Abstract
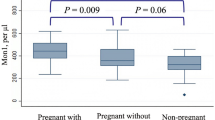
Preeclampsia (PE) is a pregnancy-specific syndrome characterized by a systemic inflammatory response that polarizes peripheral blood monocytes to the M1 phenotype. The classically activated M1 monocytes comprise immune effector cells with an acute inflammatory phenotype. CD163 is a scavenger receptor expressed by monocytes/macrophages that may be shed from their cell membrane after proteolytic cleavage, producing the soluble CD163 molecule (sCD163). This study evaluated CD163 expression by monocytes and sCD163 as well as pro- and anti-inflammatory cytokine concentration in the plasma of pregnant women with PE. Fifty-six women with PE and 28 normotensive pregnant women were included. Plasma levels of sCD163, interleukin-1 beta (IL-1β), IL-6, IL-10, transforming growth factor beta (TGF-β1), and tumor necrosis factor-alpha (TNF-α) were determined by ELISA, and CD163 expression by monocytes was assessed by flow cytometry. The expression of CD163 by monocytes was significantly lower in severe and mild PE than in normotensive pregnant. Plasma concentrations of IL-1β, TGF-β1, and TNF-α were higher in severe PE than in mild PE and normotensive pregnant women. Both groups of preeclamptic women showed decreased plasma levels of sCD163 and IL-10. Negative correlations between sCD163 and IL-1β (r = − 0.45; P = 0.014) and between sCD163 and TNF-α concentrations (r = − 0.54; P = 0.001) were observed in the severe PE group. The association between the pro-inflammatory cytokine profile and lower concentrations of sCD163 and IL-10 in plasma from women with severe PE suggests an impairment in the modulation of the systemic inflammatory response in this group of pregnant women with preeclampsia.


Similar content being viewed by others
References
Mihu D, Razvan C, Malutan A, Mihaela C. Evaluation of maternal systemic inflammatory response in preeclampsia. Taiwan J Obstet Gynecol. 2015;54(2):160–6.
Redman CW, Sargent IL. Latest advances in understanding preeclampsia. Science. 2005;308(5728):1592–4.
Borzychowski AM, Sargent IL, Redman CW. Inflammation and preeclampsia. Semin Fetal Neonatal Med. 2006;11(5):309–16.
Lok CA, Jebbink J, Nieuwland R, Faas MM, Boer K, Sturk AJ, et al. Leukocyte activation and circulating leukocyte-derived microparticles in preeclampsia. Am J Reprod Immunol. 2009;61(5):346–9.
Gotsch F, Romero R, Friel L, Kusanovic JP, Espinoza J, Erez O, et al. CXCL10/IP-10: a missing link between inflammation and anti-angiogenesis in preeclampsia? J Matern Fetal Neonatal Med. 2007;20(11):777–92.
De Oliveira L, Câmara NO, Peraçoli JC, Peraçoli MT, Moron AF, Sass N. sFlt-1 and IP-10 in women with early-onset preeclampsia. Pregnancy Hypertens. 2011;1(2):129–31.
Giorgi VS, Peracoli MT, Peracoli JC, Witkin SS, Bannwart-Castro CF. Silibinin modulates the NF-κB pathway and pro-inflammatory cytokine production by mononuclear cells from preeclamptic women. J Reprod Immunol. 2012;95(1–2):67–72.
Raghupathy R. Cytokines as key players in the pathophysiology of preeclampsia. Med Princ Pract. 2013;22(suppl 1):8–19.
Cristofalo R, Bannwart-Castro CF, Magalhães CG, Borges VT, Peraçoli JC, Witkin SS, et al. Silibinin attenuates oxidative metabolism and cytokine production by monocytes from preeclamptic women. Free Radic Res. 2013;47(4):268–75.
Peraçoli JC, Bannwart-Castro CF, Romao M, Weel IC, Ribeiro VR, Borges VT, et al. High levels of heat shock protein 70 are associated with pro-inflammatory cytokines and may differentiate early- from late-onset preeclampsia. J Reprod Immunol. 2013;100(2):129–34.
Peraçoli MT, Bannwart CF, Cristofalo R, Borges VT, Costa RA, Witkin SS, et al. Increased reactive oxygen species and tumor necrosis factor-alpha production by monocytes are associated with elevated levels of uric acid in pre-eclamptic women. Am J Reprod Immunol. 2011;66(6):460–7.
Matias ML, Romão M, Weel IC, Ribeiro VR, Nunes PR, Borges VT, et al. Endogenous and uric acid-induced activation of NLRP3 inflammasome in pregnant women with preeclampsia. PLoSOne. 2015;10(6):e0129095.
Mantovani A, Sica A, Sozzani S, Allavena P, Vecchi A, Locati M. The chemokine system in diverse forms of macrophage activation and polarization. Trends Immunol. 2004;25(12):677–86.
Gordon S, Martinez FO. Alternative activation of macrophages: mechanism and functions. Immunity. 2010;32(5):593–604.
Zhou D, Huang C, Lin Z, Zhan S, Kong L, Fang C, et al. Macrophage polarization and function with emphasis on the evolving roles of coordinated regulation of cellular signaling pathways. Cell Signal. 2014;26(2):192–7.
Sica A, Larghi P, Mancino A, Rubino L, Porta C, Totaro MG, et al. Macrophage polarization in tumor progression. Semin Cancer Biol. 2008;18(5):349–55.
Van Gorp H, Delputte PL, Nauwynck HJ. Scavenger receptor CD163, a Jack-of-all-trades and potential target for cell-directed therapy. Molecular Immunol. 2010;47(7–8):1650–60.
Porcheray F, Viaud S, Rimaniol AC, Léone C, Samah B, Dereuddre-Bosquet N, et al. Macrophage activation switching: an asset for the resolution of inflammation. Clin Exp Immunol. 2005;142(3):481–9.
Bouhlel MA, Derudas B, Rigamonti E, Dievat R, Brozek J, Haulon S, et al. PPAR gamma activation primes human monocytes into alternative M2 macrophages with anti-inflammatory properties. Cell Metab. 2007;6(2):137–43.
Groselj-Grenc M, Ihan A, Derganc M. Neutrophil and monocyte CD64 and CD163 expression in critically ill neonates and children with sepsis: comparison of fluorescence intensities and calculated indexes. Mediat Inflamm. 2008;2008:202646. https://doi.org/10.1155/2008/202646.
Satoh N, Shimatsu A, Himeno A, Sasaki Y, Yamakage H, Yamada K, et al. Unbalanced M1/M2 phenotype of peripheral blood monocytes in obese diabetic patients: effect of pioglitazone. Diabetes Care. 2010;33(1):e7. https://doi.org/10.2337/dc09-1315.
Fadini GP, Simoni F, Cappellari R, Vitturi N, Galasso S, Vigili de Kreutzenberg S, et al. Pro-inflammatory monocyte-macrophage polarization imbalance in human hypercholesterolemia and atherosclerosis. Atherosclerosis. 2014;237(2):805–8.
Awad F, Assrawi E, Jumeau C, Georgin-Lavialle S, Cobret L, Duquesnay P, et al. Impact of human monocyte and macrophage polarization on NLR expression and NLRP3 inflammasome activation. PLoS One. 2017;12(4):e0175336.
Medeiros LT, Peraçoli JC, Bannwart-Castro CF, Romão M, Weel IC, Golim MA, et al. Monocytes from pregnant women with preeclampsia are polarized to a M1 phenotype. Am J Reprod Immunol. 2014;72(1):5–13.
Møller HJ, Peterslund NA, Graversen JH, Moestrup SK. Identification of the hemoglobin scavenger receptor/CD163 as a natural soluble protein in plasma. Blood. 2002;99(1):378–80.
Buechler C, Eisinger K, Krautbauer S. Diagnostic and prognostic potential of the macrophage specific receptor CD163 in inflammatory diseases. Inflamm Allergy Drug Targets. 2013;12(6):391–402.
Tranquilli AL, Dekker G, Magee L, Roberts J, Sibai BM, Steyn W, et al. The classification, diagnosis and management of the hypertensive disorders of pregnancy: a revised statement from the ISSHP. Pregnancy Hypertens. 2014;4(2):97–104.
Mol BW, Roberts CT, Thangaratinam S, Magee LA, Groot CG, Hofmeyr GJ. Pre-eclampsia. Lancet. 2016;387(10022):999–1011, Pre-eclampsia.
American College of Obstetricians and Gynecologists. Task force on hypertension in pregnancy hypertension in pregnancy: report of the American College of Obstetricians and Gynecologists’ task force on hypertension in pregnancy. Obstet Gynecol. 2013;122(5):1122–31.
Peracoli MT, Bannwart CF, Cristofalo R, Borges VT, Costa RA, Witkin SS, et al. Increased reactive oxygen species and tumor necrosis factor-a production by monocytes are associated with elevated levels of uric acid in pre-eclamptic women. Am J Reprod Immunol. 2011;66:460–7.
Striz I, Brabcova E, Kolesar L, Liu XD, Brabcova I, Sekerkova A, et al. Epithelial cells modulate genes associated with NF-kappaB activation in co-cultured human macrophages. Immunobiology. 2011;216(10):1110–6.
Vallabhapurapu S, 15 KM. Regulation and function of NF-kappaB transcription factors in the immune system. Annu Rev Immunol. 2009;27:693–733.
Pinheiro MB, Martins-Filho OA, Mota AP, Alpoim PN, Godoi LC, Silveira AC, et al. Severe preeclampsia goes along with a cytokine network disturbance towards a systemic inflammatory state. Cytokine. 2013;62(1):165–73.
Djurovic S, Schjetlein R, Wisløff F, Haugen G, Husby H, Berg K. Plasma concentrations of Lp(a) lipoprotein and TGF-beta1 are altered in preeclampsia. Clin Genet. 1997;52(5):371–6.
Peraçoli MT, Menegon FT, Borges VT, de Araújo Costa RA, Thomazini-Santos IA, Peraçoli JC. Platelet aggregation and TGF-beta1 plasma levels in pregnant women with preeclampsia. J Reprod Immunol. 2008;79(1):79–84.
Lev PR, Salim JP, Marta RF, Osorio MJ, Goette NP, Molinas FC. Platelets possess functional TGF-beta receptors and Smad2 protein. Platelets. 2007;18(1):35–42.
Travis MA, Sheppard D. TGF-β activation and function in immunity. Annu Rev Immunol. 2014;32:51–82.
Zhou L, Lopes JE, Chong MM, Ivanov II, Min R, Victora GD, et al. TGF-β-induced Foxp3 inhibits TH17 cell differentiation by antagonizing RORγt function. Nature. 2008;453(7192):236–40.
Ribeiro VR, Romao-Veiga M, Romagnoli GG, Matias ML, Nunes PR, Borges VTM, et al. Association between cytokine profile and transcription factors produced by T-cell subsets in early- and late-onset pre-eclampsia. Immunology. 2017;152(1):163–73.
Hanna N, Hanna I, Hleb M, Wagner E, Dougherty J, Balkundi D, et al. Gestational age-dependent expression of IL-10 and its receptor in human placental tissues and isolated cytotrophoblasts. J Immunol. 2000;164(11):5721–8.
Sharma A, Satyam A, Sharma JB. Leptin, IL-10 and inflammatory markers (TNF-alpha, IL-6 and IL-8) in pre-eclamptic, normotensive pregnant and healthy non-pregnant women. Am J Reprod Immunol. 2007;58(1):21–30.
Philippidis P, Mason JC, Evans BJ, Nadra I, Taylor KM, Haskard DO, et al. Hemoglobin scavenger receptor CD163 mediates interleukin-10 release and heme oxygenase-1 synthesis: anti-inflammatory monocyte-macrophage responses in vitro, in resolving skin blisters in vivo, and after cardiopulmonary bypass surgery. Circ Res. 2004;94(1):119–26.
Kronborg CS, Knudsen UB, Moestrup SK, Allen J, Vittinghus E, Møller HJ. Serum markers of macrophage activation in pre-eclampsia: no predictive value of soluble CD163 and neopterin. Acta Obstet Gynecol Scand. 2007;86(9):1041–6.
Sulahian TH, Högger P, Wahner AE, Wardwell K, Goulding NJ, Sorg C, et al. Human monocytes express CD163, which is up-regulated by IL-10 and identical to p155. Cytokine. 2000;12(9):1312–21.
Funding
This study was supported by the Fundação de Amparo a Pesquisa do Estado de Sao Paulo (São Paulo State Research Foundation) no. 2010/20207-0 and no. 2013/12260-7.
Author information
Authors and Affiliations
Contributions
M.T.P. and P.R.N. conceived and designed the experiments. P.R.N. and M.R.V. performed the experiments. P.R.N., M.T.P., and M.R.V. analyzed the data and wrote the manuscript. J.C.P., R.A.A.C., L.G.O., and V.T.M.B. recruited the subjects for clinical evaluation and blood collection. All the authors have seen and approved the final version of the manuscript.
Corresponding author
Ethics declarations
This project was approved by the Ethics Committee of the Botucatu Medical School (Protocol number 417.350), and written informed consent was obtained from all women involved in the study.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nunes, P.R., Romão-Veiga, M., Peraçoli, J.C. et al. Downregulation of CD163 in monocytes and its soluble form in the plasma is associated with a pro-inflammatory profile in pregnant women with preeclampsia. Immunol Res 67, 194–201 (2019). https://doi.org/10.1007/s12026-019-09078-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12026-019-09078-8