Abstract
Background
Brain oxygen (PbtO2) monitoring can help guide care of poor-grade aneurysmal subarachnoid hemorrhage (aSAH) patients. The relationship between PbtO2-directed therapy and long-term outcome is unclear. We hypothesized that responsiveness to PbtO2-directed interventions is associated with outcome.
Methods
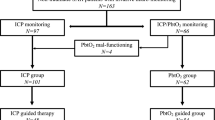
Seventy-six aSAH patients who underwent PbtO2 monitoring were included. Long-term outcome [Glasgow Outcome Score-Extended (GOS-E) and modified Rankin Scale (mRS)] was ascertained using the social security death database and structured telephone interviews. Univariate and multivariate regression were used to identify variables that correlated with outcome.
Results
Data from 64 patients were analyzed (12 were lost to follow-up). There were 530 episodes of compromised PbtO2 (<20 mmHg) during a total of 7,174 h of monitor time treated with 1,052 interventions. Forty-two patients (66 %) survived to discharge. Median follow-up was 8.5 months (range 0.1–87). At most recent follow-up 35 (55 %) patients were alive, and 28 (44 %) had a favorable outcome (mRS ≤3). In multivariate ordinal regression analysis, only age and response to PbtO2-directed intervention correlated significantly with outcome. Increased age was associated with worse outcome (coeff. 0.8, 95 % CI 0.3–1.3, p = 0.003), and response to PbtO2-directed intervention was associated with improved outcome (coeff. −2.12, 95 % CI −4.0 to −0.26, p = 0.03). Patients with favorable outcomes had a 70 % mean rate of response to PbtO2-directed interventions whereas patients with poor outcomes had a 45 % response rate (p = 0.005).
Conclusions
Response to PbtO2-directed intervention is associated with improved long-term functional outcome in aSAH patients.


Similar content being viewed by others
References
Bederson JB, Connolly ES Jr, Batjer HH, et al. Guidelines for the management of aneurysmal subarachnoid hemorrhage: a statement for healthcare professionals from a special writing group of the Stroke Council, American Heart Association. Stroke. 2009;40(3):994–1025.
Broderick JP, Thoma GB, Dudler JE, Tomsick T, Leach A. Initial and recurrent bleeding are the major causes of death following subarachnoid hemorrhage. Stroke. 1994;25:1342–7.
Johnston SC, Selvin S, Gress DR. The burden, trends, and demographics of mortality from subarachnoid hemorrhage. Neurology. 1998;50(5):1413–8.
Drake CG. Report of World Federation of Neurosurgical Societies Committee on universal subarachnoid hemorrhage grading scale. J Neurosurg. 1988;38:575–80.
Hunt WE, Hess RM. Surgical risk related to time of intervention in the repair of intracranial aneurysms. J Neurosurg. 1968;24:14–9.
Hutchinson PJ, Power DM, Tripathi P, Kirkpatrick PJ. Outcome from poor grade aneurysmal subarachnoid haemorrhage—which poor grade subarachnoid haemorrhage patients benefit from aneurysm clipping? Br J Neurosurg. 2000;14:105–9.
Le Roux P, Elliott JP, Newell DW, Grady MS, Winn HR. Predicting outcome in poor grade subarachnoid hemorrhage: a retrospective review of 159 aggressively managed patients. J Neurosurg. 1996;85:39–49.
Le Roux P, Winn HR. The poor grade aneurysm patient. Acta Neurochir Suppl. 1999;72:7–26.
Shirao S, Yoneda H, Kunitsugu I, et al. Preoperative prediction of outcome in 283 poor-grade patients with subarachnoid hemorrhage: a project of the Chugoku-Shikoku Division of the Japan Neurosurgical Society. Cerebrovasc Dis. 2010;30(2):105–13.
Sandsmark DK, Kumar MA, Park S, Levine JM. Multimodal monitoring in subarachnoid hemorrhage. Stroke. 2012;43(5):1440–5.
Meixensberger J, Vath A, Jaeger M, Kunze E, Dings J, Roosen K. Monitoring of brain tissue oxygenation following severe subarachnoid hemorrhage. Neurol Res. 2003;25(5):445–50.
Narotam PK, Morrison JF, Nathoo N. Brain tissue oxygen monitoring in traumatic brain injury and major trauma: outcome analysis of a brain tissue oxygen-directed therapy. J Neurosurg. 2009;111(4):672–82.
Spiotta AM, Stiefel MF, Gracias VH, et al. Brain tissue oxygen-directed management and outcome in patients with severe traumatic brain injury. J Neurosurg. 2010;113(3):571–80.
Stiefel MF, Spiotta A, Gracias VH, et al. Reduced mortality rate in patients with severe traumatic brain injury treated with brain tissue oxygen monitoring. J Neurosurg. 2005;103(5):805–11.
Stiefel MF, Udoetuk JD, Spiotta AM, et al. Conventional neurocritical care and cerebral oxygenation after traumatic brain injury. J Neurosurg. 2006;105(4):568–75.
Martini RP, Deem S, Yanez ND, et al. Management guided by brain tissue oxygen monitoring and outcome following severe traumatic brain injury. J Neurosurg. 2009;111(4):644–9.
Brain Trauma Foundation, American Association of Neurological Surgeons, Congress of Neurological Surgeons. Guidelines for the management of severe traumatic brain injury. J Neurotrauma. 2007;24(Suppl 1):S1–106.
Ramakrishna R, Stiefel M, Udoetuk J, et al. Brain oxygen tension and outcome in patients with aneurysmal subarachnoid hemorrhage. J Neurosurg. 2008;109(6):1075–82.
McKhann GM, Le Roux PD. Perioperative and intensive care unit care of patients with aneurysmal subarachnoid hemorrhage. Neurosurg Clin N Am. 1998;9(3):595–613.
Chen HI, Stiefel MF, Oddo M, et al. Detection of cerebral compromise with multimodality monitoring in patients with subarachnoid hemorrhage. Neurosurgery. 2011;69(1):53–63; discussion 63.
Frontera JA, Claassen J, Schmidt JM, et al. Prediction of symptomatic vasospasm after subarachnoid hemorrhage: the modified fisher scale. Neurosurgery. 2006;59(1):21–7; discussion 21–7.
Oddo M, Levine JM, Frangos S, Maloney-Wilensky E, LeRoux PD. Correlation between brain oxygen and cerebral perfusion pressure and its impact on outcome after severe traumatic brain injury. 2013. (unpublished data).
Jaeger M, Dengl M, Meixensberger J, Schuhmann MU. Effects of cerebrovascular pressure reactivity-guided optimization of cerebral perfusion pressure on brain tissue oxygenation after traumatic brain injury. Crit Care Med. 2010;38(5):1343–7.
Aries MJH, Czosnyka M, Budohoski KP, et al. Continuous determination of optimal cerebral perfusion pressure in traumatic brain injury. Crit Care Med. 2012;40(8):2456–63.
Navi BB, Kamel H, Hemphill JC III, Smith WS. Trajectory of functional recovery after hospital discharge for subarachnoid hemorrhage. Neurocrit Care. 2012;17(3):343–7.
Rordorf G, Ogilvy CS, Gress DR, Crowell RM, Choi IS. Patients in poor neurological condition after subarachnoid hemorrhage: early management and long-term outcome. Acta Neurochir. 1997;139(12):1143–51.
Mocco J, Ransom ER, Komotar RJ, et al. Preoperative prediction of long-term outcome in poor-grade aneurysmal subarachnoid hemorrhage. Neurosurgery. 2006;59(3):529–38.
Malmivaara K, Juvela S, Hernesniemi J, Lappalainen J, Siironen J. Health-related quality of life and cost-effectiveness of treatment in subarachnoid haemorrhage. Eur J Neurol. 2012;19(11):1455–61.
Ponce LL, Pillai S, Cruz J, et al. Position of probe determines prognostic information of brain tissue PO2 in severe traumatic brain injury. Neurosurgery. 2012;70(6):1492–3.
Fletcher JJ, Bergman K, Blostein PA, Kramer AH. Fluid balance, complications, and brain tissue oxygen tension monitoring following severe traumatic brain injury. Neurocrit Care. 2010;13(1):47–56.
Kim DH, Haney CL, Van Ginhoven G. Utility of outcome measures after treatment for intracranial aneurysms: a prospective trial involving 520 patients. Stroke. 2005;36(4):792–6.
Longhi L, Pagan F, Valeriani V, et al. Monitoring brain tissue oxygen tension in brain-injured patients reveals hypoxic episodes in normal-appearing and in peri-focal tissue. Intensive Care Med. 2007;33(12):2136–42.
Kiening KL, Unterberg AW, Bardt TF, Schneider GH, Lanksch WR. Monitoring of cerebral oxygenation in patients with severe head injuries: brain tissue PO2 versus jugular vein oxygen saturation. J Neurosurg. 1996;85(5):751–7.
Gupta AK, Hutchinson PJ, Al-Rawi P, et al. Measuring brain tissue oxygenation compared with jugular venous oxygen saturation for monitoring cerebral oxygenation after traumatic brain injury. Anesth Analg. 1999;88(3):549–53.
Gopinath SP, Valadka AB, Uzura M, Robertson CS. Comparison of jugular venous oxygen saturation and brain tissue PO2 as monitors of cerebral ischemia after head injury. Crit Care Med. 1999;27(11):2337–45.
Sarrafzadeh AS, Kiening KL, Bardt TF, Schneider GH, Unterberg AW, Lanksch WR. Cerebral oxygenation in contusioned vs. nonlesioned brain tissue: monitoring of PtiO2 with Licox and Paratrend. Acta Neurochir Suppl. 1998;71:186–9.
Bohman LE, Heuer GG, Macyszyn L, et al. Medical management of compromised brain oxygen in patients with severe traumatic brain injury. Neurocrit Care. 2011;14(3):361–9.
Oddo M, Levine JM, Mackenzie L, et al. Brain hypoxia is associated with short-term outcome after severe traumatic brain injury independently of intracranial hypertension and low cerebral perfusion pressure. Neurosurgery. 2011;69(5):1037–45; discussion 1045.
Brain Tissue Oxygen Monitoring in Traumatic Brain Injury (TBI) (BOOST 2), ClinicalTrials.gov Identifier: NCT00974259.
Acknowledgments
Supported by Research Grants from the Integra Foundation (PDL), Integra Neurosciences (PDL), and the Mary Elisabeth Groff Surgical and Medical Research Trust (PDL). PDL is a member of Integra’s Speaker’s Bureau and a Consultant for Integra LifeSciences.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bohman, LE., Pisapia, J.M., Sanborn, M.R. et al. Response of Brain Oxygen to Therapy Correlates with Long-Term Outcome After Subarachnoid Hemorrhage. Neurocrit Care 19, 320–328 (2013). https://doi.org/10.1007/s12028-013-9890-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-013-9890-6