U01 NS 074425) from the NIH/NINDS.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tao, WD., Wang, J., Schlaug, G. et al. A Comparative Study of Fractional Anisotropy Measures and ICH Score in Predicting Functional Outcomes After Intracerebral Hemorrhage. Neurocrit Care 21, 417–425 (2014). https://doi.org/10.1007/s12028-014-9999-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-014-9999-2