Abstract
Introduction
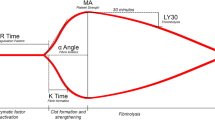
Traumatic brain injury (TBI) is associated with a hypercoagulable state, the mechanism and duration of which remain unclear. We sought to determine whether thromboelastography (TEG) analysis could identify the hypercoagulable state after TBI, as defined by elevations in maximal amplitude (MA), thrombus generation (TG), G value (G), and alpha angle (αA).
Methods
Patients with moderate–severe TBI, defined primarily as a GCS <12, admitted between 1/2012 and 8/2013 were eligible for enrolment in this prospective cohort study. TEG profiles were obtained between 0–24 h (T1), 24–48 h (T2), 48–72 h (T3), 72–96 h (T4), and 96–120 h (T5) after admission. Early TEG was defined as 0–48 h, and late TEG was defined as >48 h.
Results
Twenty five patients (80 % men) and 7 age- and sex-matched control subjects were studied. Median age was 38 years (range 18–85). Early MA was [63.6 mm (60.5, 67.4)] versus late MA [69.9 mm (65.2,73.9); p = 0.02], early TG was [763.3 mm/min (712.8, 816.2)] versus late TG [835.9 mm/min (791.2,888.3); p = 0.02], and early G was [8.8 d/cm2 (7.7,10.4)] versus late G [11.6 d/cm2 (9.4,14.1); p = 0.02]. Study patients had higher MA (p = 0.02), TG (p = 0.03), and G (p = 0.02) values at T5 compared to controls. There was a linear increase per day of MA by 2.6 mm (p = 0.001), TG 31.9 mm/min (p ≤ 0.001), and G value by 1.3 d/cm2 (p ≤ 0.001) when clustered by pairs in regression analysis. Lower MA values trended toward home discharge (p = 0.08).
Conclusion
The data suggest a progressive and delayed hypercoagulable state observed days after initial TBI. The hypercoagulable state may reflect excess platelet activity.


Similar content being viewed by others
References
Coronado VG, Xu L, Basavaraju SV, McGuire LC, Wald MM, Faul MD, Guzman BR, Hemphill JD. Surveillance for traumatic brain injury-related deaths: United States, 1997–2007. MMWR Surveill Summ. 2011;60(5):1–32.
Shackford SR, Mackersie RC, Holbrook TL, Davis JW, Hollingsworth-Fridlund P, Hoyt DB, Wolf PL. The epidemiology of traumatic death. A population-based analysis. Arch Surg. 1993;128(5):571–5.
Lustenberger T, Relja B, Puttkammer B, Gabazza EC, Geiger E, Takei Y, Morser J, Marzi I. Activated thrombin-activatable fibrinolysis inhibitor (TAFIa) levels are decreased in patients with trauma-induced coagulopathy. Thromb Res. 2013;131(1):e26–30.
Talving P, Benfield R, Hadjizacharia P, Inaba K, Chan LS, Demetriades D. Coagulopathy in severe traumatic brain injury: a prospective study. J Trauma. 2009;66(1):55–61 discussion 61-52.
Laroche M, Kutcher ME, Huang MC, Cohen MJ, Manley GT. Coagulopathy after traumatic brain injury. Neurosurgery. 2012;70(6):1334–45.
Lustenberger T, Talving P, Kobayashi L, Inaba K, Lam L, Plurad D, Demetriades D. Time course of coagulopathy in isolated severe traumatic brain injury. Injury. 2010;41(9):924–8.
Maegele M. Coagulopathy after traumatic brain injury: incidence, pathogenesis, and treatment options. Transfusion. 2013;53(Suppl 1):28S–37S.
Kunio NR, Differding JA, Watson KM, Stucke RS, Schreiber MA. Thrombelastography-identified coagulopathy is associated with increased morbidity and mortality after traumatic brain injury. Am J Surg. 2012;203(5):584–8.
Stein SC, Spettell C, Young G, Ross SE. Delayed and progressive brain injury in closed-head trauma: radiological demonstration. Neurosurgery. 1993;32(1):25–30 discussion 30-21.
Harhangi BS, Kompanje EJ, Leebeek FW, Maas AI. Coagulation disorders after traumatic brain injury. Acta Neurochir (Wien). 2008;150(2):165–75 discussion 175.
Stein SC, Smith DH. Coagulopathy in traumatic brain injury. Neurocrit Care. 2004;1(4):479–88.
Cohen MJ, Brohi K, Ganter MT, Manley GT, Mackersie RC, Pittet JF. Early coagulopathy after traumatic brain injury: the role of hypoperfusion and the protein C pathway. J Trauma. 2007;63(6):1254–61 discussion 1261-1252.
Stein SC, Graham DI, Chen XH, Smith DH. Association between intravascular microthrombosis and cerebral ischemia in traumatic brain injury. Neurosurgery. 2004;54(3):687–91 discussion 691.
Graham DI, Adams JH. Ischaemic brain damage in fatal head injuries. Lancet. 1971;1(7693):265–6.
Graham DI, Adams JH, Doyle D. Ischaemic brain damage in fatal non-missile head injuries. J Neurol Sci. 1978;39(2–3):213–34.
Knudson MM, Ikossi DG, Khaw L, Morabito D, Speetzen LS. Thromboembolism after trauma: an analysis of 1602 episodes from the American College of Surgeons National Trauma Data Bank. Ann Surg. 2004;240(3):490–6 discussion 496-498.
Phelan HA. Pharmacologic venous thromboembolism prophylaxis after traumatic brain injury: a critical literature review. J Neurotrauma. 2012;29(10):1821–8.
Rogers FB. Venous thromboembolism in trauma patients: a review. Surgery. 2001;130(1):1–12.
Gonzalez E, Pieracci FM, Moore EE, Kashuk JL. Coagulation abnormalities in the trauma patient: the role of point-of-care thromboelastography. Semin Thromb Hemost. 2010;36(7):723–37.
Windelov NA, Welling KL, Ostrowski SR, Johansson PI. The prognostic value of thrombelastography in identifying neurosurgical patients with worse prognosis. Blood Coagul Fibrinolysis. 2011;22(5):416–9.
Cotton BA, Minei KM, Radwan ZA, Matijevic N, Pivalizza E, Podbielski J, Wade CE, Kozar RA, Holcomb JB. Admission rapid thrombelastography predicts development of pulmonary embolism in trauma patients. J Trauma Acute Care Surg. 2012;72(6):1470–5 discussion 1475-1477.
Branco BC, Inaba K, Ives C, Okoye O, Shulman I, David JS, Schochl H, Rhee P, Demetriades D. Thromboelastogram evaluation of the impact of hypercoagulability in trauma patients. Shock. 2013;41(3):200–7.
Kashuk JL, Moore EE, Sabel A, Barnett C, Haenel J, Le T, Pezold M, Lawrence J, Biffl WL, Cothren CC, et al. Rapid thrombelastography (r-TEG) identifies hypercoagulability and predicts thromboembolic events in surgical patients. Surgery. 2009;146(4):764–72 discussion 772-764.
Kashuk JL, Moore EE, Wohlauer M, Johnson JL, Pezold M, Lawrence J, Biffl WL, Burlew CC, Barnett C, Sawyer M, et al. Initial experiences with point-of-care rapid thrombelastography for management of life-threatening postinjury coagulopathy. Transfusion. 2012;52(1):23–33.
Gonzalez E, Kashuk JL, Moore EE, Silliman CC. Differentiation of enzymatic from platelet hypercoagulability using the novel thrombelastography parameter delta (delta). The Journal of surgical research. 2010;163(1):96–101.
Nielsen VG. Beyond cell based models of coagulation: analyses of coagulation with clot “lifespan” resistance-time relationships. Thromb Res. 2008;122(2):145–52.
Sorensen B, Johansen P, Christiansen K, Woelke M, Ingerslev J. Whole blood coagulation thrombelastographic profiles employing minimal tissue factor activation. J Thromb Haemost. 2003;1(3):551–8.
Rivard GE, Brummel-Ziedins KE, Mann KG, Fan L, Hofer A, Cohen E. Evaluation of the profile of thrombin generation during the process of whole blood clotting as assessed by thrombelastography. J Thromb Haemost. 2005;3(9):2039–43.
Rating the severity of tissue damage. I. The abbreviated scale. JAMA 1971, 215(2):277–280
Marshall LF, Marshall SB, Klauber MR, Clark MV, Eisenberg HM, Jane JA, Luerssen TG, Marmarou A, Foulkes MA. A new classification of head-injury based on computerized-tomography. J Neurosurg. 1991;75:S14–20.
Maas AI, Hukkelhoven CW, Marshall LF, Steyerberg EW. Prediction of outcome in traumatic brain injury with computed tomographic characteristics: a comparison between the computed tomographic classification and combinations of computed tomographic predictors. Neurosurgery. 2005;57(6):1173–82 discussion 1173-1182.
Haemonetics TBMC: Modules 1–6. In: Basic clinical training. Haemonetics; 2008–2010
Abrahams JM, Torchia MB, McGarvey M, Putt M, Baranov D, Sinson GP. Perioperative assessment of coagulability in neurosurgical patients using thromboelastography. Surg Neurol. 2002;58(1):5–11 discussion 11-12.
McCrath DJ, Cerboni E, Frumento RJ, Hirsh AL, Bennett-Guerrero E. Thromboelastography maximum amplitude predicts postoperative thrombotic complications including myocardial infarction. Anesth Analg. 2005;100(6):1576–83.
Gurbel PA, Bliden KP, Guyer K, Cho PW, Zaman KA, Kreutz RP, Bassi AK, Tantry US. Platelet reactivity in patients and recurrent events post-stenting: results of the PREPARE POST-STENTING Study. J Am Coll Cardiol. 2005;46(10):1820–6.
Reiff DA, Haricharan RN, Bullington NM, Griffin RL, McGwin G Jr, Rue LW 3rd. Traumatic brain injury is associated with the development of deep vein thrombosis independent of pharmacological prophylaxis. J Trauma. 2009;66(5):1436–40.
Geerts WH, Code KI, Jay RM, Chen E, Szalai JP. A prospective study of venous thromboembolism after major trauma. N Engl J Med. 1994;331(24):1601–6.
Brohi K, Cohen MJ, Ganter MT, Matthay MA, Mackersie RC, Pittet JF. Acute traumatic coagulopathy: initiated by hypoperfusion: modulated through the protein C pathway? Ann Surg. 2007;245(5):812–8.
Brohi K, Cohen MJ, Davenport RA. Acute coagulopathy of trauma: mechanism, identification and effect. Curr Opin Crit Care. 2007;13(6):680–5.
Wafaisade A, Lefering R, Tjardes T, Wutzler S, Simanski C, Paffrath T, Fischer P, Bouillon B, Maegele M. Acute coagulopathy in isolated blunt traumatic brain injury. Neurocrit Care. 2010;12(2):211–9.
Wegner J, Popovsky MA. Clinical utility of thromboelastography: one size does not fit all. Semin Thromb Hemost. 2010;36(7):699–706.
Holcomb JB, Minei KM, Scerbo ML, Radwan ZA, Wade CE, Kozar RA, Gill BS, Albarado R, McNutt MK, Khan S, et al. Admission rapid thrombelastography can replace conventional coagulation tests in the emergency department: experience with 1974 consecutive trauma patients. Ann Surg. 2012;256(3):476–86.
Acknowlegments
Dr. Kumar has received research support from Haemonetics. Dr. Kasner has received grants from WL Gore, AstraZeneca, GlaxoSmithKline, and Acorda. He acts as a consultant for Merck, Novartis, Medtronic, and Boehringer Ingelheim. Dr. Grady has received Grant funding from the NIH. For the remaining authors, none were declared.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Massaro, A.M., Doerfler, S., Nawalinski, K. et al. Thromboelastography Defines Late Hypercoagulability After TBI: A Pilot Study. Neurocrit Care 22, 45–51 (2015). https://doi.org/10.1007/s12028-014-0051-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-014-0051-3