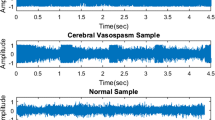
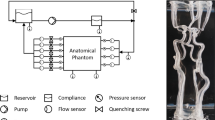
Abstract
Understanding the dynamic relationship between cerebral blood flow (CBF) and the circulation of cerebrospinal fluid (CSF) can facilitate management of cerebral pathologies. For this reason, various hydrodynamic models have been introduced in order to simulate the phenomena governing the interaction between CBF and CSF. The identification of hydrodynamic models requires an array of signals as input, with the most common of them being arterial blood pressure, intracranial pressure, and cerebral blood flow velocity; monitoring all of them is considered as a standard practice in neurointensive care. Based on these signals, physiological parameters like cerebrovascular resistance, compliances of cerebrovascular bed, and CSF space could then be estimated. Various secondary model-based indices describing cerebrovascular dynamics have been introduced, like the cerebral arterial time constant or critical closing pressure. This review presents model-derived indices that describe cerebrovascular phenomena, the nature of which is both physiological (carbon dioxide reactivity and arterial hypotension) and pathological (cerebral artery stenosis, intracranial hypertension, and cerebral vasospasm). In a neurointensive environment, real-time monitoring of a patient with these indices may be able to provide a detection of the onset of a cerebrovascular phenomenon, which could have otherwise been missed. This potentially “early warning” indicator may then prove to be important for the therapeutic management of the patient.

















Similar content being viewed by others
References
Krammer SP. On the function of the circle of Willis. J Exp Med. 1912;15:348–54.
Rogers L. A dynamic model of the circle of Willis. J Biomech. 1947;4:141–7.
Avman N, Bering EA. A plastic model for the study of pressure changes in the circle of Willis and major cerebral arteries following arterial occlusion. J Neurosurg. 1961;21:361–5.
Himwich WA, Knapp FM, Wenglarz RA, et al. The circle of Willis as simulated by an engineering model. Arch Neurol. 1965;13:164–72.
Agarwal G, Berman B, Stark L. A lumped parameter model of the cerebrospinal system. IEEE Trans Biomed Eng BME. 1969;16:45–53.
Murray KD. Dimensions of the circle of Willis and dynamic studies using electrical analogy. J Neurosurg. 1964;21:26–34.
Sorek S, Bear J, Karni Z. Resistances and compliances of a compartmental model of the cerebrovascular system. Ann Biomed Eng. 1989;17:1–12.
Takemae T, Kosugi Y, Ikebe J, et al. A simulation study of intracranial-pressure increment using an electrical circuit model of cerebral-circulation. IEEE Trans Biomed Eng. 1987;34:958–62.
Bekker A, Wolk S, Turndorf H, et al. Computer simulation of cerebrovascular circulation: assessment of intracranial hemodynamics during induction of anesthesia. J Clin Monit. 1996;12:433–44.
Czosnyka M, Harris NG, Pickard JD, et al. CO2 cerebrovascular reactivity as a function of perfusion pressure—a modeling study. Acta Neurochir Wien. 1993;121:159–65.
Czosnyka M, Pickard J, Whitehouse H, et al. The hyperemic response to a transient reduction in cerebral perfusion-pressure—a modelling study. Acta Neurochir Wien. 1992;115:90–7.
Hoffmann O. Biomathematics of intracranial CSF and haemodynamics. Simulation and analysis with the aid of a mathematical model. Acta Neurochir Suppl Wien. 1987;40:117–30.
Piechnik S, Czosnyka M, Richards H, et al. Effects of decreasing cerebral perfusion pressure on pulsatility of cerebral blood flow velocity—a modelling study. In: Nagai H, Kamiya K, Ishii S, editors. Intracranial pressure IX. Berlin: Springer; 1994. p. 496–7.
Ursino M. A mathematical study of human intracranial hydrodynamics. Part 1—the cerebrospinal fluid pulse pressure. Ann Biomed Eng. 1988;16:379–401.
Ursino M. Computer analysis of the main parameters extrapolated from the human intracranial basal artery blood flow. Comput Biomed Res. 1990;23:542–59.
Ursino M. A mathematical model of overall cerebral blood flow regulation in the rat. IEEE Trans Biomed Eng. 1991;38:795–807.
Ursino M, Cristalli C. Mathematical modeling of noninvasive blood pressure estimation techniques—part I: pressure transmission across the arm tissue. J Biomech Eng. 1995;117:107–16.
Ursino M, Di Giammarco P. A mathematical model of the relationship between cerebral blood volume and intracranial pressure changes: the generation of plateau waves. Ann Biomed Eng. 1991;19:15–42.
Ursino M, Lodi CA. A simple mathematical model of the interaction between intracranial pressure and cerebral hemodynamics. J Appl Physiol. 1997;82:1256–69.
Ursino M, Lodi CA, Rossi S, et al. Intracranial pressure dynamics in patients with acute brain damage. J Appl Physiol. 1997;82:1270–82.
Tym R, Lichtenstein S, Leutheusser J. The Munro-Kellie doctrine and the intracranial venous space at the ‘limit’ of raised intracranial pressure—an hydrodynamic experimental approach. In: Brock M, Dietz H, editors. Intracranial pressure—experimental and clinical aspects. Berlin: Springer; 1972. p. 139–43.
Hoffmann O, Zierski JT. Analysis of the ICP pulse-pressure relationship as a function of arterial blood pressure. Clinical validation of a mathematical model. Acta Neurochir Wien. 1982;66:1–21.
Nishimura H, Yasui N. A simulation study of wave transformation using a nonlinear model of artery and a physical model of intracranial vascular bed. Berlin: Springer; 1993. p. 390–393.
Chopp M, Portnoy HD, Branch C. Hydraulic model of the cerebrovascular bed: an aid to understanding the volume–pressure test. J Neurosurg. 1983;13:5–11.
Giulioni M, Ursino M. Impact of cerebral perfusion pressure and autoregulation on intracranial dynamics: a modeling study. J Neurosurg. 1996;39:1005–14.
Ursino M, Di Giammarco P, Belardinelli E. A mathematical model of cerebral blood flow chemical regulation—part II: reactivity of cerebral vascular bed. IEEE Trans Biomed Eng. 1989;36:192–201.
Czosnyka M, Piechnik S, Richards HK, et al. Contribution of mathematical modelling to the interpretation of bedside tests of cerebrovascular autoregulation. J Neurol Neurosurg Psychiatry. 1997;63:721–31.
Czosnyka M, Smielewski P, Timofeev I, et al. Intracranial pressure: more than a number. Neurosurg Focus. 2007;22(5):E10.
Sorrentino E, Budohoski KP, Kasprowicz M, et al. Critical thresholds for Transcranial Doppler indices of cerebral autoregulation in traumatic brain injury. Neurocrit Care. 2010;14(2):188–93.
Zweifel C, Lavinio A, Steiner LA, et al. Continuous monitoring of cerebrovascular pressure reactivity in patients with head injury. Neurosurg Focus. 2008;25(4):E2.
Balestreri M, Czosnyka M, Hutchinson P, et al. Impact of intracranial pressure and cerebral perfusion pressure on severe disability and mortality after head injury. Neurocrit Care. 2006;04:8–13.
Gopinath SP, Robertson CS, Narayan RG, et al. Evaluation of a microsensor intracranial pressure transducer. In: Nagai H, Kamiya K, Ishii S, editors. Intracranial pressure IX. Berlin: Springer; 1994. p. 2–5.
Czosnyka M, Czosnyka Z, Pickard JD. Laboratory testing of three intracranial pressure microtransducers: technical report. J Neurosurg. 1996;38(1):219–24.
Kirkpatrick PJ, Czosnyka M, Pickard JD. Multimodal monitoring in neurointensive care. J Neurol Neurosurg Psychiatry. 1996;60:131–9.
Carrera E, Kim DJ, Castellani G, et al. Effect of hyper- and hypocapnia on cerebral arterial compliance in normal subjects. J Neuroimaging. 2009;21(2):121–5.
Sloan MA, Alexandrov AV, Tegeler CH, et al. Transcranial Doppler ultrasonography in 2004: a comprehensive evidence-based update. Report of the American Academy of Neurology Therapeutics and Technology Assessment Subcommittee: Transcranial Doppler. J Neurol. 1990;40:680–1.
Aaslid R, Markwalder TM, Nornes H. Noninvasive transcranial Doppler recording of flow velocity in basal cerebral arteries. J Neurosurg. 1982;57:769–74 Class II–III.
Matta B, Czosnyka M. Transcranial Doppler ultrasonography in anesthesia and neurosurgery. Cottrell and Young’s Neuroanesthesia, Chapter 8. Philadelphia: Elsevier; 2010.
Aaslid R. Transcranial Doppler examination techniques. In: Aaslid R, editor. Transcranial Doppler sonography. New York: Springer; 1986. p. 39.
Nelson RJ, Czosnyka M, Pickard JD, et al. Experimental aspects of cerebrospinal haemodynamics: the relationship between blood flow velocity waveform and cerebral autoregulation. J Neurosurg. 1992;31:705–10.
Czosnyka M, Richards HK, Reinhard M, et al. Cerebrovascular time constant: dependence on cerebral perfusion pressure and end-tidal carbon dioxide concentration. Neurol Res. 2012;34(1):17–24.
Kasprowicz M, Diedler J, Reinhard M, et al. Time constant of the cerebral arterial bed. Acta Neurochir Suppl. 2012;114:17–21.
Czosnyka M, Richards H, Pickard JD, Harris N, Iyer V. Frequency-dependent properties of cerebral blood transport-an experimental study in anaesthetized rabbits. Ultrasound Med Biol. 1994;20:391–9.
Kim DJ, Kasprowicz M, Carrera E, et al. The monitoring of relative changes in compartmental compliances of brain. Physiol Meas. 2009;30(7):647–59.
Kasprowicz M, Diedler J, Reinhard M, et al. Time constant of the cerebral arterial bed in normal subjects. Ultrasound Med Biol. 2012;38(7):1129–37.
Kasprowicz M, Czosnyka M, Soehle M, et al. Vasospasm shortens cerebral arterial time constant. Neurocrit Care. 2011;16(2):213–8.
Czosnyka M, Smielewski P, Piechnik S, et al. Hemodynamic characterization of intracranial pressure plateau waves in head-injury patients. J Neurosurg. 1999;91(1):11–9.
Avezaat CJJ, van Eijndhoven JHM. Cerebrospinal fluid pulse pressure and craniospinal dynamics. A theoretical clinical and experimental study. Thesis. The Hague: A Jongbloed, 1984
Toth M, Nadasy GL, Nyar I, Kerenyi T, Monos E. Are there systemic changes in the arterial biomechanics of intracranial aneurysm patients? Pflugers Arch. 2000;439:573–8.
Alperin N, Lee SH, Sivaramakrishnan A, Hushek SG. Quantifying the effect of posture on intracranial physiology in humans by MRI flow studies. J Magn Reson Imaging. 2005;22:591–6.
Carrera E, Kim DJ, Castellani G, et al. What shapes pulse amplitude of intracranial pressure? J Neurotrauma. 2010;27:317–24.
Eide PK, Sorteberg A, Bentsen G, Marthinsen PB, Stubhaug A, Sorteberg W. Pressure-derived versus pressure wave amplitude-derived indices of cerebrovascular pressure reactivity in relation to early clinical state and 12-month outcome following aneurysmal subarachnoid hemorrhage. J Neurosurg. 2012;116(5):961–71.
Eide PK. Assessment of childhood intracranial pressure recordings using a new method of processing intracranial pressure signals. Pediatr Neurosurg. 2005;41(3):122–30.
Radolovich DK, Aries MJ, Castellani G, et al. Pulsatile intracranial pressure and cerebral autoregulation after traumatic brain injury. Neurocrit Care. 2011;15(3):379–86.
Czosnyka M, Richards HK, Whitehouse HE, Pickard JD. Relationship between transcranial Doppler-determined pulsatility index and cerebrovascular resistance: an experimental study. J Neurosurg. 1996;84(1):79–84.
Gosling RG, King DH. Arterial assessment by Doppler-shift ultrasound. Proc R Soc Med. 1974;67(6 Pt 1):447–9.
Michel E, Zernikow B. Goslig’s Doppler pulsatility index revisited. Ultrasound Med Biol. 1998;24(4):597–9.
de Riva N, Budohoski KP, Smielewski P, et al. Transcranial Doppler pulsatility index: what it is and what it isn’t. Neurocrit Care. 2012;17(1):58–66.
O’Rourke MF, Taylor MG. Input impedance of the systemic circulation. Circ Res. 1967;20(4):365–80.
Giller CA, Hodges K, Batjer HH. Transcranial Doppler pulsatility in vasodilation and stenosis. J Neurosurg. 1990;72(6):901–6.
Behrens A, Lenfeldt N, Ambarki K, Malm J, Eklund A, Koskinen LO. Transcranial Doppler pulsatility index: not an accurate method to assess intracranial pressure. J Neurosurg. 2010;66(6):1050–7.
Bellner J, Romner B, Reinstrup P, Kristiansson KA, Ryding E, Brandt L. Transcranial Doppler sonography pulsatility index (PI) reflects intracranial pressure (ICP). Surg Neurol. 2004;62(1):45–51.
Burton AC. Fundamental instability of the small blood vessels and critical closing pressure in vascular beds. Am J Physiol. 1951;164:330–1.
Brunner MJ, Greene AS, Sagawa K, Shoukas AA. Determinants of systemic zero-flow arterial pressure. Am J Physiol. 1983;245:H453–9.
Czosnyka M, Smielewski P, Piechnik S, et al. Critical closing pressure in cerebrovascular circulation. J Neurol Neurosurg Psychiatry. 1999;66:606–11.
Panerai RB. The critical closing pressure of the cerebral circulation. Med Eng Phys. 2003;25:621–32.
Dewey RC, Pierer HP, Hunt WE. Experimental cerebral hemodynamics. Vasomotor tone, critical closing pressure, and vascular bed resistance. J Neurosurg. 1974;41:597–606.
Panerai RB, Moody M, Eames PJ, Potter JF. Cerebral blood flow velocity during mental activation: interpretation with different models of the passive pressure–velocity relationship. J Appl Physiol. 2005;99:2352–62.
Aaslid R, Lash SR, Bardy GH, Gild WH, Newell DW. Dynamic pressure-flow velocity relationships in the human cerebral circulation. Stroke. 2003;34(7):1645–9.
Newell DW, Aaslid R. Transcranial Doppler: clinical and experimental uses. Cerebrovasc Brain Metab Rev. 1992;4(2):122–43.
López-Magaña JA, Richards HK, Radolovich DK, et al. Critical closing pressure: comparison of three methods. J Cereb Blood Flow Metab. 2009;29:987–93.
Panerai RB, et al. Influence of calculation method on estimates of cerebral critical closing pressure. Physiol Meas. 2011;32:1–16.
Michel E, Hillebrand S, von Twickel J, et al. Frequency dependence of cerebrovascular impedance in preterm neonates: a different view on critical closing pressure. J Cereb Blood Flow Metab. 1997;17:1127–31.
Puppo C, Camacho J, Yelicich B, Moraes L, Biestro A, Gomez H. Bedside study of cerebral critical closing pressure in patients with severe traumatic brain injury: a Transcranial Doppler study. Acta Neurosurg Suppl. 2012;114:283–8.
Soehle M, Czosnyka M, Pickard JD, Kirkpatrick PJ. Critical closing pressure in subarachnoid hemorrhage: effect of cerebral vasospasm and limitations of a transcranial Doppler-derived estimation. Stroke. 2004;35(6):1393–8.
Varsos GV, Richards H, Kasprowicz M, et al. Critical closing pressure determined with a model of cerebrovascular impedance. J Cereb Blood Flow Metab. 2012;33(2):235–43.
Richards HK, Czosnyka M, Pickard JD. Assessment of critical closing pressure in the cerebral circulation as a measure of cerebrovascular tone. Acta Neurochir Wien. 1999;141(11):1221–7.
Kontos HA, Wei EP, Navari RM, Levasseur JE, Rosenblum WI, Patterson JL Jr. Responses of cerebral arteries and arterioles to acute hypotension and hypertension. Am J Physiol. 1978;234(4):H371–83.
Serrador JM, Picot PA, Rutt BK, Shoemaker JK, Bondar RL. MRI measures of middle cerebral artery diameter in conscious humans during simulated orthostasis. Stroke. 2000;31(7):1672–8.
Giller CA, Bowman G, Dyer H, Mootz L, Krippner W. Cerebral arterial diameters during changes in blood pressure and carbon dioxide during craniotomy. J Neurosurg. 1993;32(5):737–42.
Newell DW, Aaslid R, Lam A, Mayberg TS, Winn HR. Comparison of flow and velocity during dynamic autoregulation testing in humans. Stroke. 1994;25(4):793–7.
Müller HR, Brunhölzl C, Radü EW, Buser M. Sex and side differences of cerebral arterial caliber. Neuroradiology. 1991;33(3):212–6.
Caekebeke JF, Ferrari MD, Zwetsloot CP, Jansen J, Saxena PR. Antimigraine drug sumatriptan increases blood flow velocity in large cerebral arteries during migraine attacks. J Neurology. 1992;42(8):1522–6.
Giller CA, Giller AM, Cooper CR, Hatab MR. Evaluation of the cerebral hemodynamic response to rhythmic handgrip. J Appl Physiol. 2000;88(6):2205–13.
Giller CA, Mueller M. Linearity and non-linearity in cerebral hemodynamics. Med Eng Phys. 2003;25(8):633–46.
Mitsis GD, Zhang R, Levine BD, Marmarelis VZ. Cerebral hemodynamics during orthostatic stress assessed by nonlinear modeling. J Appl Physiol. 2006;101(1):354–66.
Conflict of interests
ICM+ software is licensed by Cambridge Enterprise Ltd (www.neurosurg.cam.ac.uk/icmplus). PS and MC have a share in a fraction of licensing fee. There is no other conflict of interest regarding equipment used for monitoring of clinical and experimental signals.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Varsos, G.V., Kasprowicz, M., Smielewski, P. et al. Model-based Indices Describing Cerebrovascular Dynamics. Neurocrit Care 20, 142–157 (2014). https://doi.org/10.1007/s12028-013-9868-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-013-9868-4