Abstract
Purpose
To investigate the effect of TFF3 in the pathogenesis of Diabetic Kidney Disease (DKD), and explore the dynamic changes of TFF3 expression pattern in renal injury process.
Methods
DKD animal model was established by streptozotocin (STZ) (40 mg/kg/d, ip, for 5 days, consecutively) combined with the high fat diet (HFD) for 12 weeks. While animals were sacrificed at different time stages in DKD process (4 weeks, 8 weeks and 12 weeks, respectively).
Results
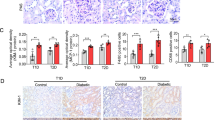
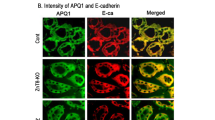
STZ combined with high-fat diet induced weight gain, increased blood glucose and decreased glucose tolerance in DKD mice. Compared to the control group, the DKD group exhibits extracellular matrix (ECM) accumulation and the renal injury was aggravated in a time-dependent manner. The TFF3 expression level was decreased in kidney, and increased in colon tissue.
Conclusion
TFF3 is not only expressed in colon, but also expressed in renal medulla and cortex. TFF3 might be play a pivotal role in renal mucosal repair by gut-kidney crosstalk, and protect renal from high glucose microenvironment damage.







Similar content being viewed by others
References
H. Sun, P. Saeedi, S. Karuranga et al. IDF diabetes atlas: global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabetes Res. Clin. Pr. 183, 109–119 (2022). https://doi.org/10.1016/j.diabres.2021.109119
C. Lin, Y. Hsu, Y. Huang, Y. Shih, C. Wang, W. Chiang, P. Chang, A KDM6A-KLF10 reinforcing feedback mechanism aggravates diabetic podocyte dysfunction. EMBO Mol. Med. 11(5), (2019). https://doi.org/10.15252/emmm.201809828
C.J. May, G.I. Welsh, M. Chesor, P.J. Lait, L.P. Schewitz-Bowers, R.W.J. Lee, M.A. Saleem, Human Th17 cells produce a soluble mediator that increases podocyte motility via signaling pathways that mimic PAR-1 activation. Am. J. Physiol. Ren. Physiol. 317(4), F913–F921 (2019). https://doi.org/10.1152/ajprenal.00093.2019
Y. Li, Y. Lu, D. Tang et al. Anthocyanin improves kidney function in diabetic kidney disease by regulating amino acid metabolism. J. Transl. Med. 20(1), 510 (2022). https://doi.org/10.1186/s12967-022-03717-9
Y. Yang, Z. Lin, Q. Lin, W. Bei, J. Guo, Pathological and therapeutic roles of bioactive peptide trefoil factor 3 in diverse diseases: recent progress and perspective. Cell Death Dis. 13(1), 62 (2022). https://doi.org/10.1038/s41419-022-04504-6
L. Thim, F. May, Structure of mammalian trefoil factors and functional insights. Cell Mol. Life Sci. 62(24), 2956–2973 (2005). https://doi.org/10.1007/s00018-005-5484-6
X. Wu, H. Zheng, R. Yang et al. Mouse trefoil factor 3 ameliorated high-fat-diet-induced hepatic steatosis via increasing peroxisome proliferator-activated receptor-alpha-mediated fatty acid oxidation. Am. J. Physiol. Endocrinol. Metab. 317(3), E436–E445 (2019). https://doi.org/10.1152/ajpendo.00454.2018
J. Zou, Z. Chen, C. Liang et al. Trefoil Factor 3, cholinesterase and homocysteine: potential predictors for parkinson’s disease dementia and vascular parkinsonism dementia in advanced stage. Aging Dis. 9(1), 51–65 (2018). https://doi.org/10.14336/AD.2017.0416
D. Taupin, J. Pedersen, M. Familari, G. Cook, N. Yeomans, A.S. Giraud, Augmented intestinal trefoil factor (TFF3) and loss of pS2 (TFF1) expression precedes metaplastic differentiation of gastric epithelium. Lab. Investig. 81(3), 397–408 (2001). https://doi.org/10.1038/labinvest.3780247
N.M. Belle, Y. Ji, K. Herbine et al. TFF3 interacts with LINGO2 to regulate EGFR activation for protection against colitis and gastrointestinal helminths. Nat. Commun. 10(1), 4408 (2019). https://doi.org/10.1038/s41467-019-12315-1
H. Cui, S. Wang, F. Song et al. CD147 receptor is essential for TFF3-mediated signaling regulating colorectal cancer progression. Signal Transduct. Target Ther. 6(1), 268 (2021). https://doi.org/10.1038/s41392-021-00677-2
T. Du, H. Luo, H. Qin, F. Wang, Q. Wang, Y. Xiang, Y. Zhang, Circulating Serum Trefoil Factor 3 (TFF3) is dramatically increased in chronic kidney disease. PLoS One 8(11), e80271 (2013). https://doi.org/10.1371/journal.pone.0080271
B.R. Griffin, S. Faubel, C.L. Edelstein, Biomarkers of drug-induced kidney toxicity. Ther. Drug Monit. 41(2), 213–226 (2019). https://doi.org/10.1097/FTD.0000000000000589
M.E. Grams, A. Surapaneni, J. Chen et al. Proteins associated with risk of kidney function decline in the general population. J. Am. Soc. Nephrol. 32(9), 2291–2302 (2021). https://doi.org/10.1681/ASN.2020111607
S. Anand, M. Bajpai, T. Khanna, A. Kumar, Influence of genetic polymorphism in renin-angiotensin system-candidate genes on urinary trefoil family factor 3 levels in children with congenital anomalies of kidney and urinary tract. Pediatr. Nephrol. 37(1), 139–145 (2022). https://doi.org/10.1007/s00467-021-05160-2
S. Anand, M. Bajpai, T. Khanna, A. Kumar, Urinary biomarkers as point-of-care tests for predicting progressive deterioration of kidney function in congenital anomalies of kidney and urinary tract: trefoil family factors (TFFs) as the emerging biomarkers. Pediatr. Nephrol. 36(6), 1465–1472 (2021). https://doi.org/10.1007/s00467-020-04841-8
Y. Yang, H. Tan, X. Zhang et al. The Chinese medicine Fufang Zhenzhu Tiaozhi capsule protects against renal injury and inflammation in mice with diabetic kidney disease. J. Ethnopharmacol. 292, 115–165 (2022). https://doi.org/10.1016/j.jep.2022.115165
P.M. Titchenell, Q. Chu, B.R. Monks, M.J. Birnbaum, Hepatic insulin signalling is dispensable for suppression of glucose output by insulin in vivo. Nat. Commun. 6, 70–78 (2015). https://doi.org/10.1038/ncomms8078
L. Xu, X. Li, F. Zhang, L. Wu, Z. Dong, D. Zhang, EGFR drives the progression of AKI to CKD through HIPK2 overexpression. Theranostics 9(9), 2712–2726 (2019). https://doi.org/10.7150/thno.31424
S. Li, J. Park, Y. Guan, K. Chung, R. Shrestha, M.B. Palmer, K. Susztak, DNMT1 in Six2 progenitor cells is essential for transposable element silencing and kidney development. J. Am. Soc. Nephrol. 30(4), 594–609 (2019). https://doi.org/10.1681/ASN.2018070687
L. Marko, E. Vigolo, C. Hinze et al. Tubular Epithelial NF-kappa B activity regulates Ischemic AKI. J. Am. Soc. Nephrol. 27(9), 2658–2669 (2016). https://doi.org/10.1681/ASN.2015070748
J. Ren, L. Han, J. Tang et al. Foxp1 is critical for the maintenance of regulatory T-cell homeostasis and suppressive function. PLoS Biol. 17(5), e3000270 (2019). https://doi.org/10.1371/journal.pbio.3000270
Y. Liu, K. Wang, X. Liang et al. Complement C3 produced by macrophages promotes renal fibrosis via IL-17A secretion. Front. Immunol. 9, 02385 (2018). https://doi.org/10.3389/fimmu.2018.02385
L. Liu, T. Tao, S. Liu et al. An RFC4/Notch1 signaling feedback loop promotes NSCLC metastasis and stemness. Nat. Commun. 12(1), 2693 (2021). https://doi.org/10.1038/s41467-021-22971-x
U. Erben, C. Loddenkemper, K. Doerfel et al. A guide to histomorphological evaluation of intestinal inflammation in mouse models. Int J. Clin. Exp. Pathol. 7(8), 4557–4576 (2014)
T. Yamanari, H. Sugiyama, K. Tanaka et al. Urine trefoil factors as prognostic biomarkers in chronic kidney disease. Biomed. Res. Int. 2018, 3024698 (2018). https://doi.org/10.1155/2018/3024698
K. Tanaka, H. Sugiyama, T. Yamanari et al. Renal expression of trefoil factor 3 mRNA in association with tubulointerstitial fibrosis in IgA nephropathy. Nephrology 23(9), 855–862 (2018). https://doi.org/10.1111/nep.13444
S. Khummuang, W. Phanphrom, W. Laopajon, W. Kasinrerk, P .Chaiyarit, S. Pata, Production of monoclonal antibodies against human trefoil factor 3 and development of a modified-sandwich ELISA for detection of trefoil factor 3 homodimer in saliva. Biol. Proced. Online. 19(14), (2017). https://doi.org/10.1186/s12575-017-0064-3
Y. Wang, Y. Liang, W. Zhao, G. Fu, Q. Li, X. Min, Y. Guo, Circulating miRNA-21 as a diagnostic biomarker in elderly patients with type 2 cardiorenal syndrome. Sci. Rep. 10(1), 4894 (2020). https://doi.org/10.1038/s41598-020-61836-z
R. Jahan, A. Shah, S.G. Kisling, M.A. Macha, S. Thayer, S.K. Batra, S. Kaur, Odyssey of trefoil factors in cancer: diagnostic and therapeutic implications. Biochim. Biophys. Acta Rev. Cancer 1873(2), 188362 (2020). https://doi.org/10.1016/j.bbcan.2020.188362
F.E.B. May, B.R. Westley, TFF3 is a valuable predictive biomarker of endocrine response in metastatic breast cancer. Endocr. Relat. Cancer 22(3), 465–479 (2015). https://doi.org/10.1530/ERC-15-0129
Y. Yu, H. Jin, D. Holder et al. Urinary biomarkers trefoil factor 3 and albumin enable early detection of kidney tubular injury. Nat. Biotechnol. 28(5), 470–477 (2010). https://doi.org/10.1038/nbt.1624
Y. Xue, L. Shen, Y. Cui et al. Tff3, as a novel peptide, regulates hepatic glucose metabolism. PloS One 8(9), e75240 (2013). https://doi.org/10.1371/journal.pone.0075240
K. Krueger, S. Schmid, F. Paulsen et al. Trefoil Factor 3 (TFF3) is involved in cell migration for skeletal repair. Int J. Mol. Sci. 20(17), 4277 (2019). https://doi.org/10.3390/ijms20174277
B.C. Astor, A. Koettgen, S. Hwang, N.A. Bhavsar, C.S. Fox, J. Coresh, Trefoil Factor 3 predicts incident chronic kidney disease: a case-control study nested within the Atherosclerosis Risk in Communities (ARIC) study. Am. J. Nephrol. 34(4), 291–297 (2011). https://doi.org/10.1159/000330699
J. Guo, M. Sun, X. Teng, L. Xu, MicroRNA-7-5p regulates the expression of TFF3 in inflammatory bowel disease. Mol. Med. Rep. 16(2), 1200–1206 (2017). https://doi.org/10.3892/mmr.2017.6730
J. Liu, S.Y. Kim, S. Shin et al. Overexpression of TFF3 is involved in prostate carcinogenesis via blocking mitochondria-mediated apoptosis. Exp. Mol. Med 50(8), 1–11 (2018). https://doi.org/10.1038/s12276-018-0137-7
Y. Zhu, S. Zhao, Y. Deng et al. Hepatic GALE regulates whole-body glucose homeostasis by modulating Tff3 expression. Diabetes 66(11), 2789–2799 (2017). https://doi.org/10.2337/db17-0323
Author contributions:
T.Z.: Writing, original draft and preparation. Y.H.Z.: Drawing and drafting; J.T.: Formal analysis, Software. X.L.R.: review, editing and Supervision. Y.Q.Y.: Writing, review & editing, Supervision, Conceptualization and Project administration.
Funding
This work was supported by Key Project of National Natural Science Foundation of China (82204704); Basic and Applied Basic Research Special Project, Guangzhou Science and Technology Bureau (SL2024A04J00581).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethics approval
All animal experiments were approved by the Animal Experiment Ethics Committee of Guangdong Pharmaceutical University (gdpulac2019180).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
These authors contributed equally: Tao Zhang, Yinghui Zhang
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, T., Zhang, Y., Tao, J. et al. Intestinal Trefoil Factor 3: a new biological factor mediating gut-kidney crosstalk in diabetic kidney disease. Endocrine 84, 109–118 (2024). https://doi.org/10.1007/s12020-023-03559-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-023-03559-5