Abstract
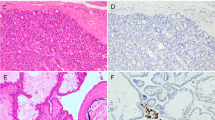
Poorly differentiated thyroid carcinoma (PDTC) is an aggressive form of thyroid cancer that currently has limited effective treatment options. Immune checkpoint inhibitors (ICIs) have shown to be an effective treatment for a variety of carcinomas. In this study, we explore whether immune checkpoint pathways, such as programmed cell death ligand 1 (PD-L1) and indoleamine 2,3-dioxygenase 1 (IDO1), are activated in a cohort of patients with PDTC to determine whether ICIs may be an effective therapy for these patients. PDTC from 28 patients were stained for IDO1, PD-L1, and CD8 using immunohistochemistry. Staining was scored using an H-score, and PD-L1 and IDO1 expression was correlated with clinicopathologic characteristics. Positivity for PD-L1 and IDO1 was set at an H-score cutoff of five. Twenty-five percent (n = 7/28) of the PDTC were positive for PD-L1 expression. Twenty-nine percent (n = 2/7) of the PD-L1 positive PDTCs also co-expressed IDO1. The expression of PD-L1 in PDTC was significantly associated with tumor size and multifocality, with a non-significant trend towards associations with older age, extrathyroidal extension, presence of metastasis, higher stage, increased number of CD8+ T cells, and decreased disease-free and overall survival. PD-L1 expression occurs in a subset of PDTC, and is associated with a subset of clinical features of aggressive thyroid disease. Given the limited effective treatments for this patient population, consideration for ICIs as monotherapy or in combination with an IDO1 inhibitor should be explored as a novel treatment modality for patients with PDTC.



Similar content being viewed by others
References
VolanteM, ColliniP, NikiforovYE, et al (2007) Poorly differentiated thyroid carcinoma: the Turin proposal for the use of uniform diagnostic criteria and an algorithmic diagnostic approach. Am J Surg Pathol31:1256–1264
KakudoK, BaiY, KatayamaS, et al (2009) Classification of follicular cell tumors of the thyroid gland: Analysis involving Japanese patients from one institute. Pathol Int59:359–367
SandersEM, LiVolsiVA, BrierleyJ, et al (2007) An evidence-based review of poorly differentiated thyroid cancer. World J Surg31:934–945
LaiHW, LeeCH, ChenJY, et al (2006) Insular Thyroid Carcinoma: Collective Analysis of Clinicohistologic Prognostic Factors and Treatment Effect with Radioiodine or Radiation Therapy. J Am Coll Surg203:715–722
PatelKN, ShahaAR (2014) Poorly differentiated thyroid cancer. Curr Opin Otolaryngol Head Neck Surg22:121–126
DettmerM, SchmittA, SteinertH, et al (2011) Poorly Differentiated Thyroid Carcinomas: How Much Poorly Differentiated is Needed? Am J Surg Pathol35:1866–1872
NikiforovYE (2004) Genetic alterations involved in the transition from well-differentiated to poorly differentiated and anaplastic thyroid carcinomas. Endocr Pathol15:319–327 . doi: https://doi.org/10.1385/EP:15:4:319
KunstmanJW, Christofer JuhlinC, GohG, et al (2015) Characterization of the mutational landscape of anaplastic thyroid cancer via whole-exome sequencing. Hum Mol Genet24:2318–2329
SykorovaV, DvorakovaS, VcelakJ, et al (2015) Search for new genetic biomarkers in poorly differentiated and anaplastic thyroid carcinomas using next generation sequencing. Anticancer Res35:2029–2036
LandaI, IbrahimpasicT, BoucaiL, et al (2016) Genomic and transcriptomic hallmarks of poorly differentiated and anaplastic thyroid cancers. J Clin Invest126:1052–1066
XuB, GhosseinR (2016) Genomic Landscape of poorly Differentiated and Anaplastic Thyroid Carcinoma. Endocr Pathol27:205–212
LiuR, XingM (2016) TERT promoter mutations in thyroid cancer. Endocr Relat Cancer23:R143–R155 . doi: https://doi.org/10.1530/ERC-15-0533
PaciniF, CastagnaMG, BrilliL, PentheroudakisG (2012) Thyroid cancer: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol23:110–119 . doi: https://doi.org/10.1093/annonc/mds230
CabanillasME, SchlumbergerM, JarzabB, et al (2015) A Phase 2 Trial of Lenvatinib (E7080) in Advanced, Progressive, Radioiodine-Refractory, Differentiated Thyroid Cancer: A Clinical Outcomes and Biomarker Assessment. Cancer121:2749–2756
CabanillasME, HabraMA (2015) Lenvatinib: Role in thyroid cancer and other solid tumors. Cancer Treat Rev42:47–55
PardollDM (2012) The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer12:252–264 . doi: https://doi.org/10.1038/nrc3239
BardhanK, AnagnostouT, BoussiotisVA (2016) The PD1:PD-L1/2 Pathway from Discovery to Clinical Implementation. Front Immunol7:550
DyckL, MillsKHG (2017) Immune checkpoints and their inhibition in cancer and infectious diseases. Eur J Immunol47:765–779
DevjiT, LevineO, NeupaneB, et al (2016) Systemic Therapy for Previously Untreated Advanced BRAF-Mutated Melanoma. JAMA Oncol150:179–185
EllisPM, VellaET, UngYC (2017) Immune Checkpoint Inhibitors for Patients With Advanced Non–Small-Cell Lung Cancer: A Systematic Review. Clin Lung Cancer
MirghaniH, AmenF, BlanchardP, et al (2015) Treatment de-escalation in HPV-positive oropharyngeal carcinoma: Ongoing trials, critical issues and perspectives. Int J Cancer136:1494–1503
KaufmanHL, RussellJ, HamidO, et al (2016) Avelumab in patients with chemotherapy-refractory metastatic Merkel cell carcinoma: a multicentre, single-group, open-label, phase 2 trial. Lancet Oncol17:1374–1385
ChaE, WallinJ, KowanetzM (2015) PD-L1 Inhibition With MPDL3280A for Solid Tumors. Semin Oncol42:484–487
PatnaikA, KangSP, RascoD, et al (2015) Phase I study of pembrolizumab (MK-3475; Anti-PD-1 monoclonal antibody) in patients with advanced solid tumors. Clin Cancer Res21:4286–4293
CarterLL, FouserLA, JussifJ, et al (2002) PD-1:PD-L inhibitory pathway affects both CD4+ and CD8+ T cells and is overcome by IL-2. Eur J Immunol32:634–643
BrahmerJR, TykodiSS, ChowLQM, et al (2012) Safety and activity of anti-PD-L1 antibody in patients with advanced cancer. N Engl J Med366:2455–2465
ChenDS, IrvingBA, HodiFS (2012) Molecular pathways: Next-generation immunotherapy-inhibiting programmed death-ligand 1 and programmed death-1. Clin Cancer Res18:6580–6587
CunhaLL, MarcelloMA, MorariEC, et al (2013) Differentiated thyroid carcinomas may elude the immune system by B7H1 upregulation. Endocr Relat Cancer20:103–110
FrenchJD, KotnisGR, SaidS, et al (2012) Programmed death-1+ T cells and regulatory T cells are enriched in tumor-involved lymph nodes and associated with aggressive features in papillary thyroid cancer. J Clin Endocrinol Metab97:934–943
BraunerE, GundaV, Borre P Vanden, et al. (2016) Combining BRAF inhibitor and anti PD-L1 antibody dramatically improves tumor regression and anti tumor immunity in an immunocompetent murine model of anaplastic thyroid cancer. Oncotarget7:17194–17211
KolliparaR, SchneiderB, RadovichM, et al (2017) Exceptional Response with Immunotherapy in a Patient with Anaplastic Thyroid Cancer. Oncologist. 22:1149–1151
MoffettJR, NamboodiriMA (2003) Tryptophan and the immune response. Immunol Cell Biol81:247–265
LobS, KonigsrainerA, RammenseeHG, et al (2009) Inhibitors of indoleamine-2,3-dioxygenase for cancer therapy: can we see the wood for the trees? Nat Rev Cancer9:445–452
AzumaK, OtaK, KawaharaA, et al (2014) Association of PD-L1 overexpression with activating EGFR mutations in surgically resected nonsmall-cell lung cancer. Ann Oncol25:1935–1940
MorettiS, MenicaliE, VoceP, et al (2014) Indoleamine 2,3-Dioxygenase 1 (IDO1) Is Up-Regulated in Thyroid Carcinoma and Drives the Development of an Immunosuppressant Tumor Microenvironment. J Clin Endocrinol Metab. 99:832–840
CarboneDP, ReckM, Paz-AresL, et al (2017) First-Line Nivolumab in Stage IV or Recurrent Non-Small-Cell Lung Cancer. N Engl J Med376:2415–2426
Lyford-PikeS, PengS, YoungGD, et al (2013) Evidence for a role of the PD-1:PD-L1 pathway in immune resistance of HPV-associated head and neck squamous cell carcinoma. Cancer Res73:1733–1741 . doi: https://doi.org/10.1158/0008-5472.CAN-12-2384
WangQ, WuX (2017) Primary and acquired resistance to PD-1/PD-L1 blockade in cancer treatment. Int Immunopharmacol46:210–219
BelloneM, EliaAR (2017) Constitutive and acquired mechanisms of resistance to immune checkpoint blockade in human cancer. Cytokine Growth Factor Rev36:17–24
SeversonJJ, SerracinoHS, MateescuV, et al (2015) PD-1+Tim-3+ CD8+ T Lymphocytes Display Varied Degrees of Functional Exhaustion in Patients with Regionally Metastatic Differentiated Thyroid Cancer. Cancer Immunol Res3:620–630
AhnS, KimTH, KimSW, et al (2017) Comprehensive screening for PD-L1 expression in thyroid cancer. Endocr Relat Cancer24:97–106
AngellTE, LechnerMG, JangJK, et al (2014) BRAF V600E in papillary thyroid carcinoma is associated with increased programmed death ligand 1 expression and suppressive immune cell infiltration. Thyroid24:1385–1393
ChowdhuryS, VeyhlJ, JessaF, et al (2016) Programmed death-ligand 1 overexpression is a prognostic marker for aggressive papillary thyroid cancer and its variants. Oncotarget7:32318–32328
WangQ, LiuF, LiuL (2017) Prognostic significance of PD-L1 in solid tumors. Medicine (Baltimore)96:e6369
LiJ, WangP, XuY (2017) Prognostic value of programmed cell death ligand 1 expression in patients with head and neck cancer: A systematic review and meta-analysis. PLoS One12:e0179536
XuF, XuL, WangQ, et al (2015) Clinicopathological and prognostic value of programmed death ligand-1 (PD-L1) in renal cell carcinoma: a meta-analysis. Int J Clin Exp Med. 8:14595–14603
PanZ, YeF, WuX, et al (2015) Clinicopathological and prognostic significance of programmed cell death ligand1 (PD-L1) expression in patients with non-small cell lung cancer: a meta-analysis. J Thorac Dis. 7:462–470. https://doi.org/10.3978/j.issn.2072-1439.2015.02.13
ShiR, QuN, LuoT, et al (2017) Programmed Death-Ligand 1 Expression in Papillary Thyroid Cancer and Its Correlation with Clinicopathologic Factors and Recurrence. Thyroid27:537–545
ChenN, FangW, LinZ, et al (2017) KRAS mutation-induced upregulation of PD-L1 mediates immune escape in human lung adenocarcinoma. Cancer Immunol Immunother. 66:1175–1187
JiangX, ZhouJ, Giobbie-HurderA, et al (2013) The activation of MAPK in melanoma cells resistant to BRAF inhibition promotes PD-L1 expression that is reversible by MEK and PI3K inhibition. Clin Cancer Res19:598–609 . doi: https://doi.org/10.1158/1078-0432.CCR-12-2731
OttPA, BhardwajN (2013) Impact of MAPK Pathway Activation in BRAFV600 Melanoma on T Cell and Dendritic Cell Function. Front Immunol4:1–7
LeeS, JangB, LeeS, et al (2006) Interferon regulatory factor-1 is prerequisite to the constitutive expression and IFN-γ-induced upregulation of B7-H1 (CD274). FEBS Lett580:755–762
RibasA (2015) Adaptive immune resistance: How cancer protects from immune attack. Cancer Discov.5:915–919
RooneyMS, ShuklaSA, WuCJ, et al (2014) Article Molecular and Genetic Properties of Tumors Associated with Local Immune Cytolytic Activity. Cell160:48–61
BalermpasP, RödelF, KrauseM, et al (2017) The PD-1/PD-L1 axis and human papilloma virus in patients with head and neck cancer after adjuvant chemoradiotherapy: A multicentre study of the German Cancer Consortium Radiation Oncology Group (DKTK-ROG). Int J Cancer141:594–603
LlosaNJ, CruiseM, TamA, et al (2014) The Vigorous Immune Microenvironment of Microsatellite Instable Colon Cancer Is Balanced by Multiple Counter-Inhibitory Checkpoints. Cancer Discov. 5:43–52
RosenbaumMW, BledsoeJR, Morales-oyarvideV, et al (2016) PD-L1 expression in colorectal cancer is associated with microsatellite instability , BRAF mutation , medullary morphology and cytotoxic tumor-infiltrating lymphocytes. Mod Pathol29:1104–1112
LeDT, DurhamJN, SmithKN, et al (2017) Mismatch-repair deficiency predicts response of solid tumors to PD-1 blockade. 357:409–413
USFDA (2017) FDA grants accelerated approval to pembrolizumab for first tissue/site agnostic indication. https://www.fda.gov/Drugs/InformationOnDrugs/ApprovedDrugs/ucm560040.htm?platform=hootsuite. Accessed 1 Jan 2017
TrottJF, KimJ, AboudOA, et al (2016) Inhibiting tryptophan metabolism enhances interferon therapy in kidney cancer. Oncotarget7:66540–66557
MorettiS, MenicaliE, NucciN, et al (2017) Signal Transducer and Activator of Transcription 1 Plays a Pivotal Role in RET/PTC3 Oncogene-induced Expression of Indoleamine 2,3-Dioxygenase 1. J Biol Chem292:1785–1797
Funding
This work is supported by the NIH/NIDCR R01DE025340 (WCF and SIP), NIH 1R01CA149738-01 (SP), NIH 5T32DK00702842 (BJG), and Stand Up To Cancer-American Cancer Society Dream Team Translation Research Grant (MMK).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Rosenbaum, M.W., Gigliotti, B.J., Pai, S.I. et al. PD-L1 and IDO1 Are Expressed in Poorly Differentiated Thyroid Carcinoma. Endocr Pathol 29, 59–67 (2018). https://doi.org/10.1007/s12022-018-9514-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12022-018-9514-y