Abstract
Background
Altered brain perfusion may play an important role in the development of sepsis-associated encephalopathy. However, whether or not cerebral autoregulation (CA) is preserved in such condition has been debated. CA is dependent on cerebral vascular tone, the main determinant of which is the concentration of carbon dioxide (CO2). The purpose of this study was to evaluate the influence of PaCO2 on the cerebral autoregulatory capacity in patients with septic shock.
Methods
Using transcranial Doppler sonography recordings from the middle cerebral artery (MCA), we evaluated the static cerebral autoregulatory responses within the first 3 days of septic shock. Changes in cerebrovascular resistance (CVR) were calculated from the changes in the mean velocity in the MCA (VMCA, cm/s), in response to an increase in mean arterial pressure (MAP, mmHg) induced by vasopressors. The cerebral autoregulation index (CAI) was calculated as the ratio of the relative changes in CVR and MAP (CAI = ΔMAP%/ΔCVR%), with normal values ranging between 0 and 2.
Results
We studied 21 mechanically ventilated patients, with a baseline MAP of 65 ± 6 mmHg, a mean VMCA of 60 ± 20 cm/s and a median PaCO2 of 35 [28–49] mmHg. Fourteen of the 21 patients had impaired CA, including 7 of the 14 patients with a PaCO2 <40 mmHg and all 7 patients with a PaCO2 >40 mmHg (Fisher’s exact test, P = 0.046).
Conclusions
According to these data, CA is impaired in the majority of patients with septic shock, especially in the presence of hypercapnia.
Similar content being viewed by others
Introduction
Septic shock and related multiple organ failure (MOF) remain major causes of morbidity and mortality in intensive care units [1]. Acute deterioration in mental status during severe infections, the so-called “sepsis-associated encephalopathy” (SAE), can develop in up to 70% of these patients [2, 3]. Clinical signs of SAE range from mild disorientation to lethargy and coma and there may be associated neurophysiological disturbances [4, 5]. Importantly, SAE can be an early sign of sepsis and may contribute to long-term cognitive complications and increased mortality [6, 7].
The pathogenesis of SAE remains unclear [8]. Various factors have been suggested to contribute to the pathogenesis, including cerebral effects of circulating inflammatory mediators [9], disruption of the blood–brain barrier [10], impaired astrocytic function [11], altered neurotransmission [12], and induced neuronal apoptosis [13]. A retrospective study identified severe hypotension as an important risk factor for SAE and the authors therefore suggested that SAE may be primarily related to ischemic damage rather than to other causes [14]. This concept was supported by an autopsy study in which ischemic lesions were the major pathologic findings in the brain of patients who died in septic shock [15]. Although impairment of tissue perfusion and blood flow redistribution are typically considered as being involved in the pathogenesis of organ failure, it remains unclear whether sepsis can induce profound and sustained brain damage by reducing global or regional cerebral blood flow (CBF).
In healthy individuals, CBF is kept constant over a wide range of mean arterial pressures (MAP). This phenomenon, called cerebral autoregulation, is accomplished by changes in cerebral vascular tone and is generally observed for MAP values between 50 and 150 mmHg [16]. There are conflicting reports regarding disturbances in cerebral autoregulation during sepsis. In a recent study, Pfister et al. [17] reported impaired cerebral autoregulation in 12/16 patients with severe sepsis and septic shock, which was associated with clinical delirium, higher levels of s100β, and worse outcome. However, Matta and Stow [18] reported intact cerebral autoregulation in ten patients with early sepsis. Impaired cerebral autoregulation may leave brain tissue unprotected against possibly harmful effects of blood pressure changes during sepsis, potentially leading to cerebral ischemia.
With its potent vasodilating properties, PaCO2 is an important regulator of the cerebral circulation [19] and hypercapnia can directly increase CBF [20]. Although the vascular response to PaCO2 in the brain has been shown to be markedly attenuated by moderate hypotension in experimental studies [21], Thees et al. recently reported normal reactivity to CO2 in patients with sepsis [22]. Interestingly, changes in CBF induced by changes in PaCO2 can influence the pressure limits of arterial blood pressure, referred to as the autoregulatory plateau, within which cerebral autoregulation operates to keep CBF constant [23]. Thus, during hypercapnia, this autoregulatory plateau is narrowed to between a lower upper limit and a higher lower limit of MAP [24].
However, the role of PaCO2 on cerebral autoregulation during sepsis has not been investigated. The purpose of this study was, therefore, to evaluate cerebral autoregulation in patients with septic shock and to assess the influence of PaCO2 concentrations on the autoregulatory cerebral capacity in these patients.
Patients and Methods
This prospective study was conducted in the 35-bed Department of Intensive Care of a University hospital. The hospital Ethics Committee approved the study protocol, and informed consent was obtained from the patient or their next of kin. We enrolled consecutive adult patients with septic shock, as defined by standard criteria [25], for less than 72 h who were being treated with mechanical ventilation. Exclusion criteria were age <18 years, intracranial infection, a history or clinical evidence of a neurologic disease, or significant arrhythmias. Patients in whom it was difficult to obtain cerebral vessel waveforms using transcranial Doppler (TCD) or who had significant stenoses of the extracranial and intracranial cerebral arteries on echo-Doppler examination were also excluded.
In all patients, demographic data, pre-existing chronic diseases, and admission diagnosis were collected. The Glasgow coma score was used to assess the neurologic status [26] on admission. The source of sepsis and relevant microbiological results were recorded. Acute lung injury/acute respiratory distress syndrome (ALI/ARDS) were diagnosed according to standard criteria [27]. Acute renal failure was defined by a sequential organ failure assessment (SOFA) renal score >2 [28]. Other treatments, including administration of adrenergic agents, hydrocortisone, and activated protein C were also recorded daily. The severity of critical illness was assessed by the Acute Physiology and Chronic Health Evaluation (APACHE) II [29] and SOFA score on the day of the study. Biochemical data, including complete blood count, electrolytes, urea, creatinine, bilirubin, total protein, albumin, and C-reactive protein (CRP) concentrations were recorded on the day of the study. Intensive care unit (ICU) and hospital length of stay, overall mortality and cause of death were noted.
Each patient had continuous monitoring of heart rate, systemic MAP and central venous pressure (CVP). MAP was measured using a radial or femoral arterial catheter; pulmonary artery pressures and cardiac output were continuously measured using the thermal dilution technique via a triple-lumen pulmonary artery catheter (Vigilance; Baxter Edwards Critical-Care, Irvine, CA), previously inserted for diagnostic or therapeutic purposes. Cardiac index (CI) and systemic vascular resistance index (SVRI) were calculated using standard formulas. Arterial and venous blood gases, oxygen saturation, and hemoglobin were determined at baseline (ABL 700; Radiometer, Copenhagen, Denmark). End-tidal expired carbon dioxide (ETCO2) was continuously measured (SC9000; Dräger Medical, Germany). Volume-control ventilation was provided with a tidal volume of 6–8 ml/kg and a plateau pressure not exceeding 30 cmH2O. The inspiratory oxygen fraction was set to maintain a PaO2 above 70 mmHg. Morphine was used for analgesia. Sedation was provided by a continuous infusion of midazolam, titrated as clinically required to target a Brussels sedation score of 4 [30]. No other agents were used for analgesia or sedation during the study period. Fluid and drug administration remained unchanged throughout the study.
Mean velocity in the middle cerebral artery (VMCA) was measured using a 2-MHz TCD probe (EME, Pioneer, Germany). The ultrasonic probe was placed on a temporal window and fixed using a specially designed holder apparatus to ensure a constant angle of insonation. The right side of the brain was chosen in each patient. TCD adjustments (depth, sample volume, gain, and power) were kept constant during the study.
After inclusion in the study, MAP and VMCA were recorded simultaneously at baseline and after increasing doses of a norepinephrine infusion to increase MAP by approximately 10–15 mmHg and to achieve three or four different levels of MAP. The interval between two measurements was at least 20 min. Each measurement was averaged over a period of two respiratory cycles. All subjects were kept in the 30-degree head up position throughout the procedure. PaCO2 was measured immediately before starting each test and at the end of the procedure to confirm stable levels. Cerebral vascular resistance (CVR) was calculated as CVR = MAP/VMCA. Changes in cerebrovascular resistance (ΔCVR) were estimated from the changes in MCA velocity (ΔVMCA) in response to changes in MAP (ΔMAP). The cerebral autoregulation index (CAI) was calculated as the ratio of the relative changes in CVR and MAP (CAI = ΔMAP%/ΔCVR%); the normal limits are between 0 and 2 [31].
Statistical Analysis
Statistical analyses were performed using a SPSS 13.0 program for Windows NT software package (2004). Descriptive statistics were computed for all study variables. A Kolmogorov–Smirnov test was used, and histograms and normal-quantile plots were examined to verify the normality of distribution of continuous variables. Discrete variables were expressed as counts (percentage) and continuous variables as means ± SD or median with interquartile ranges (IQR: 25th–75th percentiles), depending on the data distribution. Categorical variables were compared by chi-square or Fisher’s exact test, as appropriate. Continuous variables were compared using a Student’s t test. Non-parametric tests were used if the data were not normally distributed. We conducted a receiver operating characteristic (ROC) analysis to determine the best PaCO2 cut-off of, in terms of sensitivity and specificity, for the prediction of impaired cerebral autoregulation. Differences at a level of P < 0.05 were considered statistically significant.
Results
The study included 21 patients with septic shock (2 women, 19 men); their clinical characteristics are summarized in Table 1. Ten patients (47%) had a history of arterial hypertension and 2 (9%) of insulin-requiring diabetes. GCSs at ICU admission, before mechanical ventilation and sedation, ranged from 5 to 15 (median: 13). The duration of septic shock was 2 days for 14 patients and 3 days for 7; 17 patients (80%) had acute renal failure (median creatinine 1.9 [ranges: 1.3–4.1] mg/dl and median urea 84 mg/dl [ranges: 45–125] mg/dl) and 14 (66%) had ALI/ARDS on the study day. Twelve patients (57%) were being treated with hydrocortisone, 12 with dobutamine, and 8 (39%) with activated protein C. Eight patients (38%) eventually died because of complications related to sepsis. The remaining patients were discharged without gross neurologic sequelae.
At study inclusion, MAP was 65 ± 6 mmHg, VMCA 60 ± 20 cm/s, and median PaCO2 35 [28–49] mmHg. Norepinephrine infusion was increased from 7 [2–70] to 20 [8–110] mcg/min to raise MAP from 65 ± 6 to 96 ± 13 mmHg. All other major variables, including CI, remained constant throughout the study (Table 2), except for a significant increase in SVRI with the higher doses of norepinephrine. No relevant changes in PaCO2 were observed between the start and the end of the procedure.
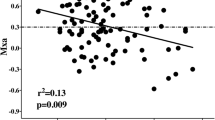
The results of individual cerebral autoregulation testing are shown in Fig. 1. VMCA increased from 60 ± 20 to 78 ± 28 cm/s. The mean increase in VMCA was 0.48 (cm/s)/mmHg MAP [0.09–1.95]. There was no correlation between PaCO2 and the initial VMCA. Fourteen patients (66%) had impaired cerebral autoregulation, including seven of the 14 patients (50%) with a PaCO2 < 40 mmHg [28–37] and all seven of the patients with a PaCO2 > 40 mmHg [41–49] (P = 0.046). Specifically, 4/9 (44%) patients with PaCO2 < 35 mmHg, 7/9 (77%) with PaCO2 between 35 and 42 mmHg, and 3/3 (100%) with PaCO2 > 42 mmHg had impaired autoregulation. The ROC analysis showed, for a PaCO2 threshold of 38 mmHg, a sensitivity of 50% and a specificity of 100% for the prediction of impaired cerebral autoregulation; the area under the ROC curve was 0.76 (95% confidence interval: 0.52–0.91). There were no correlations between baseline VMCA or any measured variables and impaired autoregulation. During the ICU stay, 17 patients underwent cerebral computed tomography (CT)-scan, which was normal in each case, except for one patient who had left frontal ischemia but normal cerebral autoregulation.
Discussion
The major finding of this study is that cerebral autoregulation is often impaired in the early phase of septic shock. Impaired cerebral autoregulation seemed to be more frequent in patients with hypercapnia, suggesting that PaCO2 may alter the responsiveness of cerebral vasculature to arterial pressure stimuli. Steady state conditions were maintained during the study, and we avoided hyperglycemia, extreme acidosis, and uremia, and changes in temperature and hemoglobin concentration, all factors that are known to affect cerebral autoregulation [18]. The drugs used for sedation or analgesia and norepinephrine do not influence cerebral autoregulation [32–34], so that one can assume that modifications in VMCA were due only to changes in MAP.
Reduced cerebral perfusion is a potential cause of or a precipitating factor for cerebral dysfunction in septic patients [35]. Experimental investigations have demonstrated a wide range of CBF values during severe infections [36–39], probably because of the different models used. In human studies, CBF was in the lower range, suggesting a role of cerebral hypoperfusion in the development of SAE [40, 41]. However, in these studies, CBF was estimated from absolute values of VMCA and intracranial vessel velocities do not provide a reliable measure of CBF [42, 43]. In our study, VMCA was quite variable and it is hard to define a normal range in such a group of patients. Direct measurements of CBF with the 133Xe clearance technique [40] or the transcranial double-indicator dilution technique [22] have yielded conflicting results. However, the reduction in CBF during sepsis occurs before alteration of systemic hemodynamics or hypotension [40] and is due to an early increase in CVR [41, 44]. Early after endotoxin administration, a decrease in PaCO2 increased cerebral vascular tone in septic animals [45] and in healthy volunteers [46], suggesting that moderate hyperventilation, and not sepsis per se, reduces CBF.
Changes in CBF in response to changes in PaCO2 are referred to as cerebral CO2 reactivity (COR) [47]. COR was reported to be normal in three clinical studies on sepsis [18, 22, 40], but impaired in two others [48, 49]. However, one of these latter studies [48] investigated septic patients with pre-existing neurological disease, which may already have impaired COR, while in the other [49], three patients had a COR within the normal range, seven had a reduced COR, and two patients had an increased response. Importantly, hyperventilatory hypocapnia probably reduces CBF in septic patients without compromising cerebral metabolism, as indicated by an unchanged cerebral metabolic rate of oxygen (CMRO2) [22]. However, in a canine model of sepsis, only normocapnic animals showed an increase in CMRO2 of over 40% [39], suggesting that, in the presence of normal or high PaCO2 levels, CBF and cerebral metabolism could be uncoupled during sepsis, probably because of loss of CA. Autoregulation of CBF is a sensitive mechanism, which can be impaired by various pathological conditions, and has a direct impact on delayed ischemic events and poor outcome [50, 51]. Although cerebral autoregulation was maintained in several experimental models of sepsis [39, 44, 52, 53], most patients with septic shock had impaired cerebral autoregulation in our study. We found only three human studies that have previously evaluated cerebral autoregulation in septic patients, and these yielded conflicting results. In a recent study by Pfister et al. [17], patients with septic shock had similar CBF to septic patients without shock, but they had disturbed cerebral autoregulation and clinical signs of delirium, as well as higher levels of serum biomarkers of brain injury (s100β); they also had higher mortality rates. In another study [54], septic shock patients had impaired cerebral autoregulation and CBF changes were directly related to changes in cardiac index. However, CBF was calculated by carotid Doppler velocities, and this is only a gross estimate of intracerebral vessel blood flow. In the third study, Matta and Stow [18] reported intact cerebral autoregulation and COR in the early phase of sepsis, concluding that sepsis-induced widespread vasoplegia does not involve the cerebral vasculature. However, individual results were not provided and it is possible that some of these patients had impaired cerebral autoregulation. Moreover, patients with severe sepsis and septic shock were evaluated together, although they may not have the same vascular reactivity [17]. Finally, PaCO2 ranged from 33 to 38 mmHg, and we showed that PaCO2 concentrations above 40 mmHg may significantly influence the autoregulatory cerebral capacity during sepsis.
Even though the precise mechanisms of cerebral autoregulation remain controversial, vascular caliber changes are mediated by a complex interplay of myogenic and metabolic mechanisms [16]. In a study describing the effects of CO2 on cerebral autoregulation [24], the autoregulatory state of the cerebral blood vessels depended on their resting tone. If, as in hypercarbia, the vascular tone was reduced, a further dilation in response to reduced MAP was either impaired or unnecessary. Moreover, elevations in PaCO2 increase autonomic neural activity and this may alter the arterial baroreflex [19]. Mild hypercapnia can significantly impair cerebral autoregulation during general anesthesia [55]. Additionally, PaCO2 levels greater than 55 mmHg have been shown to impair cerebral autoregulation in healthy adults [24]. The presence of hyperemia, usually associated with normocapnia, has been associated with impaired cerebral autoregulation in brain-injured patients [56]. In contrast, cerebral autoregulation may be regained by hyperventilatory hypocapnia in many disease states, such as acute liver failure [57].
Interestingly, the vasodilatory effects of hypercapnia are in part mediated by an increased production of nitric oxide (NO) from the vascular endothelium—a phenomenon called chemoregulation. During sepsis, activation of the inducible form of NO synthase (iNOS) by endotoxins can play an important role in mediating a reduction in vascular resistance [58], with excessive vasodilatation and loss of autoregulatory reserve [59]. In the presence of hypercapnia these effects could be further enhanced, and participate to the loss of autoregulation even in the cerebral circulation. Data about endothelial dysfunction in the brain of septic patients are scarce and experimental studies in sepsis showed that NOS inhibition reversed hypotension and the hyperdynamic state [60] but had no effects on the brain vasculature [52]. Our study showed that, during septic shock, normal or high levels of PaCO2 were associated with loss of cerebral autoregulation in all patients, whereas only 50% of patients with low PaCO2 levels had impaired cerebral autoregulation. In septic ICU patients, hypercapnia is a common clinical event in the presence of chronic obstructive pulmonary disease, status asthmaticus, obesity hypoventilation syndrome, major pulmonary resection, neurological impairment of respiratory muscles, such as in amyotrophic lateral sclerosis, and in severe ALI/ARDS [61–63]. Nevertheless, no data are available about the brain autoregulatory capacity in these patients and an increased frequency of encephalopathy or ischemic brain lesions has never been associated with higher PaCO2 levels.
There are several limitations to this study. First, the number of patients was relatively small and these results should be confirmed in a larger group. Second, other techniques, such as Xe133 methods [40] or transcranial double-indicator dilution technique [22] may more reliably estimate CBF than TCD although they are more difficult to use. However, TCD has been previously validated as a non-invasive, inexpensive, and non-radioactive method for the assessment of cerebral autoregulation [24]. When TCD is used, one assumes the diameter of the insonated vessel is constant to interpret relative changes in VMCA as relative changes in CBF. There is some evidence that the MCA is a conductance vessel and its diameter does not significantly change with MAP [64, 65], while changes in CVR occur primarily though dilatation of arterioles and pial arteries, which are the main determinants of cerebral autoregulation [66, 67]. Third, for the sake of simplicity, we did not study different PaCO2 concentrations in each patient individually, which limits our ability to ascribe all the changes in cerebral autoregulation to CO2 alone. However, modifications in ventilator conditions would have been problematic in the 14 patients with ALI/ARDS. In addition, brain tissue or jugular bulb venous PCO2 monitoring may be preferable to PaCO2 for the interpretation of the effects of CO2 on CBF and cerebral autoregulation [68]. Fourth, we could not evaluate the potential relationship between impaired cerebral autoregulation and the occurrence of SAE, but this association has already been reported elsewhere [17]. Moreover, as in previous studies [17, 18, 22], we lack data on cerebral autoregulation in septic patients with low GCS scores. Fifth, we examined cerebral autoregulation only within defined limits of MAP and some patients may have a right-shift of their autoregulation curve with a higher threshold limit for cerebral autoregulation. However, we investigated MAP levels that are clinically acceptable in the management of septic shock [69]. An assessment of cerebral autoregulation in the patients recovering from septic shock was not feasible because of lack of cooperation and inability to maintain pre-defined concentrations of PaCO2. Finally, we explored blood flow in the large intracranial basal vessels and cannot extrapolate our results to regional CBF in the cortex or deep brain structures, such as the basal ganglia or hypothalamus.
Conclusions
The present study indicates that cerebral autoregulation is impaired in most patients with septic shock. The clinical implications of this impaired cerebral autoregulation are that, in such patients, even modest hypotension can lead to cerebral hypoperfusion. This stresses the importance of achieving hemodynamic stabilization to avoid a critical decrease in blood pressure in these patients. Our results suggest that PaCO2 concentrations above 40 mmHg are more likely to alter cerebral autoregulation. Further investigations are necessary to define the role of cerebral hemodynamics and the potential role of PaCO2 in the pathogenesis of SAE.
References
Vincent JL, Sakr Y, Sprung CL, et al. Sepsis in European intensive care units: results of the SOAP study. Crit Care Med. 2006;34:344–53.
Young GB, Bolton CF, Austin TW, Archibald YM, Gonder J, Wells GA. The encephalopathy associated with septic illness. Clin Invest Med. 1990;13:297–304.
Eggers V, Schilling A, Kox WJ, Spies C. Septic encephalopathy. Diagnosis und therapy. Anaesthesist. 2003;52:294–303.
Zauner C, Gendo A, Kramer L, et al. Impaired subcortical and cortical sensory evoked potential pathways in septic patients. Crit Care Med. 2002;30:1136–9.
Young GB, Bolton CF, Archibald YM, Austin TW, Wells GA. The electroencephalogram in sepsis-associated encephalopathy. J Clin Neurophysiol. 1992;9:145–52.
Wratten ML. Therapeutic approaches to reduce systemic inflammation in septic-associated neurologic complications. Eur J Anaesthesiol Suppl. 2008;42:1–7.
Barichello T, Martins MR, Reinke A, et al. Cognitive impairment in sepsis survivors from cecal ligation and perforation. Crit Care Med. 2005;33:221–3.
Ebersoldt M, Sharshar T, Annane D. Sepsis-associated delirium. Intensive Care Med. 2007;33:941–50.
Goris RJ. Mediators of multiple organ failure. Intensive Care Med. 1990;16(Suppl 3):S192–6.
Davies DC. Blood–brain barrier breakdown in septic encephalopathy and brain tumours. J Anat. 2002;200:639–46.
Papadopoulos MC, Lamb FJ, Moss RF, Davies DC, Tighe D, Bennett ED. Faecal peritonitis causes oedema and neuronal injury in pig cerebral cortex. Clin Sci (Lond). 1999;96:461–6.
Freund HR, Muggia-Sullam M, Peiser J, Melamed E. Brain neurotransmitter profile is deranged during sepsis and septic encephalopathy in the rat. J Surg Res. 1985;38:267–71.
Sharshar T, Gray F, Lorin DLG, et al. Apoptosis of neurons in cardiovascular autonomic centres triggered by inducible nitric oxide synthase after death from septic shock. Lancet. 2003;362:1799–805.
Wijdicks EF, Stevens M. The role of hypotension in septic encephalopathy following surgical procedures. Arch Neurol. 1992;49:653–6.
Sharshar T, Annane D, de la Grandmaison GL, Brouland JP, Hopkinson NS, Francoise G. The neuropathology of septic shock. Brain Pathol. 2004;14:21–33.
Strandgaard S, Paulson OB. Cerebral autoregulation. Stroke. 1984;15:413–6.
Pfister D, Siegemund M, Dell-Kuster S, et al. Cerebral perfusion in sepsis-associated delirium. Crit Care. 2008;12:R63.
Matta BF, Stow PJ. Sepsis-induced vasoparalysis does not involve the cerebral vasculature: indirect evidence from autoregulation and carbon dioxide reactivity studies. Br J Anaesth. 1996;76:790–4.
Ainslie PN, Celi L, McGrattan K, Peebles K, Ogoh S. Dynamic cerebral autoregulation and baroreflex sensitivity during modest and severe step changes in arterial PCO2. Brain Res. 2008;1230:115–24.
Xie A, Skatrud JB, Morgan B, et al. Influence of cerebrovascular function on the hypercapnic ventilatory response in healthy humans. J Physiol. 2006;577:319–29.
Harper AM, Glass HI. Effect of alterations in the arterial carbon dioxide tension on the blood flow through the cerebral cortex at normal and low arterial blood pressures. J Neurol Neurosurg Psychiatry. 1965;28:449–52.
Thees C, Kaiser M, Scholz M, et al. Cerebral haemodynamics, carbon dioxide reactivity during sepsis syndrome. Crit Care. 2007;11:R123.
van Lieshout JJ, Wieling W, Karemaker JM, Secher NH. Syncope, cerebral perfusion, and oxygenation. J Appl Physiol. 2003;94:833–48.
Aaslid R, Lindegaard KF, Sorteberg W, Nornes H. Cerebral autoregulation dynamics in humans. Stroke. 1989;20:45–52.
Levy MM, Fink MP, Marshall JC, et al. 2001 SCCM/ESICM/ACCP/ATS/SIS International Sepsis Definitions Conference. Crit Care Med. 2003;31:1250–6.
Teasdale G, Jennett B. Assessment of coma and impaired consciousness. A practical scale. Lancet. 1974;2:81–4.
Bernard GR, Artigas A, Brigham KL, et al. The American-European Consensus Conference on ARDS. Definitions, mechanisms, relevant outcomes, and clinical trial coordination. Am J Respir Crit Care Med. 1994;149:818–24.
Vincent JL, Moreno R, Takala J, et al. The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the Working Group on Sepsis-Related Problems of the European Society of Intensive Care Medicine. Intensive Care Med. 1996;22:707–10.
Knaus WA, Draper EA, Wagner DP, Zimmerman JE. APACHE II: a severity of disease classification system. Crit Care Med. 1985;13:818–29.
Detriche O, Berre J, Massaut J, Vincent JL. The Brussels sedation scale: use of a simple clinical sedation scale can avoid excessive sedation in patients undergoing mechanical ventilation in the intensive care unit. Br J Anaesth. 1999;83:698–701.
Bouma GJ, Muizelaar JP. Cerebral blood flow, cerebral blood volume, and cerebrovascular reactivity after severe head injury. J Neurotrauma. 1992;9(Suppl 1):S333–48.
Forster A, Juge O, Morel D. Effects of midazolam on cerebral hemodynamics and cerebral vasomotor responsiveness to carbon dioxide. J Cereb Blood Flow Metab. 1983;3:246–9.
de Nadal M, Munar F, Poca MA, Sahuquillo J, Garnacho A, Rossello J. Cerebral hemodynamic effects of morphine and fentanyl in patients with severe head injury: absence of correlation to cerebral autoregulation. Anesthesiology. 2000;92:11–9.
Moppett IK, Sherman RW, Wild MJ, Latter JA, Mahajan RP. Effects of norepinephrine and glyceryl trinitrate on cerebral haemodynamics: transcranial Doppler study in healthy volunteers. Br J Anaesth. 2008;100:240–4.
Eidelman LA, Putterman D, Putterman C, Sprung CL. The spectrum of septic encephalopathy. Definitions, etiologies, and mortalities. JAMA. 1996;275:470–3.
Martin CM, Sibbald WJ. Modulation of hemodynamics and organ blood flow by nitric oxide synthase inhibition is not altered in normotensive, septic rats. Am J Respir Crit Care Med. 1994;150:1539–44.
Raper RF, Sibbald WJ, Hobson J, Rutledge FS. Effect of PGE1 on altered distribution of regional blood flows in hyperdynamic sepsis. Chest. 1991;100:1703–11.
Meyer J, Hinder F, Stothert J Jr, et al. Increased organ blood flow in chronic endotoxemia is reversed by nitric oxide synthase inhibition. J Appl Physiol. 1994;76:2785–93.
Ekstrom-Jodal B, Haggendal E, Larsson LE. Cerebral blood flow and oxygen uptake in endotoxic shock. An experimental study in dogs. Acta Anaesthesiol Scand. 1982;26:163–70.
Bowton DL, Bertels NH, Prough DS, Stump DA. Cerebral blood flow is reduced in patients with sepsis syndrome. Crit Care Med. 1989;17:399–403.
Maekawa T, Fujii Y, Sadamitsu D, et al. Cerebral circulation and metabolism in patients with septic encephalopathy. Am J Emerg Med. 1991;9:139–43.
Bishop CC, Powell S, Rutt D, Browse NL. Transcranial Doppler measurement of middle cerebral artery blood flow velocity: a validation study. Stroke. 1986;17:913–5.
Poulin MJ, Robbins PA. Indexes of flow and cross-sectional area of the middle cerebral artery using Doppler ultrasound during hypoxia and hypercapnia in humans. Stroke. 1996;27:2244–50.
Parker JL, Emerson TE Jr. Cerebral hemodynamics, vascular reactivity, and metabolism during canine endotoxin shock. Circ Shock. 1977;4:41–53.
Hinkelbein J, Schroeck H, Peterka A, Schubert C, Kuschinsky W, Kalenka A. Local cerebral blood flow is preserved in sepsis. Curr Neurovasc Res. 2007;4:39–47.
Moller K, Strauss GI, Qvist J, et al. Cerebral blood flow and oxidative metabolism during human endotoxemia. J Cereb Blood Flow Metab. 2002;22:1262–70.
Ackerman RH, Zilkha E, Bull JW, et al. The relationship of the CO2 reactivity of cerebral vessels to blood pressure and mean resting blood flow. Neurology. 1973;23:21–6.
Terborg C, Schummer W, Albrecht M, Reinhart K, Weiller C, Rother J. Dysfunction of vasomotor reactivity in severe sepsis and septic shock. Intensive Care Med. 2001;27:1231–4.
Bowie RA, O’Connor PJ, Mahajan RP. Cerebrovascular reactivity to carbon dioxide in sepsis syndrome. Anaesthesia. 2003;58:261–5.
Pickard JD, Matheson M, Patterson J, Wyper D. Prediction of late ischemic complications after cerebral aneurysm surgery by the intraoperative measurement of cerebral blood flow. J Neurosurg. 1980;53:305–8.
Czosnyka M, Smielewski P, Kirkpatrick P, Menon DK, Pickard JD. Monitoring of cerebral autoregulation in head-injured patients. Stroke. 1996;27:1829–34.
Booke M, Westphal M, Hinder F, Traber LD, Traber DL. Cerebral blood flow is not altered in sheep with Pseudomonas aeruginosa sepsis treated with norepinephrine or nitric oxide synthase inhibition. Anesth Analg. 2003;96:1122–8, table.
Pedersen M, Brandt CT, Knudsen GM, et al. The effect of S. pneumoniae bacteremia on cerebral blood flow autoregulation in rats. J Cereb Blood Flow Metab. 2008;28:126–34.
Smith SM, Padayachee S, Modaresi KB, Smithies MN, Bihari DJ. Cerebral blood flow is proportional to cardiac index in patients with septic shock. J Crit Care. 1998;13:104–9.
McCulloch TJ, Visco E, Lam AM. Graded hypercapnia and cerebral autoregulation during sevoflurane or propofol anesthesia. Anesthesiology. 2000;93:1205–9.
Vavilala MS, Lee LA, Boddu K, et al. Cerebral autoregulation in pediatric traumatic brain injury. Pediatr Crit Care Med. 2004;5:257–63.
Strauss GI. The effect of hyperventilation upon cerebral blood flow and metabolism in patients with fulminant hepatic failure. Dan Med Bull. 2007;54:99–111.
Szabo C. Alterations in nitric oxide production in various forms of circulatory shock. New Horiz. 1995;3:2–32.
Avontuur JA, Bruining HA, Ince C. Nitric oxide causes dysfunction of coronary autoregulation in endotoxemic rats. Cardiovasc Res. 1997;35:368–76.
Avontuur JA, Tutein Nolthenius RP, van Bodegom JW, Bruining HA. Prolonged inhibition of nitric oxide synthesis in severe septic shock: a clinical study. Crit Care Med. 1998;26:660–7.
Lee WY, Mokhlesi B. Diagnosis and management of obesity hypoventilation syndrome in the ICU. Crit Care Clin. 2008;24:533–49. vii.
Pieretti P, Alifano M, Roche N, et al. Predictors of an appropriate admission to an ICU after a major pulmonary resection. Respiration. 2006;73:157–65.
Chakrabarti B, Angus RM. Ventilatory failure on acute take. Clin Med. 2005;5:630–4.
Matta BF, Mayberg TS, Lam AM. Direct cerebrovasodilatory effects of halothane, isoflurane, and desflurane during propofol-induced isoelectric electroencephalogram in humans. Anesthesiology. 1995;83:980–5.
Matta BF, Lam AM, Strebel S, Mayberg TS. Cerebral pressure autoregulation and carbon dioxide reactivity during propofol-induced EEG suppression. Br J Anaesth. 1995;74:159–63.
Kontos HA, Wei EP, Navari RM, Levasseur JE, Rosenblum WI, Patterson JL Jr. Responses of cerebral arteries and arterioles to acute hypotension and hypertension. Am J Physiol. 1978;234:H371–83.
Baumbach GL, Heistad DD. Regional, segmental, and temporal heterogeneity of cerebral vascular autoregulation. Ann Biomed Eng. 1985;13:303–10.
Peebles K, Celi L, McGrattan K, Murrell C, Thomas K, Ainslie PN. Human cerebrovascular and ventilatory CO2 reactivity to end-tidal, arterial and internal jugular vein PCO2. J Physiol. 2007;584:347–57.
Dellinger RP, Levy MM, Carlet JM, et al. Surviving Sepsis Campaign: international guidelines for management of severe sepsis and septic shock: 2008. Crit Care Med. 2008;36:296–327.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Taccone, F.S., Castanares-Zapatero, D., Peres-Bota, D. et al. Cerebral Autoregulation is Influenced by Carbon Dioxide Levels in Patients with Septic Shock. Neurocrit Care 12, 35–42 (2010). https://doi.org/10.1007/s12028-009-9289-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-009-9289-6