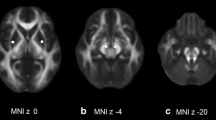
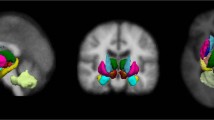
Abstract
There is a compelling need for early, accurate diagnosis of Parkinson’s disease (PD). Various magnetic resonance imaging modalities are being explored as an adjunct to diagnosis. A significant challenge in using MR imaging for diagnosis is developing appropriate algorithms for extracting diagnostically relevant information from brain images. In previous work, we have demonstrated that individual subject variability can have a substantial effect on identifying and determining the borders of regions of analysis, and that this variability may impact on prediction accuracy. In this paper we evaluate a new statistical algorithm to determine if we can improve accuracy of prediction using a subjects left-out validation of a DTI analysis. Twenty subjects with PD and 22 healthy controls were imaged to evaluate if a full brain diffusion tensor imaging-fractional anisotropy (DTI-FA) map might be capable of segregating PD from controls. In this paper, we present a new statistical algorithm based on bootstrapping. We compare the capacity of this algorithm to classify the identity of subjects left out of the analysis with the accuracy of other statistical techniques, including standard cluster-thresholding. The bootstrapped analysis approach was able to correctly discriminate the 20 subjects with PD from the 22 healthy controls (area under the receiver operator curve or AUROC 0.90); however the sensitivity and specificity of standard cluster-thresholding techniques at various voxel-specific thresholds were less effective (AUROC 0.72–0.75). Based on these results sufficient information to generate diagnostically relevant statistical maps may already be collected by current MRI scanners. We present one statistical technique that might be used to extract diagnostically relevant information from a full brain analysis.




Similar content being viewed by others
References
Bennet, C. M., Wolford, G. L., & Miller, M. B. (2009). The principled control of false positives in neuroimaging. Social Cognitive and Affective Neuroscience, 4(4), 417–422.
Brodoehl, S., Klingner, C., Volk, G. F., Bitter, T., Witte, O. W., & Redecker, C. (2012). Decreased olfactory bulb volume in idiopathic Parkinson’s disease detected by 3.0-Tesla magnetic resonance imaging. Movement Disorders, 27(8), 1019–1025.
Caprihan, A., Pearlson, G. D., & Calhoun, V. D. (2008). Application of principal component analysis to distinguish patients with schizophrenia from healthy controls based on fractional anisotropy measurements. NeuroImage, 42(2), 675–682.
Cox, R. W. (1996). AFNI: software for analysis and visualization of functional magnetic resonance neuroimages. Computers and Biomedical Research, 29, 162–173.
DiCiccio, T. J., & Romano, J. P. (1989). A review of bootstrap confidence intervals. Journal of the Royal Statistical Society, Series B, 50, 338–354.
Efron, B. (1979). Bootstrap methods: another look at the jackknife. Annals of Statistics, 7, 1–26.
Eidelberg, D. (2009). Metabolic brain networks in neurodegenerative disorders: a functional imaging approach. Trends in Neurosciences, 32(10), 548–557.
Focke, N. K., Helms, G., Scheewe, S., Pantel, P. M., Bachmann, C. G., Dechent, P., et al. (2011). Individual voxel-based subtype prediction can differentiate progressive supranuclear palsy from idiopathic Parkinson syndrome and healthy controls. Human Brain Mapping, 32(11), 1905–1915.
Forman, S. D., Cohen, J. D., Fitzgerald, M., Eddy, W. F., Mintun, M. A., & Noll, D. C. (1995). Improved assessment of significant activation in functional magnetic resonance imaging (fMRI): use of a cluster-size threshold. Magnetic Resonance in Medicine, 33(5), 636–647.
Gallivan, J. P., McLean, D. A., Flanagan, J. R., & Culham, J. C. (2013). Where one hand meets the other: limb-specific and action-dependent movement plans decoded from preparatory signals in single human frontoparietal brain areas. The Journal of Neuroscience, 30(5), 1991–2008. 33.
Gorell, J. M., Ordidge, R. J., Brown, G. G., Deniau, J. C., & Buderer, N. M. (1995). Helpern. Increased iron‐related MRI contrast in the substantia nigra in Parkinson’s disease. Neurology, 459(6), 1138–1143.
Habeck, C., Foster, N. L., Perneczky, R., Kurz, A., Alexopoulos, P., Koeppe, R. A., et al. (2008). Multivariate and univariate neuroimaging biomarkers of Alzheimer’s disease. NeuroImage, 40, 1503–1515.
Haller, S., Badoud, S., Nguyen, D., Barnaure, I., Montandon, M. L., Lovblad, K. O., et al. (2012). Differentiation between Parkinson disease and other forms of Parkinsonism using support vector machine analysis of susceptibility-weighted imaging (SWI): Initial results. European Radiology http://us.datscan.com/patient/support-resources/locate-datscan-imaging-center.
Huang, C., Mattis, P., Tang, C., Perine, K., Carbon, M., & Eidelberg, D. (2007). Metabolic brain networks associated with cognitive function in Parkinson’s disease. NeuroImage, 34(2), 714–723.
Huang, C., Mattis, P., Perine, K., Brown, N., Dhawan, V., & Eidelberg, D. (2008). Metabolic abnormalities associated with mild cognitive impairment in Parkinson disease. Neurology, 70(16 pt 2), 1470–1477.
Junger, J., Pauly, K., Brohr, S., Birkholz, P., Neuschaefer-Rue, C., Kohler, C., et al. (2013). Sex matters: neural correlates of voice gender perception. NeuroImage, 79, 275–287.
Lauzon, et al. (2013). Simultaneous analysis and quality assurance for diffusion tensor imaging. PLOS One, 8(4), e61737.
Leunissen, I., Coxon, J. P., Geurts, M., Caeyenberghs, K., Michiels, K., Sunaert, S., et al. (2013). Disturbed cortic-subcortical interactions during motor task switching in traumatic brain injury. Human Brain Mapping, 34, 1254–1271.
Ma, Y., Huang, C., Dyke, J. P., Pan, H., Alsop, D., Feigin, A., et al. (2010). Parkinson’s disease spatial covariance pattern: noninvasive quantification with perfusion MRI. Journal of Cerebral Blood Flow and Metabolism, 30(3), 505–509.
Melzer, et al. (2013). White matter microstructure deteriorates across cognitive stages in Parkinson disease. Neurology, 80, 1841.
Monti, M. M., Pickard, J. D., & Owen, A. M. (2013). Visual cognition in disorders of consciousness: from VI to top-down attention. Human Brain Mapping, 34, 1245–1253.
Mure, H., Hirano, S., Tang, C. C., Isaias, I. U., Antonini, A., Ma, Y., et al. (2011). Parkinson’s disease tremor-related metabolic network: characterization, progression, and treatment effects. NeuroImage, 54, 1244–1253.
Nichols, T., & Hayasaka, S. (2003). Controlling the family wise error rate in functional neuroimaging: a comparative review. Statistical Methods in Medical Research, 12(5), 419–446.
Péran, P., Cherubini, A., Assogna, F., Piras, F., Quattrocchi, C., Peppe, A., et al. (2010). Magnetic resonance imaging markers of Parkinson’s disease nigrostriatal signature. Brain, 33(11), 3423–3433.
Shalom Michaeli, S., Oz, G., Sorce, D. J., Garwood, M., Ugurbil, K., Majestic, S., et al. (2007). Assessment of brain iron and neuronal integrity in patients with Parkinson’s disease using novel MRI contrasts. Movement Disorders, 22(3), 334–340.
Skidmore, F. M., Yang, M., Baxter, L., von Deneen, K. D., Collingwood, J., He, G., et al. (2011a). Reliability analysis of the resting state can sensitively and specifically identify the presence of Parkinson disease. NeuroImage, 75, 249–261.
Skidmore, F. M., Spetsieris, P., Yang, M., Gold, M., Heilman, K. M., Collingwood, J., et al. (2011b). Diagnosis of Parkinson’s disease using resting state fMRI. Poster LB22, Movement Disorders, Toronto, June 2011.
Skidmore, F. M., Yang, M., Baxter, L., von Deneen, K., Collingwood, J., He, G., et al. (2011c). Apathy, depression, and motor symptoms have distinct and separable resting activity patterns in idiopathic Parkinson disease. NeuroImage, 81, 484–495.
Smith, S. M., & Nichols, T. E. (2009). Threshold-free cluster enhancement: addressing problems of smoothing, threshold dependence and localisation in cluster inference. NeuroImage, 44(1), 83–98.
Tang, C. C., Poston, K. L., Dhawan, V., & Eidelberg, D. (2010a). Abnormalities in metabolic network activity precede the onset of motor symptoms in Parkinson’s disease. Journal of Neuroscience, 30(3), 1049–1056.
Tang, C. C.*., Poston, K. L.*., Eckveer, T., Feigin, A., Frucht, S., Gudesblatt, M., et al. (2010b). Differential diagnosis of Parkinsonism: a metabolic imaging study using pattern analysis. Lancet Neurology, 9, 149–158. * Equal first author contribution.
Theilmann, et al. (2013). White-matter changes correlate with cognitive functioning in Parkinson’s disease. Frontiers in Neurology, 4, 37.
Vaillancourt, D. E., Spraker, M. B. BS, Prodoehl, J., Abraham, I., Corcos, D. M., Zhou, X. J., et al. (2009). High-resolution diffusion tensor imaging in the substantia nigra of de novo Parkinson disease. Neurology, 72(16), 1378–1384.
Villalon, D., et al. (2013). White matter microstructural abnormalities in girls with chromosome 22q11.2 deletion syndrome. NeuroImage. doi:10.1016/j.neuroimage.2013.04.028.
Zhan, W., Kang, G. A., Glass, G. A., Zhang, Y., Shirley, C., Millin, R., et al. (2012). Regional alterations of brain microstructure in Parkinson’s disease using diffusion tensor imaging. Movement Disorders, 27(1), 90–97.
Zhang, K., Yua, C., Zhang, Y., Wu, X., Zhuc, C., Chan, P., et al. (2011). Regional alterations of brain microstructure in Parkinson’s disease using diffusion tensor imaging. European Journal of Radiology, 77, 269–273.
Acknowledgments
Funding for this project was provided by the nonprofit fund Hawg Wild for the Cure with special thanks to Capital City Harley Davidson and Gainesville Harley Davidson, as well as by the National Center for Advancing Translational Sciences of the National Institutes of Health under award number UL1TR00165. Additional funding for this project came from the Micanopy Doc Hollywood Fund.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Skidmore, F.M., Spetsieris, P.G., Anthony, T. et al. A Full-Brain, Bootstrapped Analysis of Diffusion Tensor Imaging Robustly Differentiates Parkinson Disease from Healthy Controls. Neuroinform 13, 7–18 (2015). https://doi.org/10.1007/s12021-014-9222-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12021-014-9222-9