Abstract
Objective
To correlate regional brain saturations (RSO2) measured by cerebral Near-infrared spectroscopy (cNIRS) with serological markers indicative of neurological injury (neuron-specific enolase (NSE) and S100β).
Methods
Children with at least one organ failure who were undergoing cNIRS monitoring were eligible for enrollment, while children with hyperbilirubinemia and cyanotic heart disease were excluded. Children were further analyzed based on the presence of an acute neurological injury (defined as hypoxic/ischemic injury after cardiac arrest, status epilepticus, meningitis, encephalopathy) as well as survival. RSO2 was measured continuously (every 30 s) and averages were obtained at 6 h and 24 h epochs prior to serum collection (E6 and E24, respectively). Serum was collected for NSE and S100β, which were both determined by ELISA. Serum from children undergoing evaluation for fever in the Emergency department served as serological controls. Correlations were determined using the Pearson Product Moment Correlations.
Results
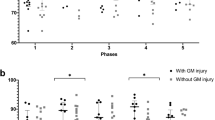
A total of 26 children underwent cNIRS monitoring for a total of 47 days. Overall NSE was greater in critically ill children compared to controls, as well as in all subsets of children analyzed (acute CNS injuries, no acute CNS injuries, survivors and non-survivors). S100β tended to be greater in critically ill children, but this did not reach statistical significance. Average RSO2 in E6 and E24 was 68.0% ± 1.5 and 68.6% ± 1.6, respectively, in a total of 131,036 measurements and E6 RSO2 was strongly, negatively correlated with S100β in children with acute neurological injuries.
Conclusions
This is the first study to correlate averaged RSO2 measured by cNIRS with neurological injury markers in critically ill children. We believe that this data can be used to establish thresholds for RSO2 that can be tested in future trials to determine if this technology is predictive of long-term neurological outcome.

Similar content being viewed by others
References
Bone RC, Sprung CL, Sibbald WJ. Definitions for sepsis and organ failure. Crit Care Med. 1992;20:724–6.
Osterlundh G, Kjellmer I, Lannering B, et al. Neurochemical markers of brain damage in cerebrospinal fluid during induction treatment of acute lymphoblastic leukemia in children. Pediatr Blood Cancer. 2008;50:793–8.
Berger RP, Beers SR, Richichi R, et al. Serum biomarker concentrations and outcome after pediatric traumatic brain injury. J Neurotrauma. 2007;24:1793–801.
Nguyen DN, Spapen H, Su F, et al. Elevated serum levels of S-100beta protein and neuron-specific enolase are associated with brain injury in patients with severe sepsis and septic shock. Crit Care Med. 2006;34:1967–74.
Thorngren-Jerneck K, Alling C, Herbst A, et al. S100 protein in serum as a prognostic marker for cerebral injury in term newborn infants with hypoxic ischemic encephalopathy. Pediatr Res. 2004;55:406–12.
Tiainen M, Roine RO, Pettila V, Takkunen O. Serum neuron-specific enolase and S-100B protein in cardiac arrest patients treated with hypothermia. Stroke. 2003;34:2881–6.
Abdul-Khaliq H, Schubert S, Stoltenburg-Didinger G, et al. Release patterns of astrocytic and neuronal biochemical markers in serum during and after experimental settings of cardiac surgery. Restor Neurol Neurosci. 2003;21:141–50.
Tanabe T, Suzuki S, Hara K, et al. Cerebrospinal fluid and serum neuron-specific enolase levels after febrile seizures. Epilepsia. 2001;42:504–7.
Pleines UE, Morganti-Kossmann MC, Rancan M, et al. S-100 beta reflects the extent of injury and outcome, whereas neuronal specific enolase is a better indicator of neuroinflammation in patients with severe traumatic brain injury. J Neurotrauma. 2001;18:491–8.
Piazza O, Storti MP, Cotena S, et al. S100B is not a reliable prognostic index in paediatric TBI. Pediatr Neurosurg. 2007;43:258–64.
Anderson RE, Hansson LO, Nilsson O, et al. Increase in serum S100A1-B and S100BB during cardiac surgery arises from extracerebral sources. Ann Thorac Surg. 2001;71:1512–7.
Soul JS, du Plessis AJ. New technologies in pediatric neurology. Near-infrared spectroscopy. Semin Pediatr Neurol. 1999;6:101–10.
Zeltzer PM, Marangos PJ, Parma AM, et al. Raised neuron-specific enolase in serum of children with metastatic neuroblastoma. A report from the Children’s Cancer Study Group. Lancet. 1983;2:361–3.
Hsu AA, Fenton K, Weinstein S, et al. Neurological injury markers in children with septic shock. Pediatr Crit Care. 2008;9:245–51.
American College of Chest Physicians/Society of Critical Care Medicine Consensus Conference: definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. Crit Care Med. 1992;20:864–74.
Pollack MM, Patel KM, Ruttimann UE. PRISM III: an updated pediatric risk of mortality score. Crit Care Med. 1996;24:743–52.
Trachsel D, McCrindle BW, Nakagawa S, Bohn D. Oxygenation index predicts outcome in children with acute hypoxemic respiratory failure. Am J Respir Crit Care Med. 2005;172:206–11.
Rivera RA, Butt W, Shann F. Predictors of mortality in children with respiratory failure: possible indications for ECMO. Anaesth Intensive Care. 1990;18:385–9.
Leteurtre S, Martinot A, Duhamel A, et al. Validation of the paediatric logistic organ dysfunction (PELOD) score: prospective, observational, multicentre study. Lancet. 2003;362:192–7.
Shore PM, Berger RP, Varma S, et al. Cerebrospinal fluid biomarkers versus glasgow coma scale and glasgow outcome scale in pediatric traumatic brain injury: the role of young age and inflicted injury. J Neurotrauma. 2007;24:75–86.
Bandyopadhyay S, Hennes H, Gorelick MH, et al. Serum neuron-specific enolase as a predictor of short-term outcome in children with closed traumatic brain injury. Acad Emerg Med. 2005;12:732–8.
Petzold A, Keir G, Lim D, et al. Cerebrospinal fluid (CSF) and serum S100B: release and wash-out pattern. Brain Res Bull. 2003;61:281–5.
Pelinka LE, Bahrami S, Szalay L, et al. Hemorrhagic shock induces an S 100 B increase associated with shock severity. Shock. 2003;19:422–6.
Dimopoulou I, Korfias S, Dafni U, et al. Protein S-100b serum levels in trauma-induced brain death. Neurology. 2003;60:947–51.
Berger RP, Pierce MC, Wisniewski SR, et al. Neuron-specific enolase and S100B in cerebrospinal fluid after severe traumatic brain injury in infants and children. Pediatrics. 2002;109:E31.
Berger RP, Pierce MC, Wisniewski SR, et al. Serum S100B concentrations are increased after closed head injury in children: a preliminary study. J Neurotrauma. 2002;19:1405–9.
Gazzolo D, Marinoni E, Di Iorio R, et al. Measurement of urinary S100B protein concentrations for the early identification of brain damage in asphyxiated full-term infants. Arch Pediatr Adolesc Med. 2003;157:1163–8.
Berger RP, Adelson PD, Richichi R, Kochanek PM. Serum biomarkers after traumatic and hypoxemic brain injuries: insight into the biochemical response of the pediatric brain to inflicted brain injury. Dev Neurosci. 2006;28:327–35.
Schmitt B, Bauersfeld U, Schmid ER, et al. Serum and CSF levels of neuron-specific enolase (NSE) in cardiac surgery with cardiopulmonary bypass: a marker of brain injury? Brain Dev. 1998;20:536–9.
Kirshbom PM, Forbess JM, Kogon BE, et al. Cerebral near infrared spectroscopy is a reliable marker of systemic perfusion in awake single ventricle children. Pediatr Cardiol. 2007;28:42–5.
Gottlieb EA, Fraser CD Jr., Andropoulos DB, Diaz LK. Bilateral monitoring of cerebral oxygen saturation results in recognition of aortic cannula malposition during pediatric congenital heart surgery. Paediatr Anaesth. 2006;16:787–9.
Hayashida M, Kin N, Tomioka T, et al. Cerebral ischaemia during cardiac surgery in children detected by combined monitoring of BIS and near-infrared spectroscopy. Br J Anaesth. 2004;92:662–9.
Berens RJ, Stuth EA, Robertson FA, et al. Near infrared spectroscopy monitoring during pediatric aortic coarctation repair. Paediatr Anaesth. 2006;16:777–81.
Huang L, Ding H, Hou X, et al. Assessment of the hypoxic-ischemic encephalopathy in neonates using non-invasive near-infrared spectroscopy. Physiol Meas. 2004;25:749–61.
Kurth CD, Steven JL, Montenegro LM, et al. Cerebral oxygen saturation before congenital heart surgery. Ann Thorac Surg. 2001;72:187–92.
Petrova A, Mehta R. Near-infrared spectroscopy in the detection of regional tissue oxygenation during hypoxic events in preterm infants undergoing critical care. Pediatr Crit Care Med. 2006;7:449–54.
Bhutta AT, Ford JW, Parker JG, et al. Noninvasive cerebral oximeter as a surrogate for mixed venous saturation in children. Pediatr Cardiol. 2007;28:34–41.
Tortoriello TA, Stayer SA, Mott AR, et al. A noninvasive estimation of mixed venous oxygen saturation using near-infrared spectroscopy by cerebral oximetry in pediatric cardiac surgery patients. Paediatr Anaesth. 2005;15:495–503.
Yoxall CW, Weindling AM, Dawani NH, Peart I. Measurement of cerebral venous oxyhemoglobin saturation in children by near-infrared spectroscopy and partial jugular venous occlusion. Pediatr Res. 1995;38:319–23.
Gopinath SP, Cormio M, Ziegler J, et al. Intraoperative jugular desaturation during surgery for traumatic intracranial hematomas. Anesth Analg. 1996;83:1014–21.
Gopinath SP, Robertson CS, Contant CF, et al. Jugular venous desaturation and outcome after head injury. J Neurol Neurosurg Psychiatr. 1994;57:717–23.
Acknowledgment
Dr. Bell is funded by NIH (HD 044716).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Subbaswamy, A., Hsu, A.A., Weinstein, S. et al. Correlation of Cerebral Near-Infrared Spectroscopy (cNIRS) and Neurological Markers in Critically Ill Children. Neurocrit Care 10, 129–135 (2009). https://doi.org/10.1007/s12028-008-9122-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-008-9122-7