Abstract
Diabetes mellitus (DM) is a chronic and relentlessly progressive metabolic disease characterized by a relative or absolute deficiency of insulin in the body, leading to increased production of advanced glycosylation end products that further enhance oxidative and nitrosative stresses, often leading to multiple macrovascular (cardiovascular disease) and microvascular (e.g., diabetic nephropathy, diabetic retinopathy, and neuropathy) complications, representing the ninth leading cause of death worldwide. Existing medical treatments do not provide a complete cure for DM; thus, stem cell transplantation therapy has become the focus of research on DM and its complications. Urine-derived stem cells (USCs), which are isolated from fresh urine and have biological properties similar to those of mesenchymal stem cells (MSCs), were demonstrated to exert antiapoptotic, antifibrotic, anti-inflammatory, and proangiogenic effects through direct differentiation or paracrine mechanisms and potentially treat patients with DM. USCs also have the advantages of simple noninvasive sample collection procedures, minimal ethical issues, low cost, and easy cell isolation methods and thus have received more attention in regenerative therapies in recent years. This review outlines the biological properties of USCs and the research progress and current limitations of their role in DM and related complications. In summary, USCs have shown good versatility in treating hyperglycemia-impaired target organs in preclinical models, and many challenges remain in translating USC therapies to the clinic.


Similar content being viewed by others
Abbreviations
- DM:
-
Diabetes mellitus
- USCs:
-
Urine-derived stem cells
- MSCs:
-
Mesenchymal stem cells
- T1DM:
-
Type 1 diabetes mellitus
- T2DM:
-
Type 2 diabetes mellitus
- SC:
-
Stem cell
- IPCs:
-
Insulin-producing cells
- iPSCs:
-
Induced pluripotent stem cells
- ECM:
-
Extracellular matrix
- EVs:
-
Extracellular vesicles
- hUSCs:
-
Human USCs
- USC-Exos:
-
USC-derived exosomes
- BMSCs:
-
Bone mesenchymal stem cells
- DCM:
-
Diabetic cardiomyopathy
- DED:
-
Diabetic erectile dysfunction
- DOP:
-
Diabetic osteoporosis
- ESRD:
-
End-stage renal disease
- miRNAs:
-
MicroRNAs
- d-USC:
-
USCs from patients with DN
- CCs:
-
Corpora cavernosa
- LV:
-
Left ventricular
- DR:
-
Diabetic retinopathy
References
H. Sun, P. Saeedi, S. Karuranga, M. Pinkepank, K. Ogurtsova, B.B. Duncan, C. Stein, A. Basit, J.C.N. Chan, J.C. Mbanya, M.E. Pavkov, A. Ramachandaran, S.H. Wild, S. James, W.H. Herman, P. Zhang, C. Bommer, S. Kuo, E.J. Boyko, D.J. Magliano, IDF Diabetes Atlas: Global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabetes Res. Clin. Pract. 183, 109–119 (2022). https://doi.org/10.1016/j.diabres.2021.109119
A.M. Shapiro, J.R. Lakey, E.A. Ryan, G.S. Korbutt, E. Toth, G.L. Warnock, N.M. Kneteman, R.V. Rajotte, Islet transplantation in seven patients with type 1 diabetes mellitus using a glucocorticoid-free immunosuppressive regimen. N. Engl. J. Med. 343(4), 230–238 (2000). https://doi.org/10.1056/nejm200007273430401
M.R. Rickels, S.M. Kong, C. Fuller, C. Dalton-Bakes, J.F. Ferguson, M.P. Reilly, K.L. Teff, A. Naji, Improvement in insulin sensitivity after human islet transplantation for type 1 diabetes. J. Clin. Endocrinol. Metab. 98(11), E1780–1785 (2013). https://doi.org/10.1210/jc.2013-1764
M.R. Rickels, C. Fuller, C. Dalton-Bakes, E. Markmann, M. Palanjian, K. Cullison, J. Tiao, S. Kapoor, C. Liu, A. Naji, K.L. Teff, Restoration of glucose counterregulation by islet transplantation in long-standing type 1 diabetes. Diabetes 64(5), 1713–1718 (2015). https://doi.org/10.2337/db14-1620
B.J. Hering, W.R. Clarke, N.D. Bridges, T.L. Eggerman, R. Alejandro, M.D. Bellin, K. Chaloner, C.W. Czarniecki, J.S. Goldstein, L.G. Hunsicker, D.B. Kaufman, O. Korsgren, C.P. Larsen, X. Luo, J.F. Markmann, A. Naji, J. Oberholzer, A.M. Posselt, M.R. Rickels, C. Ricordi, M.A. Robien, P.A. Senior, A.M. Shapiro, P.G. Stock, N.A. Turgeon, Phase 3 trial of transplantation of human islets in type 1 diabetes complicated by severe hypoglycemia. Diabetes Care 39(7), 1230–1240 (2016). https://doi.org/10.2337/dc15-1988
J.F. Markmann, M.R. Rickels, T.L. Eggerman, N.D. Bridges, D.E. Lafontant, J. Qidwai, E. Foster, W.R. Clarke, M. Kamoun, R. Alejandro, M.D. Bellin, K. Chaloner, C.W. Czarniecki, J.S. Goldstein, B.J. Hering, L.G. Hunsicker, D.B. Kaufman, O. Korsgren, C.P. Larsen, X. Luo, A. Naji, J. Oberholzer, A.M. Posselt, C. Ricordi, P.A. Senior, A.M.J. Shapiro, P.G. Stock, N.A. Turgeon, Phase 3 trial of human islet-after-kidney transplantation in type 1 diabetes. Am. J. Transplant. 21(4), 1477–1492 (2021). https://doi.org/10.1111/ajt.16174
Y.F. Smets, R.G. Westendorp, J.W. van der Pijl, F.T. de Charro, J. Ringers, J.W. de Fijter, H.H. Lemkes, Effect of simultaneous pancreas-kidney transplantation on mortality of patients with type-1 diabetes mellitus and end-stage renal failure. Lancet 353(9168), 1915–1919 (1999). https://doi.org/10.1016/s0140-6736(98)07513-8
B.N. Becker, P.C. Brazy, Y.T. Becker, J.S. Odorico, T.J. Pintar, B.H. Collins, J.D. Pirsch, G.E. Leverson, D.M. Heisey, H.W. Sollinger, Simultaneous pancreas-kidney transplantation reduces excess mortality in type 1 diabetic patients with end-stage renal disease. Kidney Int. 57(5), 2129–2135 (2000). https://doi.org/10.1046/j.1523-1755.2000.00064.x
R. Giannarelli, A. Coppelli, M.S. Sartini, M. Del Chiaro, F. Vistoli, G. Rizzo, M. Barsotti, S. Del Prato, F. Mosca, U. Boggi, P. Marchetti, Pancreas transplant alone has beneficial effects on retinopathy in type 1 diabetic patients. Diabetologia 49(12), 2977–2982 (2006). https://doi.org/10.1007/s00125-006-0463-5
R.P. Pelletier, A.A. Rajab, A. Diez, N.R. DiPaola, G.L. Bumgardner, E.A. Elkhammas, M.L. Henry, Early immunosuppression treatment correlates with later de novo donor-specific antibody development after kidney and pancreas transplantation. Clin. Transplant. 29(12), 1119–1127 (2015). https://doi.org/10.1111/ctr.12636
F. Vendrame, Y.Y. Hopfner, S. Diamantopoulos, S.K. Virdi, G. Allende, I.V. Snowhite, H.K. Reijonen, L. Chen, P. Ruiz, G. Ciancio, J.C. Hutton, S. Messinger, G.W. Burke 3rd, A. Pugliese, Risk factors for type 1 diabetes recurrence in immunosuppressed recipients of simultaneous pancreas-kidney transplants. Am. J. Transplant. 16(1), 235–245 (2016). https://doi.org/10.1111/ajt.13426
Y. Zhang, E. McNeill, H. Tian, S. Soker, K.E. Andersson, J.J. Yoo, A. Atala, Urine derived cells are a potential source for urological tissue reconstruction. J. Urol. 180(5), 2226–2233 (2008). https://doi.org/10.1016/j.juro.2008.07.023
S. Bharadwaj, G. Liu, Y. Shi, R. Wu, B. Yang, T. He, Y. Fan, X. Lu, X. Zhou, H. Liu, A. Atala, J. Rohozinski, Y. Zhang, Multipotential differentiation of human urine-derived stem cells: potential for therapeutic applications in urology. Stem Cells 31(9), 1840–1856 (2013). https://doi.org/10.1002/stem.1424
H.S. Kang, S.H. Choi, B.S. Kim, J.Y. Choi, G.B. Park, T.G. Kwon, S.Y. Chun, Advanced properties of urine derived stem cells compared to adipose tissue derived stem cells in terms of cell proliferation, immune modulation and multi differentiation. J. Korean Med. Sci. 30(12), 1764–1776 (2015). https://doi.org/10.3346/jkms.2015.30.12.1764
D. Zhang, G. Wei, P. Li, X. Zhou, Y. Zhang, Urine-derived stem cells: a novel and versatile progenitor source for cell-based therapy and regenerative medicine. Genes Dis. 1(1), 8–17 (2014). https://doi.org/10.1016/j.gendis.2014.07.001
C. Wu, L. Chen, Y.Z. Huang, Y. Huang, O. Parolini, Q. Zhong, X. Tian, L. Deng, Comparison of the Proliferation and Differentiation Potential of Human Urine-, Placenta Decidua Basalis-, and Bone Marrow-Derived Stem Cells. Stem Cells Int 2018, 7131532 (2018). https://doi.org/10.1155/2018/7131532
A.J. Chen, J.K. Pi, J.G. Hu, Y.Z. Huang, H.W. Gao, S.F. Li, J. Li-Ling, H.Q. Xie, Identification and characterization of two morphologically distinct stem cell subpopulations from human urine samples. Sci. China Life Sci. 63(5), 712–723 (2020). https://doi.org/10.1007/s11427-018-9543-1
J. Zhou, X. Wang, S. Zhang, Y. Gu, L. Yu, J. Wu, T. Gao, F. Chen, Generation and characterization of human cryptorchid-specific induced pluripotent stem cells from urine. Stem Cells Dev. 22(5), 717–725 (2013). https://doi.org/10.1089/scd.2012.0260
M. Culenova, A. Nicodemou, Z.V. Novakova, M. Debreova, V. Smolinská, S. Bernatova, D. Ivanisova, O. Novotna, J. Vasicek, I. Varga, S. Ziaran, L. Danisovic, Isolation, culture and comprehensive characterization of biological properties of human urine-derived stem cells. Int. J. Mol. Sci. 22(22), 12503 (2021). https://doi.org/10.3390/ijms222212503
J.J. Guan, X. Niu, F.X. Gong, B. Hu, S.C. Guo, Y.L. Lou, C.Q. Zhang, Z.F. Deng, Y. Wang, Biological characteristics of human-urine-derived stem cells: potential for cell-based therapy in neurology. Tissue Eng. A 20(13-14), 1794–1806 (2014). https://doi.org/10.1089/ten.TEA.2013.0584
G. Liu, R.A. Pareta, R. Wu, Y. Shi, X. Zhou, H. Liu, C. Deng, X. Sun, A. Atala, E.C. Opara, Y. Zhang, Skeletal myogenic differentiation of urine-derived stem cells and angiogenesis using microbeads loaded with growth factors. Biomaterials 34(4), 1311–1326 (2013). https://doi.org/10.1016/j.biomaterials.2012.10.038
B. Ouyang, X. Sun, D. Han, S. Chen, B. Yao, Y. Gao, J. Bian, Y. Huang, Y. Zhang, Z. Wan, B. Yang, H. Xiao, Z. Songyang, G. Liu, Y. Zhang, C. Deng, Human urine-derived stem cells alone or genetically-modified with FGF2 Improve type 2 diabetic erectile dysfunction in a rat model. PLoS One 9(3), e92825 (2014). https://doi.org/10.1371/journal.pone.0092825
E. Lazzeri, E. Ronconi, M.L. Angelotti, A. Peired, B. Mazzinghi, F. Becherucci, S. Conti, G. Sansavini, A. Sisti, F. Ravaglia, D. Lombardi, A. Provenzano, A. Manonelles, J.M. Cruzado, S. Giglio, R.M. Roperto, M. Materassi, L. Lasagni, P. Romagnani, Human urine-derived renal progenitors for personalized modeling of genetic kidney disorders. J. Am. Soc. Nephrol. 26(8), 1961–1974 (2015). https://doi.org/10.1681/asn.2014010057
J.Y. Choi, S.Y. Chun, Y.S. Ha, D.H. Kim, J. Kim, P.H. Song, H.T. Kim, E.S. Yoo, B.S. Kim, T.G. Kwon, Potency of human urine-derived stem cells for renal lineage differentiation. Tissue Eng. Regen. Med. 14(6), 775–785 (2017). https://doi.org/10.1007/s13770-017-0081-y
Y. Hwang, S.H. Cha, Y. Hong, A.R. Jung, H.S. Jun, Direct differentiation of insulin-producing cells from human urine-derived stem cells. Int. J. Med. Sci. 16(12), 1668–1676 (2019). https://doi.org/10.7150/ijms.36011
G. Sun, B. Ding, M. Wan, L. Chen, J. Jackson, A. Atala, Formation and optimization of three-dimensional organoids generated from urine-derived stem cells for renal function in vitro. Stem Cell Res. Ther. 11(1), 309 (2020). https://doi.org/10.1186/s13287-020-01822-4
M. Zhou, L. Shen, Y. Qiao, Z. Sun, Inducing differentiation of human urine-derived stem cells into hepatocyte-like cells by coculturing with human hepatocyte L02 cells. J. Cell. Biochem. 121(1), 566–573 (2020). https://doi.org/10.1002/jcb.29301
D. Jafari, S. Malih, S.S. Eslami, R. Jafari, L. Darzi, P. Tarighi, A. Samadikuchaksaraei, The relationship between molecular content of mesenchymal stem cells derived exosomes and their potentials: Opening the way for exosomes based therapeutics. Biochimie 165, 76–89 (2019). https://doi.org/10.1016/j.biochi.2019.07.009
X. Dong, T. Zhang, Q. Liu, J. Zhu, J. Zhao, J. Li, B. Sun, G. Ding, X. Hu, Z. Yang, Y. Zhang, L. Li, Beneficial effects of urine-derived stem cells on fibrosis and apoptosis of myocardial, glomerular and bladder cells. Mol. Cell. Endocrinol. 427, 21–32 (2016). https://doi.org/10.1016/j.mce.2016.03.001
C.Y. Chen, S.S. Rao, L. Ren, X.K. Hu, Y.J. Tan, Y. Hu, J. Luo, Y.W. Liu, H. Yin, J. Huang, J. Cao, Z.X. Wang, Z.Z. Liu, H.M. Liu, S.Y. Tang, R. Xu, H. Xie, Exosomal DMBT1 from human urine-derived stem cells facilitates diabetic wound repair by promoting angiogenesis. Theranostics 8(6), 1607–1623 (2018). https://doi.org/10.7150/thno.22958
X. Li, J. Liao, X. Su, W. Li, Z. Bi, J. Wang, Q. Su, H. Huang, Y. Wei, Y. Gao, J. Li, L. Liu, C. Wang, Human urine-derived stem cells protect against renal ischemia/reperfusion injury in a rat model via exosomal miR-146a-5p which targets IRAK1. Theranostics 10(21), 9561–9578 (2020). https://doi.org/10.7150/thno.42153
K.C. Herold, D.A. Vignali, A. Cooke, J.A. Bluestone, Type 1 diabetes: translating mechanistic observations into effective clinical outcomes. Nat. Rev. Immunol. 13(4), 243–256 (2013). https://doi.org/10.1038/nri3422
C.J. Nolan, P. Damm, M. Prentki, Type 2 diabetes across generations: from pathophysiology to prevention and management. Lancet 378(9786), 169–181 (2011). https://doi.org/10.1016/s0140-6736(11)60614-4
Y. Yang, L. Chan, Monogenic diabetes: what it teaches us on the common forms of type 1 and type 2 diabetes. Endocr. Rev. 37(3), 190–222 (2016). https://doi.org/10.1210/er.2015-1116
K.A. D’Amour, A.G. Bang, S. Eliazer, O.G. Kelly, A.D. Agulnick, N.G. Smart, M.A. Moorman, E. Kroon, M.K. Carpenter, E.E. Baetge, Production of pancreatic hormone-expressing endocrine cells from human embryonic stem cells. Nat. Biotechnol. 24(11), 1392–1401 (2006). https://doi.org/10.1038/nbt1259
D. Zhang, W. Jiang, M. Liu, X. Sui, X. Yin, S. Chen, Y. Shi, H. Deng, Highly efficient differentiation of human ES cells and iPS cells into mature pancreatic insulin-producing cells. Cell Res. 19(4), 429–438 (2009). https://doi.org/10.1038/cr.2009.28
S. Hrvatin, C.W. O’Donnell, F. Deng, J.R. Millman, F.W. Pagliuca, P. DiIorio, A. Rezania, D.K. Gifford, D.A. Melton, Differentiated human stem cells resemble fetal, not adult, β cells. Proc. Natl Acad. Sci. USA 111(8), 3038–3043 (2014). https://doi.org/10.1073/pnas.1400709111
A. Rezania, J.E. Bruin, P. Arora, A. Rubin, I. Batushansky, A. Asadi, S. O’Dwyer, N. Quiskamp, M. Mojibian, T. Albrecht, Y.H. Yang, J.D. Johnson, T.J. Kieffer, Reversal of diabetes with insulin-producing cells derived in vitro from human pluripotent stem cells. Nat. Biotechnol. 32(11), 1121–1133 (2014). https://doi.org/10.1038/nbt.3033
L. Sui, N. Danzl, S.R. Campbell, R. Viola, D. Williams, Y. Xing, Y. Wang, N. Phillips, G. Poffenberger, B. Johannesson, J. Oberholzer, A.C. Powers, R.L. Leibel, X. Chen, M. Sykes, D. Egli, β-cell replacement in mice using human type 1 diabetes nuclear transfer embryonic stem cells. Diabetes 67(1), 26–35 (2018). https://doi.org/10.2337/db17-0120
Y.M. Park, C.M. Yang, H.Y. Cho, Therapeutic effects of insulin-producing human umbilical cord-derived mesenchymal stem cells in a type 1 diabetes mouse model. Int. J. Mol. Sci. 23(13), 6877 (2022). https://doi.org/10.3390/ijms23136877
D. Balboa, T. Barsby, V. Lithovius, J. Saarimäki-Vire, M. Omar-Hmeadi, O. Dyachok, H. Montaser, P.E. Lund, M. Yang, H. Ibrahim, A. Näätänen, V. Chandra, H. Vihinen, E. Jokitalo, J. Kvist, J. Ustinov, A.I. Nieminen, E. Kuuluvainen, V. Hietakangas, P. Katajisto, J. Lau, P.O. Carlsson, S. Barg, A. Tengholm, T. Otonkoski, Functional, metabolic and transcriptional maturation of human pancreatic islets derived from stem cells. Nat. Biotechnol. 40(7), 1042–1055 (2022). https://doi.org/10.1038/s41587-022-01219-z
T. Shi, M. Cheung, Urine-derived induced pluripotent/neural stem cells for modeling neurological diseases. Cell Biosci. 11(1), 85 (2021). https://doi.org/10.1186/s13578-021-00594-5
T. Zhao, D. Luo, Y. Sun, X. Niu, Y. Wang, C. Wang, W. Jia, Human urine-derived stem cells play a novel role in the treatment of STZ-induced diabetic mice. J. Mol. Histol. 49(4), 419–428 (2018). https://doi.org/10.1007/s10735-018-9772-5
R.A. Galhom, H.E. Korayem, M.A. Ibrahim, A. Abd-Eltawab Tammam, M.M. Khalifa, E.K. Rashwan, M.H. Al Badawi, Urine-derived stem cells versus their lysate in ameliorating erectile dysfunction in a rat model of type 2 diabetes. Front. Physiol. 13, 854949 (2022). https://doi.org/10.3389/fphys.2022.854949
Z.Z. Jiang, Y.M. Liu, X. Niu, J.Y. Yin, B. Hu, S.C. Guo, Y. Fan, Y. Wang, N.S. Wang, Exosomes secreted by human urine-derived stem cells could prevent kidney complications from type I diabetes in rats. Stem Cell Res. Ther. 7, 24 (2016). https://doi.org/10.1186/s13287-016-0287-2
R. Wu, M. Soland, G. Liu, Y. Shi, C. Zhang, Y. Tang, G. Almeida-Porada, Y. Zhang, Functional characterization of the immunomodulatory properties of human urine-derived stem cells. Transl. Androl. Urol. 10(9), 3566–3578 (2021). https://doi.org/10.21037/tau-21-506
C. Zhou, X.R. Wu, H.S. Liu, X.H. Liu, G.H. Liu, X.B. Zheng, T. Hu, Z.X. Liang, X.W. He, X.J. Wu, L.C. Smith, Y. Zhang, P. Lan, Immunomodulatory effect of urine-derived stem cells on inflammatory bowel diseases via downregulating Th1/Th17 Immune Responses in a PGE2-dependent Manner. J. Crohns. Colitis 14(5), 654–668 (2020). https://doi.org/10.1093/ecco-jcc/jjz200
B. Ouyang, Y. Xie, C. Zhang, C. Deng, L. Lv, J. Yao, Y. Zhang, G. Liu, J. Deng, C. Deng, Extracellular vesicles from human urine-derived stem cells ameliorate erectile dysfunction in a diabetic rat model by delivering proangiogenic MicroRNA. Sex. Med. 7(2), 241–250 (2019). https://doi.org/10.1016/j.esxm.2019.02.001
G. Xiong, L. Tao, W.J. Ma, M.J. Gong, L. Zhao, L.J. Shen, C.L. Long, D.Y. Zhang, Y.Y. Zhang, G.H. Wei, Urine-derived stem cells for the therapy of diabetic nephropathy mouse model. Eur. Rev. Med. Pharmacol. Sci. 24(3), 1316–1324 (2020). https://doi.org/10.26355/eurrev_202002_20189
Y.R. Duan, B.P. Chen, F. Chen, S.X. Yang, C.Y. Zhu, Y.L. Ma, Y. Li, J. Shi, Exosomal microRNA-16-5p from human urine-derived stem cells ameliorates diabetic nephropathy through protection of podocyte. J. Cell. Mol. Med. 25(23), 10798–10813 (2021). https://doi.org/10.1111/jcmm.14558
Y. Fu, J. Guan, S. Guo, F. Guo, X. Niu, Q. Liu, C. Zhang, H. Nie, Y. Wang, Human urine-derived stem cells in combination with polycaprolactone/gelatin nanofibrous membranes enhance wound healing by promoting angiogenesis. J. Transl. Med. 12, 274 (2014). https://doi.org/10.1186/s12967-014-0274-2
Y. Zhang, X. Niu, X. Dong, Y. Wang, H. Li, Bioglass enhanced wound healing ability of urine-derived stem cells through promoting paracrine effects between stem cells and recipient cells. J. Tissue Eng. Regen. Med. 12(3), e1609–e1622 (2018). https://doi.org/10.1002/term.2587
X.R. Zhang, Y.Z. Huang, H.W. Gao, Y.L. Jiang, J.G. Hu, J.K. Pi, A.J. Chen, Y. Zhang, L. Zhou, H.Q. Xie, Hypoxic preconditioning of human urine-derived stem cell-laden small intestinal submucosa enhances wound healing potential. Stem Cell Res. Ther. 11(1), 150 (2020). https://doi.org/10.1186/s13287-020-01662-2
C. Zhang, D. Luo, T. Li, Q. Yang, Y. Xie, H. Chen, L. Lv, J. Yao, C. Deng, X. Liang, R. Wu, X. Sun, Y. Zhang, C. Deng, G. Liu, Transplantation of human urine-derived stem cells ameliorates erectile function and cavernosal endothelial function by promoting autophagy of corpus cavernosal endothelial cells in diabetic erectile dysfunction rats. Stem Cells Int 2019, 2168709 (2019). https://doi.org/10.1155/2019/2168709
D. Zhang, J. Du, M. Yu, L. Suo, Urine-derived stem cells-extracellular vesicles ameliorate diabetic osteoporosis through HDAC4/HIF-1α/VEGFA axis by delivering microRNA-26a-5p. Cell Biol. Toxicol. (2022). https://doi.org/10.1007/s10565-022-09713-5
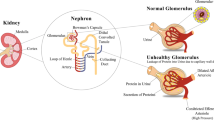
R.A. DeFronzo, W.B. Reeves, A.S. Awad, Pathophysiology of diabetic kidney disease: impact of SGLT2 inhibitors. Nat. Rev. Nephrol. 17(5), 319–334 (2021). https://doi.org/10.1038/s41581-021-00393-8
Y.S. Kanwar, L. Sun, P. Xie, F.Y. Liu, S. Chen, A glimpse of various pathogenetic mechanisms of diabetic nephropathy. Annu. Rev. Pathol. 6, 395–423 (2011). https://doi.org/10.1146/annurev.pathol.4.110807.092150
F.P. Schena, L. Gesualdo, Pathogenetic mechanisms of diabetic nephropathy. J. Am. Soc. Nephrol. 16(Suppl 1), S30–33 (2005). https://doi.org/10.1681/asn.2004110970
S. Wang, Y. Li, J. Zhao, J. Zhang, Y. Huang, Mesenchymal stem cells ameliorate podocyte injury and proteinuria in a type 1 diabetic nephropathy rat model. Biol. Blood Marrow Transplant. 19(4), 538–546 (2013). https://doi.org/10.1016/j.bbmt.2013.01.001
L. Peng, Y. Chen, S. Shi, H. Wen, Stem cell-derived and circulating exosomal microRNAs as new potential tools for diabetic nephropathy management. Stem Cell Res. Ther. 13(1), 25 (2022). https://doi.org/10.1186/s13287-021-02696-w
S. Wang, X. Wen, X.R. Han, Y.J. Wang, M. Shen, S.H. Fan, J. Zhuang, Z.F. Zhang, Q. Shan, M.Q. Li, B. Hu, C.H. Sun, D.M. Wu, J. Lu, Y.L. Zheng, Repression of microRNA-382 inhibits glomerular mesangial cell proliferation and extracellular matrix accumulation via FoxO1 in mice with diabetic nephropathy. Cell Prolif. 51(5), e12462 (2018). https://doi.org/10.1111/cpr.12462
Q. Zhu, Q. Li, X. Niu, G. Zhang, X. Ling, J. Zhang, Y. Wang, Z. Deng, Extracellular vesicles secreted by human urine-derived stem cells promote ischemia repair in a mouse model of hind-limb ischemia. Cell. Physiol. Biochem. 47(3), 1181–1192 (2018). https://doi.org/10.1159/000490214
G. Xiong, W. Tang, D. Zhang, D. He, G. Wei, A. Atala, X.J. Liang, A.J. Bleyer, M.E. Bleyer, J. Yu, J.A. Aloi, J.X. Ma, C.M. Furdui, Y. Zhang, Impaired regeneration potential in urinary stem cells diagnosed from the patients with diabetic nephropathy. Theranostics 9(14), 4221–4232 (2019). https://doi.org/10.7150/thno.34050
G. Broughton 2nd, J.E. Janis, C.E. Attinger, Wound healing: an overview. Plast. Reconstr. Surg. 117(7 Suppl), 1e–S-32e-S (2006). https://doi.org/10.1097/01.prs.0000222562.60260.f9
T.A. Mustoe, K. O’Shaughnessy, O. Kloeters, Chronic wound pathogenesis and current treatment strategies: a unifying hypothesis. Plast. Reconstr. Surg. 117(7 Suppl), 35s–41s (2006). https://doi.org/10.1097/01.prs.0000225431.63010.1b
J. Holl, C. Kowalewski, Z. Zimek, P. Fiedor, A. Kaminski, T. Oldak, M. Moniuszko, A. Eljaszewicz, Chronic diabetic wounds and their treatment with skin substitutes. Cells 10(3), 655 (2021). https://doi.org/10.3390/cells10030655
D. Baltzis, I. Eleftheriadou, A. Veves, Pathogenesis and treatment of impaired wound healing in diabetes mellitus: new insights. Adv. Ther. 31(8), 817–836 (2014). https://doi.org/10.1007/s12325-014-0140-x
Y.M. Cao, M.Y. Liu, Z.W. Xue, Y. Qiu, J. Li, Y. Wang, Q.K. Wu, Surface-structured bacterial cellulose loaded with hUSCs accelerate skin wound healing by promoting angiogenesis in rats. Biochem. Biophys. Res. Commun. 516(4), 1167–1174 (2019). https://doi.org/10.1016/j.bbrc.2019.06.161
J.M. Souren, M. Ponec, R: van Wijk, Contraction of collagen by human fibroblasts and keratinocytes. Vitr. Cell. Dev. Biol. 25(11), 1039–1045 (1989). https://doi.org/10.1007/bf02624138
H. Brem, M. Tomic-Canic, Cellular and molecular basis of wound healing in diabetes. J. Clin. Invest. 117(5), 1219–1222 (2007). https://doi.org/10.1172/jci32169
R.R. Driskell, B.M. Lichtenberger, E. Hoste, K. Kretzschmar, B.D. Simons, M. Charalambous, S.R. Ferron, Y. Herault, G. Pavlovic, A.C. Ferguson-Smith, F.M. Watt, Distinct fibroblast lineages determine dermal architecture in skin development and repair. Nature 504(7479), 277–281 (2013). https://doi.org/10.1038/nature12783
B. Bucalo, W.H. Eaglstein, V. Falanga, Inhibition of cell proliferation by chronic wound fluid. Wound Repair. Regen. 1(3), 181–186 (1993). https://doi.org/10.1046/j.1524-475X.1993.10308.x
N.J. Trengove, H. Bielefeldt-Ohmann, M.C. Stacey, Mitogenic activity and cytokine levels in non-healing and healing chronic leg ulcers. Wound Repair. Regen. 8(1), 13–25 (2000). https://doi.org/10.1046/j.1524-475x.2000.00013.x
G.S. Schultz, A. Wysocki, Interactions between extracellular matrix and growth factors in wound healing. Wound Repair. Regen. 17(2), 153–162 (2009). https://doi.org/10.1111/j.1524-475X.2009.00466.x
Y. Kouidrat, D. Pizzol, T. Cosco, T. Thompson, M. Carnaghi, A. Bertoldo, M. Solmi, B. Stubbs, N. Veronese, High prevalence of erectile dysfunction in diabetes: a systematic review and meta-analysis of 145 studies. Diabet. Med 34(9), 1185–1192 (2017). https://doi.org/10.1111/dme.13403
V.S. Thorve, A.D. Kshirsagar, N.S. Vyawahare, V.S. Joshi, K.G. Ingale, R.J. Mohite, Diabetes-induced erectile dysfunction: epidemiology, pathophysiology and management. J. Diabetes Complic. 25(2), 129–136 (2011). https://doi.org/10.1016/j.jdiacomp.2010.03.003
J.J. Joseph, S.H. Golden, Cortisol dysregulation: the bidirectional link between stress, depression, and type 2 diabetes mellitus. Ann. N. Y. Acad. Sci. 1391(1), 20–34 (2017). https://doi.org/10.1111/nyas.13217
J. Zhuang, P. Gao, H. Chen, Z. Fang, J. Zheng, D. Zhu, J. Hou, Extracellular vesicles from human urine-derived stem cells merged in hyaluronic acid ameliorate erectile dysfunction in type 2 diabetic rats by glans administration. Andrology 10(8), 1673–1686 (2022). https://doi.org/10.1111/andr.13293
Q. Yang, W. Chen, D. Han, C. Zhang, Y. Xie, X. Sun, G. Liu, C. Deng, Intratunical injection of human urine-derived stem cells derived exosomes prevents fibrosis and improves erectile function in a rat model of Peyronie’s disease. Andrologia 52(11), e13831 (2020). https://doi.org/10.1111/and.13831
S.C. DeShields, T.D. Cunningham, Comparison of osteoporosis in US adults with type 1 and type 2 diabetes mellitus. J. Endocrinol. Invest. 41(9), 1051–1060 (2018). https://doi.org/10.1007/s40618-018-0828-x
R. Ma, R. Zhu, L. Wang, Y. Guo, C. Liu, H. Liu, F. Liu, H. Li, Y. Li, M. Fu, D. Zhang, Diabetic osteoporosis: a review of its traditional Chinese medicinal use and clinical and preclinical research. Evid.-Based Complement. Altern. Med. 2016, 3218313 (2016). https://doi.org/10.1155/2016/3218313
V.V. Shanbhogue, S. Hansen, M. Frost, K. Brixen, A.P. Hermann, Bone disease in diabetes: another manifestation of microvascular disease? Lancet Diabetes Endocrinol. 5(10), 827–838 (2017). https://doi.org/10.1016/s2213-8587(17)30134-1
J.H. Kim, A.R. Kim, Y.H. Choi, S. Jang, G.H. Woo, J.H. Cha, E.J. Bak, Y.J. Yoo, Tumor necrosis factor-α antagonist diminishes osteocytic RANKL and sclerostin expression in diabetes rats with periodontitis. PLoS One 12(12), e0189702 (2017). https://doi.org/10.1371/journal.pone.0189702
S.E. Kahn, B. Zinman, J.M. Lachin, S.M. Haffner, W.H. Herman, R.R. Holman, B.G. Kravitz, D. Yu, M.A. Heise, R.P. Aftring, G. Viberti, Rosiglitazone-associated fractures in type 2 diabetes: an Analysis from A Diabetes Outcome Progression Trial (ADOPT). Diabetes Care 31(5), 845–851 (2008). https://doi.org/10.2337/dc07-2270
C.Y. Chen, S.S. Rao, Y.J. Tan, M.J. Luo, X.K. Hu, H. Yin, J. Huang, Y. Hu, Z.W. Luo, Z.Z. Liu, Z.X. Wang, J. Cao, Y.W. Liu, H.M. Li, Y. Chen, W. Du, J.H. Liu, Y. Zhang, T.H. Chen, H.M. Liu, B. Wu, T. Yue, Y.Y. Wang, K. Xia, P.F. Lei, S.Y. Tang, H. Xie, Extracellular vesicles from human urine-derived stem cells prevent osteoporosis by transferring CTHRC1 and OPG. Bone Res. 7, 18 (2019). https://doi.org/10.1038/s41413-019-0056-9
S. Rubler, J. Dlugash, Y.Z. Yuceoglu, T. Kumral, A.W. Branwood, A. Grishman, New type of cardiomyopathy associated with diabetic glomerulosclerosis. Am. J. Cardiol. 30(6), 595–602 (1972). https://doi.org/10.1016/0002-9149(72)90595-4
W.B. Kannel, M. Hjortland, W.P. Castelli, Role of diabetes in congestive heart failure: the Framingham study. Am. J. Cardiol. 34(1), 29–34 (1974). https://doi.org/10.1016/0002-9149(74)90089-7
Y. Tan, Z. Zhang, C. Zheng, K.A. Wintergerst, B.B. Keller, L. Cai, Mechanisms of diabetic cardiomyopathy and potential therapeutic strategies: preclinical and clinical evidence. Nat. Rev. Cardiol. 17(9), 585–607 (2020). https://doi.org/10.1038/s41569-020-0339-2
E. Konduracka, G. Cieslik, D. Galicka-Latala, P. Rostoff, A. Pietrucha, P. Latacz, G. Gajos, M.T. Malecki, J. Nessler, Myocardial dysfunction and chronic heart failure in patients with long-lasting type 1 diabetes: a 7-year prospective cohort study. Acta Diabetol. 50(4), 597–606 (2013). https://doi.org/10.1007/s00592-013-0455-0
S. Bouthoorn, G.B. Valstar, A. Gohar, H.M. den Ruijter, H.B. Reitsma, A.W. Hoes, F.H. Rutten, The prevalence of left ventricular diastolic dysfunction and heart failure with preserved ejection fraction in men and women with type 2 diabetes: a systematic review and meta-analysis. Diabetes Vasc. Dis. Res. 15(6), 477–493 (2018). https://doi.org/10.1177/1479164118787415
W.H. Dillmann, Diabetic cardiomyopathy. Circ. Res. 124(8), 1160–1162 (2019). https://doi.org/10.1161/circresaha.118.314665
D.C. Raev, Which left ventricular function is impaired earlier in the evolution of diabetic cardiomyopathy? An echocardiographic study of young type I diabetic patients. Diabetes Care 17(7), 633–639 (1994). https://doi.org/10.2337/diacare.17.7.633
M. E. Hölscher, C. Bode, H. Bugger, Diabetic cardiomyopathy: does the type of diabetes matter? Int. J. Mol. Sci. 17(12) (2016). https://doi.org/10.3390/ijms17122136
G. Jia, M.A. Hill, J.R. Sowers, Diabetic cardiomyopathy: an update of mechanisms contributing to this clinical entity. Circ. Res. 122(4), 624–638 (2018). https://doi.org/10.1161/circresaha.117.311586
L.V. Heerebeek, N. Hamdani, M.L. Handoko, I. Falcao-Pires, W.J.J.C. Paulus, Diastolic stiffness of the failing diabetic heart: importance of fibrosis, advanced glycation end products, and myocyte resting tension. Circulation 117(1), 43–51 (2008). https://doi.org/10.1161/circulationaha.107.728550
B. Vulesevic, B. McNeill, F. Giacco, K. Maeda, N.J. Blackburn, M. Brownlee, R.W. Milne, E.J. Suuronen, Methylglyoxal-induced endothelial cell loss and inflammation contribute to the development of diabetic cardiomyopathy. Diabetes 65(6), 1699–1713 (2016). https://doi.org/10.2337/db15-0568
A. Wan, B. Rodrigues, Endothelial cell-cardiomyocyte crosstalk in diabetic cardiomyopathy. Cardiovasc. Res. 111(3), 172–183 (2016). https://doi.org/10.1093/cvr/cvw159
J.W. Yau, S.L. Rogers, R. Kawasaki, E.L. Lamoureux, J.W. Kowalski, T. Bek, S.J. Chen, J.M. Dekker, A. Fletcher, J. Grauslund, S. Haffner, R.F. Hamman, M.K. Ikram, T. Kayama, B.E. Klein, R. Klein, S. Krishnaiah, K. Mayurasakorn, J.P. O’Hare, T.J. Orchard, M. Porta, M. Rema, M.S. Roy, T. Sharma, J. Shaw, H. Taylor, J.M. Tielsch, R. Varma, J.J. Wang, N. Wang, S. West, L. Xu, M. Yasuda, X. Zhang, P. Mitchell, T.Y. Wong, Global prevalence and major risk factors of diabetic retinopathy. Diabetes Care 35(3), 556–564 (2012). https://doi.org/10.2337/dc11-1909
A.W. Stitt, T.M. Curtis, M. Chen, R.J. Medina, G.J. McKay, A. Jenkins, T.A. Gardiner, T.J. Lyons, H.P. Hammes, R. Simó, N. Lois, The progress in understanding and treatment of diabetic retinopathy. Prog. Retin. Eye Res. 51, 156–186 (2016). https://doi.org/10.1016/j.preteyeres.2015.08.001
J. Lechner, O.E. O’Leary, A.W. Stitt, The pathology associated with diabetic retinopathy. Vis. Res 139, 7–14 (2017). https://doi.org/10.1016/j.visres.2017.04.003
T. Hamadneh, S. Aftab, N. Sherali, R. Vetrivel Suresh, N. Tsouklidis, M. An, Choroidal changes in diabetic patients with different stages of diabetic retinopathy. Cureus 12(10), e10871 (2020). https://doi.org/10.7759/cureus.10871
D.A. Antonetti, P.S. Silva, A.W. Stitt, Current understanding of the molecular and cellular pathology of diabetic retinopathy. Nat. Rev. Endocrinol. 17(4), 195–206 (2021). https://doi.org/10.1038/s41574-020-00451-4
X. Gu, X. Yu, C. Zhao, P. Duan, T. Zhao, Y. Liu, S. Li, Z. Yang, Y. Li, C. Qian, Z. Yin, Y. Wang, Efficacy and safety of autologous bone marrow mesenchymal stem cell transplantation in patients with diabetic retinopathy. Cell. Physiol. Biochem. 49(1), 40–52 (2018). https://doi.org/10.1159/000492838
B. Mathew, S. Ravindran, X. Liu, L. Torres, M. Chennakesavalu, C.C. Huang, L. Feng, R. Zelka, J. Lopez, M. Sharma, S. Roth, Mesenchymal stem cell-derived extracellular vesicles and retinal ischemia-reperfusion. Biomaterials 197, 146–160 (2019). https://doi.org/10.1016/j.biomaterials.2019.01.016
C.H. Gil, D. Chakraborty, C.P. Vieira, N. Prasain, S. Li Calzi, S.D. Fortmann, P. Hu, K. Banno, M. Jamal, C. Huang, M.S. Sielski, Y. Lin, X. Huang, M.D. Dupont, J.L. Floyd, R. Prasad, A.L.F. Longhini, T.J. McGill, H.M. Chung, M.P. Murphy, D.N. Kotton, M.E. Boulton, M.C. Yoder, M.B. Grant, Specific mesoderm subset derived from human pluripotent stem cells ameliorates microvascular pathology in type 2 diabetic mice. Sci. Adv. 8(9), eabm5559 (2022). https://doi.org/10.1126/sciadv.abm5559
S. Bharadwaj, G. Liu, Y. Shi, C. Markert, K.E. Andersson, A. Atala, Y. Zhang, Characterization of urine-derived stem cells obtained from upper urinary tract for use in cell-based urological tissue engineering. Tissue Eng. A 17(15-16), 2123–2132 (2011). https://doi.org/10.1089/ten.TEA.2010.0637
S.F. Tian, Z.Z. Jiang, Y.M. Liu, X. Niu, B. Hu, S.C. Guo, N.S. Wang, Y. Wang, Human urine-derived stem cells contribute to the repair of ischemic acute kidney injury in rats. Mol. Med. Rep. 16(4), 5541–5548 (2017). https://doi.org/10.3892/mmr.2017.7240
M. Talmon, E. Massara, G. Pruonto, M. Quaregna, F. Boccafoschi, B. Riva, L.G. Fresu, Characterization of a functional Ca(2+) toolkit in urine-derived stem cells and derived skeletal muscle cells. Cell Calcium 103, 102548 (2022). https://doi.org/10.1016/j.ceca.2022.102548
G. Liu, X. Wang, X. Sun, C. Deng, A. Atala, Y. Zhang, The effect of urine-derived stem cells expressing VEGF loaded in collagen hydrogels on myogenesis and innervation following after subcutaneous implantation in nude mice. Biomaterials 34(34), 8617–8629 (2013). https://doi.org/10.1016/j.biomaterials.2013.07.077
X. Ling, G. Zhang, Y. Xia, Q. Zhu, J. Zhang, Q. Li, X. Niu, G. Hu, Y. Yang, Y. Wang, Z. Deng, Exosomes from human urine-derived stem cells enhanced neurogenesis via miR-26a/HDAC6 axis after ischaemic stroke. J. Cell. Mol. Med. 24(1), 640–654 (2020). https://doi.org/10.1111/jcmm.14774
W. Chen, M. Xie, B. Yang, S. Bharadwaj, L. Song, G. Liu, S. Yi, G. Ye, A. Atala, Y. Zhang, Skeletal myogenic differentiation of human urine-derived cells as a potential source for skeletal muscle regeneration. J. Tissue Eng. Regen. Med. 11(2), 334–341 (2017). https://doi.org/10.1002/term.1914
R. Wu, C. Huang, Q. Wu, X. Jia, M. Liu, Z. Xue, Y. Qiu, X. Niu, Y. Wang, Exosomes secreted by urine-derived stem cells improve stress urinary incontinence by promoting repair of pubococcygeus muscle injury in rats. Stem Cell Res. Ther. 10(1), 80 (2019). https://doi.org/10.1186/s13287-019-1182-4
G. Liu, R. Wu, B. Yang, Y. Shi, C. Deng, A. Atala, S. Mou, T. Criswell, Y. Zhang, A cocktail of growth factors released from a heparin hyaluronic-acid hydrogel promotes the myogenic potential of human urine-derived stem cells in vivo. Acta Biomater. 107, 50–64 (2020). https://doi.org/10.1016/j.actbio.2020.02.005
Funding
This work was supported by The Key Research and development project of Jiangxi Province [grant number 20201BBG71006] and the National Natural Science Foundation of China (Grant number 81460018).
Author information
Authors and Affiliations
Contributions
J.X. contributed to the study of conception and design. Material preparation, data collection and analysis were performed by Y.Z. and S.L. W.C. prepared the table and figure. The first draft of the manuscript was written by all authors and all authors commented on previous versions of the manuscript. J.X. contributed to Writing-review, editing and supervision. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zou, Y., Li, S., Chen, W. et al. Urine-derived stem cell therapy for diabetes mellitus and its complications: progress and challenges. Endocrine 83, 270–284 (2024). https://doi.org/10.1007/s12020-023-03552-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-023-03552-y