Abstract
Purpose
(1) to compare clinical, biochemical features in female patients with hypoestrogenism due to childhood- and adult-onset CP; (2) to reveal effects of estrogen replacement therapy in female patients with childhood-onset CP.
Methods
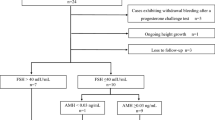
Thirty-seven women that received specific treatment for CP in the period from 1980 to 2019 were recruited: 21 with childhood-onset and 16 with adult-onset CP. Clinical and hormonal characteristics were evaluated. Seventeen-beta-estradiol 2 mg and dydrogesterone 10 mg in sequential regiment was used in 18 childhood-onset cases. Mean follow-up was 31 months.
Results
Amenorrheic women with childhood- and adult-onset CP presented with the same complaints except for lack of genital hair and breast hypoplasia, which were common in patients with childhood-onset CP. BMI was lower in childhood-onset CP group, as was the proportion of overweight patients. They had more favorable lipid profile. The levels of estradiol, testosterone and DHEA-S were low and did not differ. Uterine and ovary volumes were reduced in all patients, but the decline was noticeable in the childhood-onset group. Mineral bone density of lumbar vertebrae was diminished in childhood-onset group. Estrogen therapy in these patients led to clinical improvement: increase in BMD in lumbar spine without negative changes in BMI and/or lipid profile.
Conclusions
Study showed that women with childhood-onset CP had less negative metabolic changes. However, they have more pronounced breast and uterus hypoplasia and lower BMD in lumbar spine. The estrogen replacement therapy led to clinical improvement and BMD increase in lumbar spine without increase of BMI and/or lipid profile changes.





Similar content being viewed by others
References
D.N. Louis, A. Perry, P. Wesseling, D.J. Brat, I.A. Cree, D. Figarella-Branger, C. Hawkins et al. The 2021 WHO classification of tumors of the central nervous system: A Summary. Neuro-Oncol. 23(8), 1231–1251 (2021). https://doi.org/10.1093/neuonc/noab106
N. Karavitaki, C. Brufani, J.T. Warner, C.B.T. Adams, P. Richards, O. Ansorge, B. Shine, H.E. Turner, J.A.H. Wass, Craniopharyngiomas in children and adults: systematic analysis of 121 cases with long-term follow-up. Clin. Endocrinol. 62(4), 397–409 (2005). https://doi.org/10.1111/j.1365-2265.2005.02231.x
M.J. Diaz, S.H. Kwak, K.T. Root, A. Fadil, A. Nguyen, L. Ladehoff, S. Batchu, B. Lucke-Wold, Current approaches to craniopharyngioma management. Front. Biosci. (Landmark Ed.) 27(12), 328 (2022). https://doi.org/10.31083/j.fbl2712328
E.M. Erfurth, Endocrine aspects and sequel in patients with craniopharyngioma. J. Pediatr. Endocrinol. Metab.: JPEM 28(1–2), 19–26 (2015). https://doi.org/10.1515/jpem-2014-0419
A.M. Pereira, E.M. Schmid, P.J. Schutte, J.H.C. Voormolen, N.R. Biermasz, S.W. van Thiel, E.P.M. Corssmit, J.W.A. Smit, F. Roelfsema, J.A. Romijn, High prevalence of long-term cardiovascular, neurological and psychosocial morbidity after treatment for craniopharyngioma. Clin. Endocrinol. 62(2), 197–204 (2005). https://doi.org/10.1111/j.1365-2265.2004.02196.x
A. Serbis, V.R. Tsinopoulou, A. Papadopoulou, S. Kolanis, E.I. Sakellari, K. Margaritis, E. Litou et al. Predictive factors for pediatric craniopharyngioma recurrence: an extensive narrative review. Diagnostics (Basel, Switz.) 13(9), 1588 (2023). https://doi.org/10.3390/diagnostics13091588
A.S. Sterkenburg, A. Hoffmann, U. Gebhardt, M. Warmuth-Metz, A.M.M. Daubenbüchel, H.L. Müller, Survival, hypothalamic obesity, and neuropsychological/psychosocial status after childhood-onset craniopharyngioma: newly reported long-term outcomes. Neuro-Oncol. 17(7), 1029–1038 (2015). https://doi.org/10.1093/neuonc/nov044
Z. Zhou, S. Zhang, F. Hu, Endocrine disorder in patients with craniopharyngioma. Front. Neurol. 12 (2021). https://www.frontiersin.org/articles/10.3389/fneur.2021.737743
Y. Guo, L. Pei, Y. Li, C. Li, S. Gui, M. Ni, P. Liu, Y. Zhang, L. Zhong, Characteristics and factors influencing hypothalamic pituitary dysfunction in patients with craniopharyngioma. Front. Endocrinol. 14, 1180591 (2023). https://doi.org/10.3389/fendo.2023.1180591
G. Patti, E. Calandra, A. De Bellis, A. Gallizia, M. Crocco, F. Napoli, A.M.E. Allegri et al. Antibodies against hypothalamus and pituitary gland in childhood-onset brain tumors and pituitary dysfunction. Front. Endocrinol. 11, 16 (2020). https://doi.org/10.3389/fendo.2020.00016
B. Ahmed, C.N. Bairey Merz, B.D. Johnson, V. Bittner, S.L. Berga, G.D. Braunstein, T.K. Hodgson et al. Diabetes mellitus, hypothalamic hypoestrogenemia, and coronary artery disease in premenopausal women (from the national heart, lung, and blood institute sponsored WISE study). Am. J. Cardiol. 102(2), 150–154 (2008). https://doi.org/10.1016/j.amjcard.2008.03.029
C.N. Bairey Merz, B.D. Johnson, B.L. Sharaf, V. Bittner, S.L. Berga, G.D. Braunstein, Hodgson et al. Hypoestrogenemia of hypothalamic origin and coronary artery disease in premenopausal women: A Report from the NHLBI-Sponsored WISE Study. J. Am. Coll. Cardiol. 41(3), 413–419 (2003). https://doi.org/10.1016/s0735-1097(02)02763-8
N.A. Tritos, S.L. Greenspan, D. King, A. Hamrahian, D.M. Cook, P.J. Jönsson, M.P. Wajnrajch, M. Koltowska-Häggstrom, B.M.K. Biller, Unreplaced sex steroid deficiency, corticotropin deficiency, and lower IGF-I are associated with lower bone mineral density in adults with growth hormone deficiency: A KIMS database analysis. J. Clin. Endocrinol. Metab. 96(5), 1516–1523 (2011). https://doi.org/10.1210/jc.2010-2662
H. Holmer, V. Popovic, B. Ekman, C. Follin, A.B. Siversson, E.M. Erfurth, Hypothalamic involvement and insufficient sex steroid supplementation are associated with low bone mineral density in women with childhood onset craniopharyngioma. Eur. J. Endocrinol. 165(1), 25–31 (2011). https://doi.org/10.1530/EJE-11-0229
D. Matthews, L. Bath, W. Högler, A. Mason, A. Smyth, M. Skae, Hormone supplementation for pubertal induction in girls. Arch. Dis. Child. 102(10), 975–980 (2017). https://doi.org/10.1136/archdischild-2016-311372
K.K. Miller, G. Sesmilo, A. Schiller, D. Schoenfeld, S. Burton, A. Klibanski, Androgen deficiency in women with hypopituitarism. J. Clin. Endocrinol. Metab. 86(2), 561–567 (2001). https://doi.org/10.1210/jcem.86.2.7246
C. Olivius, K. Landin-Wilhelmsen, D.S. Olsson, G. Johannsson, Å. Tivesten, Prevalence and treatment of central hypogonadism and hypoandrogenism in women with hypopituitarism. Pituitary 21(5), 445–453 (2018). https://doi.org/10.1007/s11102-018-0895-1
N. Pluchino, A. Carmignani, A. Cubeddu, A. Santoro, V. Cela, T. Errasti, Androgen therapy in women: For whom and when. Arch. Gynecol. Obstet. 288(4), 731–737 (2013). https://doi.org/10.1007/s00404-013-2969-7
I. Ilovayskaya, V. Zektser, L. Lazebnik, Similarity of female central (hypogonadotropic) hypogonadism and postmenopause. Climacteric: J. Int. Menopause Soc. 20(4), 356–361 (2017). https://doi.org/10.1080/13697137.2017.1315086
K.K. Miller, B.M.K. Biller, C. Beauregard, J.G. Lipman, J. Jones, D. Schoenfeld, J.C. Sherman, B. Swearingen, J. Loeffler, A. Klibanski, Effects of testosterone replacement in androgen-deficient women with hypopituitarism: A randomized, double-blind, placebo-controlled study. J. Clin. Endocrinol. Metab. 91(5), 1683–1690 (2006). https://doi.org/10.1210/jc.2005-2596
H. Zang, S.R. Davis, Androgen replacement therapy in androgen-deficient women with hypopituitarism. Drugs 68(15), 2085–2093 (2008). https://doi.org/10.2165/00003495-200868150-00001
T. Tsilchorozidou, G.S. Conway, Uterus size and ovarian morphology in women with isolated growth hormone deficiency, hypogonadotrophic hypogonadism and hypopituitarism. Clin. Endocrinol. 61(5), 567–572 (2004). https://doi.org/10.1111/j.1365-2265.2004.02126.x
H. Okinaga, A. Matsuno, R. Okazaki, High risk of osteopenia and bone derangement in postsurgical patients with craniopharyngiomas, pituitary adenomas and other parasellar lesions. Endocr. J. 52(6), 751–756 (2005). https://doi.org/10.1507/endocrj.52.751
N.A. Tritos, A.H. Hamrahian, D. King, S.L. Greenspan, D.M. Cook, P.J. Jönsson, M.P. Wajnrajch, M. Koltowska-Häggstrom, B.M.K. Biller, A longer interval without gh replacement and female gender are associated with lower bone mineral density in adults with childhood-onset GH deficiency: A KIMS database analysis. Eur. J. Endocrinol. 167(3), 343–351 (2012). https://doi.org/10.1530/EJE-12-0070
P. Dimitri, Treatment of acquired hypothalamic obesity: Now and the future. Front. Endocrinol. 13, 846880 (2022). https://doi.org/10.3389/fendo.2022.846880
M. Hage, O. Plesa, I. Lemaire, M.L. Raffin Sanson, Estrogen and progesterone therapy and meningiomas. Endocrinology 163(2), bqab259 (2022). https://doi.org/10.1210/endocr/bqab259
A. Martínez-Ortega, Á. Flores-Martinez, E. Venegas-Moreno, E. Dios, D. Del Can, E. Rivas, A. Kaen et al. Sex hormone receptor expression in craniopharyngiomas and association with tumor aggressiveness characteristics. J. Clin. Med. 11(1), 281 (2022). https://doi.org/10.3390/jcm11010281
M. Fleseriu, I.A. Hashim, N. Karavitaki, S. Melmed, M.H. Murad, R. Salvatori, M.H. Samuels, Hormonal replacement in hypopituitarism in adults: an endocrine society clinical practice guideline. J. Clin. Endocrinol. Metab. 101(11), 3888–3921 (2016). https://doi.org/10.1210/jc.2016-2118
P.M. Mah, J. Webster, P. Jönsson, U. Feldt-Rasmussen, M. Koltowska-Häggström, R.J.M. Ross, Estrogen replacement in women of fertile years with hypopituitarism. J. Clin. Endocrinol. Metab. 90(11), 5964–5969 (2005). https://doi.org/10.1210/jc.2005-1207
N. Phelan, S.H. Conway, S. Llahana, G.S. Conway, Quantification of the adverse effect of ethinylestradiol containing oral contraceptive pills when used in conjunction with growth hormone replacement in routine practice. Clin. Endocrinol. 76(5), 729–733 (2012). https://doi.org/10.1111/j.1365-2265.2011.04277.x
M.V. Dragoman, The combined oral contraceptive pill- recent developments, risks and benefits. Best. Pract. Res. Clin. Obstet. Gynaecol. 28(6), 825–834 (2014). https://doi.org/10.1016/j.bpobgyn.2014.06.003
V. Birzniece, K.K.Y. Ho, Growth and development: patching up a better pill for GH-deficient women. Nat. Rev. Endocrinol. 8(4), 197–198 (2012). https://doi.org/10.1038/nrendo.2012.9
N. Shoung, K.K.Y. Ho, Managing estrogen therapy in the pituitary patient. J. Endocr. Soc. 7(5), bvad051 (2023). https://doi.org/10.1210/jendso/bvad051
S. Khosla, D.G. Monroe, Regulation of bone metabolism by sex steroids. Cold Spring Harb. Perspect. Med. 8(1), a031211 (2018). https://doi.org/10.1101/cshperspect.a031211
S.S. van Santen, D.S. Olsson, M.M. van den Heuvel-Eibrink, M. Wijnen, C. Hammarstrand, J.A.M.J.L. Janssen, G. Johansson, A.J. van der Lely, S.J.C.M.M. Neggers, Body composition and bone mineral density in craniopharyngioma patients: A longitudinal study over 10 years. J. Clin. Endocrinol. Metab. 105(12), dgaa607 (2020). https://doi.org/10.1210/clinem/dgaa607
S.S. van Santen, D.S. Olsson, M.M. van den Heuvel-Eibrink, M. Wijnen, C. Hammarstrand, J.A.M.J.L. Janssen, G. Johansson, A.J. van der Lely, S.J.C.M.M. Neggers, Fractures, bone mineral density, and final height in craniopharyngioma patients with a follow-up of 16 years. J. Clin. Endocrinol. Metab. 105(4), e1397–e1407 (2020). https://doi.org/10.1210/clinem/dgz279
E. Burt, M.C. Davies, E. Yasmin, A. Cameron-Pimblett, D. Mavrelos, V. Talaulikar, G.S. Conway, Reduced uterine volume after induction of puberty in women with hypogonadism. Clin. Endocrinol. 91(6), 798–804 (2019). https://doi.org/10.1111/cen.14092
D. Lin, Y. Yang, Z. Zhou, Natural pregnancy after gross total resection of a large craniopharyngioma with hypothalamic involvement: a case report. Acta. Neurol. Belg. 122(6), 1641–1644 (2022).
P. Sowithayasakul, S. Boekhoff, B. Bison, HL. Müller, Pregnancies after Childhood Craniopharyngioma: Results of KRANIOPHARYNGEOM 2000/2007 and Review of the Literature. Neuroendocrinol 111(1-2), 16–26 (2021).
M. Hayashi, K. Tomobe, K. Hoshimoto, T.Ohkura, Successful pregnancy following gonadotropin therapy in a patient with hypogonadotropic hypogonadism resulting from craniopharyngioma. Int. J. Clin. Pract. 56(2), 149–151 (2002).
Acknowledgements
This is a short text to acknowledge the contributions of specific colleagues, institutions, or agencies that aided the efforts of the authors.
Author contributions
II and VZ curated the data, performed the investigation and methodology of the study. VZ performed formal analysis and statistical analysis using the software. II was responsible for conceptualization, supervision, validation of the methodology. II and VZ were writing the paper, II performed a full review and editing. Both authors contributed to the article and approved the submitted version.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethics approval
Authors of research confirm that the study was approved by the local appropriate institutional State Budget Health Agency Moscow Region Moscow Regional Research Clinical Institute, Moscow, RU research ethics committee (the protocol number 16, 28.10.2021).
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent to publish
Our manuscript doesn’t contain any individual person’s data in any form (including any individual details, images or videos).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ilovayskaya, I., Zektser, V. Hypogonadism as a consequence of craniopharyngioma in female patients: comparison of childhood and adult onset and effects of estrogen replacement therapy. Endocrine (2024). https://doi.org/10.1007/s12020-024-03872-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12020-024-03872-7