Abstract
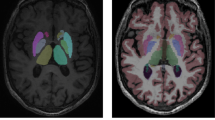
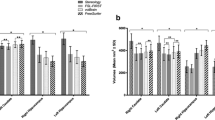
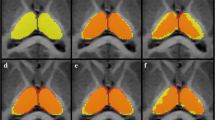
Volumetric and morphometric neuroimaging studies of the basal ganglia and thalamus in pediatric populations have utilized existing automated segmentation tools including FIRST (Functional Magnetic Resonance Imaging of the Brain’s Integrated Registration and Segmentation Tool) and FreeSurfer. These segmentation packages, however, are mostly based on adult training data. Given that there are marked differences between the pediatric and adult brain, it is likely an age-specific segmentation technique will produce more accurate segmentation results. In this study, we describe a new automated segmentation technique for analysis of 7-year-old basal ganglia and thalamus, called Pediatric Subcortical Segmentation Technique (PSST). PSST consists of a probabilistic 7-year-old subcortical gray matter atlas (accumbens, caudate, pallidum, putamen and thalamus) combined with a customized segmentation pipeline using existing tools: ANTs (Advanced Normalization Tools) and SPM (Statistical Parametric Mapping). The segmentation accuracy of PSST in 7-year-old data was compared against FIRST and FreeSurfer, relative to manual segmentation as the ground truth, utilizing spatial overlap (Dice’s coefficient), volume correlation (intraclass correlation coefficient, ICC) and limits of agreement (Bland-Altman plots). PSST achieved spatial overlap scores ≥90 % and ICC scores ≥0.77 when compared with manual segmentation, for all structures except the accumbens. Compared with FIRST and FreeSurfer, PSST showed higher spatial overlap (p FDR < 0.05) and ICC scores, with less volumetric bias according to Bland-Altman plots. PSST is a customized segmentation pipeline with an age-specific atlas that accurately segments typical and atypical basal ganglia and thalami at age 7 years, and has the potential to be applied to other pediatric datasets.





Similar content being viewed by others
Abbreviations
- ANTs:
-
Advanced Neuroimaging Tools
- BET:
-
Brain Extraction Tool
- CMA:
-
Center for Morphometric Analysis
- CSF:
-
Cerebrospinal fluid
- FDR:
-
False discovery rate
- FIRST:
-
Functional Magnetic Resonance Imaging of the Brain’s Integrated Registration and Segmentation Tool
- FMRIB:
-
Functional Magnetic Resonance Imaging of the Brain
- FSL:
-
Functional Magnetic Resonance Imaging of the Brain’s Software Library
- FWHM:
-
Full width half maximum
- ICC:
-
Intraclass correlation coefficient
- ITK:
-
Insight Toolkit
- LDDM:
-
Large Deformation Diffeomorphic Metric
- MRI:
-
Magnetic resonance imaging
- PSST:
-
Pediatric Subcortical Segmentation Technique
- SPM:
-
Statistical Parametric Mapping
- VIBeS:
-
Victorian Infant Brain Study
References
Ahn, M. S., Breeze, J. L., Makris, N., Kennedy, D. N., Hodge, S. M., Herbert, M. R., et al. (2007). Anatomic brain magnetic resonance imaging of the basal ganglia in pediatric bipolar disorder. Journal of Affective Disorders, 104(1–3), 147–154.
Alexander, G. E., DeLong, M. R., & Strick, P. L. (1986). Parallel organization of functionally segregated circuits linking basal ganglia and cortex. Annual Review of Neuroscience, 9(1), 357–381.
Ashburner, J., & Friston, K. J. (2005). Unified segmentation. NeuroImage, 26(3), 839–851.
Avants, B. B., Epstein, C. L., Grossman, M., & Gee, J. C. (2008). Symmetric diffeomorphic image registration with cross-correlation: evaluating automated labeling of elderly and neurodegenerative brain. Medical Image Analysis, 12(1), 26–41.
Avants, B. B., Yushkevich, P., Pluta, J., Minkoff, D., Korczykowski, M., Detre, J., et al. (2010). The optimal template effect in hippocampus studies of diseased populations. NeuroImage, 49(3), 2457–2466.
Aylward, E. H., Nopoulos, P. C., Ross, C. A., Langbehn, D. R., Pierson, R. K., Mills, J. A., et al. (2011). Longitudinal change in regional brain volumes in prodromal Huntington disease. Journal of Neurology, Neurosurgery and Psychiatry, 82(4), 405–410.
Babalola, K. O., Patenaude, B., Aljabar, P., Schnabel, J., Kennedy, D., Crum, W., et al. (2009). An evaluation of four automatic methods of segmenting the subcortical structures in the brain. NeuroImage, 47(4), 1435–1447.
Bigler, E. D., Abildskov, T. J., Wilde, E. A., McCauley, S. R., Li, X., Merkley, T. L., et al. (2010). Diffuse damage in pediatric traumatic brain injury: a comparison of automated versus operator-controlled quantification methods. NeuroImage, 50(3), 1017–1026.
Cardenas, V. A., Price, M., Infante, M. A., Moore, E. M., Mattson, S. N., Riley, E. P., et al. (2014). Automated cerebellar segmentation: validation and application to detect smaller volumes in children prenatally exposed to alcohol. NeuroImage: Clinical, 4, 295–301.
Dennison, M., Whittle, S., Yücel, M., Vijayakumar, N., Kline, A., Simmons, J., et al. (2013). Mapping subcortical brain maturation during adolescence: evidence of hemisphere- and sex-specific longitudinal changes. Developmental Science, 16(5), 772–791.
Dewey, J., Hana, G., Russell, T., Price, J., McCaffrey, D., Harezlak, J., et al. (2010). Reliability and validity of MRI-based automated volumetry software relative to auto-assisted manual measurement of subcortical structures in HIV-infected patients from a multisite study. NeuroImage, 51(4), 1334–1344.
Fischl, B., Salat, D. H., Busa, E., Albert, M., Dieterich, M., Haselgrove, C., et al. (2002). Whole brain segmentation: automated labeling of neuroanatomical structures in the human brain. Neuron, 33(3), 341–355.
Fleisher, A. S., Sun, S., Taylor, C., Ward, C. P., Gamst, A. C., Petersen, R. C., et al. (2008). Volumetric MRI vs clinical predictors of Alzheimer disease in mild cognitive impairment. Neurology, 70(3), 191–199.
Fonov, V., Evans, A. C., Botteron, K., Almli, C. R., McKinstry, R. C., & Collins, D. L. (2011). Unbiased average age-appropriate atlases for pediatric studies. NeuroImage, 54(1), 313–327.
Giedd, J. N., & Rapoport, J. L. (2010). Structural MRI of pediatric brain development: what have we learned and where are we going? Neuron, 67(5), 728–734.
Giedd, J. N., Snell, J. W., Lange, N., Rajapakse, J. C., Casey, B. J., Kozuch, P. L., et al. (1996). Quantitative magnetic resonance imaging of human brain development: ages 4–18. Cerebral Cortex, 6(4), 551–559.
Glenthoj, A., Glenthoj, B. Y., Mackeprang, T., Pagsberg, A. K., Hemmingsen, R. P., Jernigan, T. L., et al. (2007). Basal ganglia volumes in drug-naive first-episode schizophrenia patients before and after short-term treatment with either a typical or an atypical antipsychotic drug. Psychiatry Research, 154(3), 199–208.
Haber, S. N. (2003). The primate basal ganglia: parallel and integrative networks. Journal of Chemical Neuroanatomy, 26(4), 317–330.
Haber, S. N., Kim, K.-S., Mailly, P., & Calzavara, R. (2006). Reward-related cortical inputs define a large Striatal region in primates that interface with associative cortical connections, providing a substrate for incentive-based learning. The Journal of Neuroscience, 26(32), 8368–8376.
Helmich, R. C., Derikx, L. C., Bakker, M., Scheeringa, R., Bloem, B. R., & Toni, I. (2010). Spatial remapping of cortico-striatal connectivity in Parkinson’s disease. Cerebral Cortex, 20(5), 1175–1186.
Hoeksma, M. R., Kenemans, J. L., Kemner, C., & van Engeland, H. (2005). Variability in spatial normalization of pediatric and adult brain images. Clinical Neurophysiology, 116(5), 1188–1194.
Kawadler, J. M., Clayden, J. D., Kirkham, F. J., Cox, T. C., Saunders, D. E., & Clark, C. A. (2013). Subcortical and cerebellar volumetric deficits in paediatric sickle cell anaemia. British Journal of Haematology, 163(3), 373–376.
Keller, S., Gerdes, J., Mohammadi, S., Kellinghaus, C., Kugel, H., Deppe, K., et al. (2012). Volume estimation of the thalamus using Freesurfer and stereology: consistency between methods. Neuroinformatics, 10(4), 341–350.
Khan, A. R., Wang, L., & Beg, M. F. (2008). FreeSurfer-initiated fully-automated subcortical brain segmentation in MRI using large deformation diffeomorphic metric mapping. NeuroImage, 41(3), 735–746.
Langen, M., Durston, S., Staal, W. G., Palmen, S. J. M. C., & van Engeland, H. (2007). Caudate nucleus is enlarged in high-functioning medication-naive subjects with autism. Biological Psychiatry, 62(3), 262–266.
Lee, H. M., Kwon, K.-Y., Kim, M.-J., Jang, J.-W., Suh, S.-i., & Koh, S.-B. (2014). Subcortical grey matter changes in untreated, early stage Parkinson’s disease without dementia. Parkinsonism & Related Disorders, 20(6), 622–626.
Lenroot, R. K., & Giedd, J. N. (2006). Brain development in children and adolescents: insights from anatomical magnetic resonance imaging. Neuroscience & Biobehavioral Reviews, 30(6), 718–729.
Lenroot, R. K., Gogtay, N., Greenstein, D. K., Wells, E. M., Wallace, G. L., Clasen, L. S., et al. (2007). Sexual dimorphism of brain developmental trajectories during childhood and adolescence. NeuroImage, 36(4), 1065–1073.
Lin, J. J., Siddarth, P., Riley, J. D., Gurbani, S. G., Ly, R., Yee, V. W., et al. (2013). Neurobehavioral comorbidities of pediatric epilepsies are linked to thalamic structural abnormalities. Epilepsia, 54(12), 2116–2124.
Morey, R. A., Petty, C. M., Xu, Y., Pannu Hayes, J., Wagner Ii, H. R., Lewis, D. V., et al. (2009). A comparison of automated segmentation and manual tracing for quantifying hippocampal and amygdala volumes. NeuroImage, 45(3), 855–866.
Muzik, O., Chugani, D. C., Juhász, C., Shen, C., & Chugani, H. T. (2000). Statistical parametric mapping: assessment of application in children. NeuroImage, 12(5), 538–549.
Omizzolo, C., Thompson, D. K., Scratch, S. E., Stargatt, R., Lee, K. J., Cheong, J., et al. (2013). Hippocampal volume and memory and learning outcomes at 7 years in children born very preterm. Journal of the International Neuropsychological Society, 19(10), 1065–1075.
Østby, Y., Tamnes, C. K., Fjell, A. M., Westlye, L. T., Due-Tønnessen, P., & Walhovd, K. B. (2009). Heterogeneity in subcortical brain development: a structural magnetic resonance imaging study of brain maturation from 8 to 30 years. The Journal of Neuroscience, 29(38), 11772–11782.
Pardoe, H. R., Pell, G. S., Abbott, D. F., & Jackson, G. D. (2009). Hippocampal volume assessment in temporal lobe epilepsy: how good is automated segmentation? Epilepsia, 50(12), 2586–2592.
Patenaude, B., Smith, S. M., Kennedy, D. N., & Jenkinson, M. (2011). A Bayesian model of shape and appearance for subcortical brain segmentation. NeuroImage, 56(3), 907–922.
Qiu, A., Crocetti, D., Adler, M., Mahone, E. M., Denckla, M. B., Miller, M. I., et al. (2009). Basal ganglia volume and shape in children with attention deficit hyperactivity disorder. The American Journal of Psychiatry, 166(1), 74–82.
Raznahan, A., Shaw, P. W., Lerch, J. P., Clasen, L. S., Greenstein, D., Berman, R., et al. (2014). Longitudinal four-dimensional mapping of subcortical anatomy in human development. Proceedings of the National Academy of Sciences, 111(4), 1592–1597.
Redgrave, P., Rodriguez, M., Smith, Y., Rodriguez-Oroz, M. C., Lehericy, S., Bergman, H., et al. (2010). Goal-directed and habitual control in the basal ganglia: implications for Parkinson’s disease. Nature Reviews Neuroscience, 11(11), 760–772.
Sánchez-Benavides, G., Gómez-Ansón, B., Sainz, A., Vives, Y., Delfino, M., & Peña-Casanova, J. (2010). Manual validation of FreeSurfer’s automated hippocampal segmentation in normal aging, mild cognitive impairment, and Alzheimer disease subjects. Psychiatry Research: Neuroimaging, 181(3), 219–225.
Sandman, C. A., Head, K., Muftuler, L. T., Su, L., Buss, C., & Davis, E. P. (2014). Shape of the basal ganglia in preadolescent children is associated with cognitive performance. NeuroImage, 99, 93–102.
Silk, T. J., Vance, A., Rinehart, N., Bradshaw, J. L., & Cunnington, R. (2009). Structural development of the basal ganglia in attention deficit hyperactivity disorder: a diffusion tensor imaging study. Psychiatry Research: Neuroimaging, 172(3), 220–225.
Smith, S. M. (2002). Fast robust automated brain extraction. Human Brain Mapping, 17(3), 143–155.
Sowell, E. R., Trauner, D. A., Gamst, A., & Jernigan, T. L. (2002). Development of cortical and subcortical brain structures in childhood and adolescence: a structural MRI study. Developmental Medicine and Child Neurology, 44(1), 4–16.
Thompson, P. M., Mega, M. S., Woods, R. P., Zoumalan, C. I., Lindshield, C. J., Blanton, R. E., et al. (2001). Cortical change in Alzheimer’s disease detected with a disease-specific population-based brain atlas. Cerebral Cortex, 11(1), 1–16.
Thompson, D. K., Adamson, C., Roberts, G., Faggian, N., Wood, S. J., Warfield, S. K., et al. (2013). Hippocampal shape variations at term equivalent age in very preterm infants compared with term controls: perinatal predictors and functional significance at age 7. NeuroImage, 70, 278–287.
Wierenga, L., Langen, M., Ambrosino, S., van Dijk, S., Oranje, B., & Durston, S. (2014). Typical development of basal ganglia, hippocampus, amygdala and cerebellum from age 7 to 24. NeuroImage, 96, 67–72.
Wilke, M., Schmithorst, V. J., & Holland, S. K. (2003). Normative pediatric brain data for spatial normalization and segmentation differs from standard adult data. Magnetic Resonance in Medicine, 50(4), 749–757.
Wilke, M., Holland, S. K., Altaye, M., & Gaser, C. (2008). Template-O-Matic: a toolbox for creating customized pediatric templates. NeuroImage, 41(3), 903–913.
Yoon, U., Fonov, V. S., Perusse, D., & Evans, A. C. (2009). The effect of template choice on morphometric analysis of pediatric brain data. NeuroImage, 45(3), 769–777.
Yushkevich, P. A., Piven, J., Hazlett, H. C., Smith, R. G., Ho, S., Gee, J. C., et al. (2006). User-guided 3D active contour segmentation of anatomical structures: significantly improved efficiency and reliability. NeuroImage, 31(3), 1116–1128.
Information Sharing Statement
The age-specific (7 years) basal ganglia and thalamus atlas which includes the T 1 template and the structure priors is available upon request from W.Y. Loh (corresponding author) and D. K. Thompson.
Acknowledgments
We gratefully recognize the efforts of Terrie Inder who provided insight into the study, Merilyn Bear who recruited the participants, Michael Kean and the radiographers at Melbourne Children’s MRI Centre who scanned the participants, the VIBeS and Developmental Imaging groups at the Murdoch Childrens Research Institute where the research was conducted. We also thank the families and children who participated in this study.
Funding
This study was supported by Australia’s National Health & Medical Research Council (Centre for Clinical Research Excellence 546519 to L.D. and P.A.; Centre for Research Excellence 1060733 to L.D., P.A., J.C., D.T., A.S., and W.Y.L.; Project Grants 237117 to L.D., 491209 to P.A.; Senior Research Fellowship 628371 to P.A., Early Career Fellowships 1053767 to A.S., 1012236 to D.T., 1053787 to J.C., National Institutes of Health (HD058056), the Victorian Government’s Operational Infrastructure Support Program, and The Royal Children’s Hospital Foundation.
Conflict of Interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Appendices
Appendix A
Manual tracing boundaries
A. Basal ganglia
The basal ganglia nuclei were segmented in the following order: caudate, putamen, pallidum and accumbens. Segmentation of the caudate began at the slice it first appeared and tracing stopped when it was difficult to visualize (the tail of the caudate was not included). The medial and lateral borders of the caudate were respectively defined by the lateral border of the lateral ventricle and the medial border of the internal capsule. Delineation of the putamen began anteriorly when it was first visualized at the ventrolateral section of the caudate and ended when it could no longer be seen. The medial border of the putamen was marked by the structure of the pallidum, while its lateral border was defined by the medial border of the external capsule. Striations in between the caudate and putamen were excluded. For the pallidum, tracing began when it was first seen medial to the putamen and ended when poorly visualized. Segmentation of the accumbens was done last as this structure was inferiorly connected to both the caudate and putamen. The first anterior slice of the accumbens was defined to start five slices after the putamen was first seen, while the last slice was defined to be two slices anterior to the anterior commissure. Before the caudate and putamen were connected inferiorly, the accumbens was superiorly bordered by a line which was drawn from the most inferiolateral voxel of the lateral ventricle to the most inferiomedial voxel of the putamen. Once the caudate and putamen were visualized as an integrated structure, the superior border of the accumbens was marked by a line connecting the most inferiolateral voxel of the lateral ventricle to the most inferiolateral point of the internal capsule; while its lateral border was defined by a vertical line drawn from the internal capsule’s most inferiolateral voxel.
B. Thalamus
Tracing of the thalamus began when it was first visualized a few slices after the anterior commissure appeared and ended when it could no longer be seen. Segmentation of the thalamus included both the lateral and medial geniculate nucleus, as well as the interthalamic adhesion.
Appendix B
Rights and permissions
About this article
Cite this article
Loh, W.Y., Connelly, A., Cheong, J.L.Y. et al. A New MRI-Based Pediatric Subcortical Segmentation Technique (PSST). Neuroinform 14, 69–81 (2016). https://doi.org/10.1007/s12021-015-9279-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12021-015-9279-0