Abstract
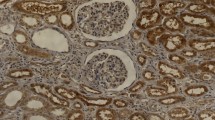
Functional chemokine (C-X-C motif) receptor 4 (CXCR4) is well known to be over-expressed in papillary thyroid carcinoma (PTC). The aim of this study was to evaluate whether or not the expression of CXCR4 is different by histological subtypes of PTC and to elucidate the relationship between the expression of CXCR4 and clinicopathologic factors. CXCR4 expression in 127 PTC samples was assessed using immunohistochemical staining. The expression of CXCR4 showed different patterns according to the histological subtype of PTC (p < 0.001). A strong expression of CXCR4 was observed more frequently in the poorly differentiated region of PTC (81.0 %) than in classical PTC (50.0 %). Strong CXCR4 expression was less frequently shown in follicular variant (33.9 %) and in diffuse sclerosing variant (14.3 %) of PTC. CXCR4 expression showed a distinct pattern according to the histological subtype of PTC although not associated with other clinicopathological parameters.

Similar content being viewed by others
References
Sherman SI. Thyroid carcinoma. Lancet 361:501–11, 2003.
Arturi F, Russo D, Giuffrida D, et al. Early diagnosis by genetic analysis of differentiated thyroid cancer metastases in small lymph nodes. J Clin Endocrinol Metab 82:1638–41, 1997.
Chow SM, Law SC, Chan JK, Au SK, Yau S, Lau WH. Papillary microcarcinoma of the thyroid—prognostic significance of lymph node metastasis and multifocality. Cancer 98:31–40, 2003.
Qubain SW, Nakano S, Baba M, Takao S, Aikou T. Distribution of lymph node micrometastasis in pN0 well-differentiated thyroid carcinoma. Surgery 131:249–56, 2002.
Baudin E, Schlumberger M. New therapeutic approaches for metastatic thyroid carcinoma. Lancet Oncol 8:148–56, 2007.
Rossi D, Zlotnik A. The biology of chemokines and their receptors. Annu Rev Immunol 18:217–42, 2000.
Zlotnik A. Chemokines and cancer. Int J Cancer 119:2026–9, 2006.
Kim CH, Broxmeyer HE. SLC/exodus2/6Ckine/TCA4 induces chemotaxis of hematopoietic progenitor cells: differential activity of ligands of CCR7, CXCR3, or CXCR4 in chemotaxis vs. suppression of progenitor proliferation. J Leukoc Biol 66:455–61, 1999.
Balkwill F. The significance of cancer cell expression of the chemokine receptor CXCR4. Semin Cancer Biol 14:171–9, 2004.
Aust G, Steinert M, Kiessling S, Kamprad M, Simchen C. Reduced expression of stromal-derived factor 1 in autonomous thyroid adenomas and its regulation in thyroid-derived cells. J Clin Endocrinol Metab 86:3368–76, 2001.
Castellone MD, Guarino V, De Falco V, et al. Functional expression of the CXCR4 chemokine receptor is induced by RET/PTC oncogenes and is a common event in human papillary thyroid carcinomas. Oncogene 23:5958–67, 2004.
De Falco V, Guarino V, Avilla E, et al. Biological role and potential therapeutic targeting of the chemokine receptor CXCR4 in undifferentiated thyroid cancer. Cancer Res 67:11821–9, 2007.
Hwang JH, Hwang JH, Chung HK, et al. CXC chemokine receptor 4 expression and function in human anaplastic thyroid cancer cells. J Clin Endocrinol Metab 88:408–16, 2003.
González HE, Leiva A, Tobar H, et al. Altered chemokine receptor expression in papillary thyroid cancer. Thyroid 19:957–65, 2009.
Wagner PL, Moo T-A, Arora N, et al. The chemokine receptors CXCR4 and CCR7 are associated with tumor size and pathologic indicators of tumor aggressiveness in papillary thyroid carcinoma. Ann Surg Oncol 15:2833–41, 2008.
Muller A, Homey B, Soto H, et al. Involvement of chemokine receptors in breast cancer metastasis. Nature 410:50–6, 2001.
Orimo A, Gupta PB, Sgroi DC, et al. Stromal fibroblasts present in invasive human breast carcinomas promote tumor growth and angiogenesis through elevated SDF-1/CXCL12 secretion. Cell 121:335–48, 2005.
Borrello MG, Alberti L, Fischer A, et al. Induction of a proinflammatory program in normal human thyrocytes by the RET/PTC1 oncogene. Proc Natl Acad Sci USA 102:14825–30, 2005.
Yasuoka H, Kodama R, Hirokawa M, et al. CXCR4 expression in papillary thyroid carcinoma: induction by nitric oxide and correlation with lymph node metastasis. BMC Cancer 8:274, 2008.
Burningham AR, Krishnan J, Davidson BJ, Ringel MD, Burman KD. Papillary and follicular variant of papillary carcinoma of the thyroid: initial presentation and response to therapy. Otolaryngol Head Neck Surg 132:840–4, 2005.
Passler C, Prager G, Scheuba C, et al. Follicular variant of papillary thyroid carcinoma: a long-term follow-up. Arch Surg 138:1362–6, 2003.
Zidan J, Karen D, Stein M, Rosenblatt E, Basher W, Kuten A. Pure versus follicular variant of papillary thyroid carcinoma: clinical features, prognostic factors, treatment, and survival. Cancer 97:1181–5, 2003.
Chow SM, Chan JK, Law SC, et al. Diffuse sclerosing variant of papillary thyroid carcinoma—clinical features and outcome. Eur J Surg Oncol 29:446–9, 2003.
Vickery AL, Jr., Carcangiu ML, Johannessen JV, Sobrinho-Simoes M. Papillary carcinoma. Semin Diagn Pathol 2:90–100, 1985.
Albareda M, Puig-Domingo M, Wengrowicz S, et al. Clinical forms of presentation and evolution of diffuse sclerosing variant of papillary carcinoma and insular variant of follicular carcinoma of the thyroid. Thyroid 8:385–91, 1998.
Carcangiu ML, Bianchi S. Diffuse sclerosing variant of papillary thyroid carcinoma. Clinicopathologic study of 15 cases. Am J Surg Pathol 13:1041–9, 1989.
Koo JS, Hong S, Park CS. Diffuse sclerosing variant is a major subtype of papillary thyroid carcinoma in the young. Thyroid 19:1225–31, 2009.
Ito Y, Hirokawa M, Fukushima M, et al. Prevalence and prognostic significance of poor differentiation and tall cell variant in papillary carcinoma in Japan. World J Surg 32:1535–43; discussion 44–5, 2008.
Volante M, Collini P, Nikiforov YE, et al. Poorly differentiated thyroid carcinoma: the Turin proposal for the use of uniform diagnostic criteria and an algorithmic diagnostic approach. Am J Surg Pathol 31:1256–64, 2007.
He X, Wei Q, Zhang X, et al. Immunohistochemical expression of CXCR4 in thyroid carcinomas and thyroid benign lesions. Pathol Res Pract 206:712–5, 2010.
Nikiforova MN, Kimura et, Gandhi M, et al. BRAF mutations in thyroid tumors are restricted to papillary carcinomas and anaplastic or poorly differentiated carcinomas arising from papillary carcinomas. J Clin Endocrinol Metab 88:5399–404, 2003.
Zhu Z, Gandhi M, Nikiforova MN, Fischer AH, Nikiforov YE. Molecular profile and clinical-pathologic features of the follicular variant of papillary thyroid carcinoma. An unusually high prevalence of ras mutations. Am J Clin Pathol 120:71–7, 2003.
Lam AK, Lo CY. Diffuse sclerosing variant of papillary carcinoma of the thyroid: a 35-year comparative study at a single institution. Ann Surg Oncol 13:176–81, 2006.
Thompson LD, Wieneke JA, Heffess CS. Diffuse sclerosing variant of papillary thyroid carcinoma: a clinicopathologic and immunophenotypic analysis of 22 cases. Endocr Pathol 16:331–48, 2005.
Acknowledgments
This work was supported by Yonsei University College of Medicine faculty research grant 2010 (No. 6-2010-0137).
Conflict of interest
The authors declare that there is no conflict of interest that could be perceived as prejudicing the impartiality of the research reported.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shin, D.Y., Kim, K.J., Ku, C.R. et al. Different CXCR4 Expression According to Various Histologic Subtype of Papillary Thyroid Carcinoma. Endocr Pathol 24, 169–176 (2013). https://doi.org/10.1007/s12022-013-9259-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12022-013-9259-6