Abstract
Objective
The study aimed to investigate the association of TyG index with chronic kidney disease (CKD) progression in type 2 diabetes mellitus (T2DM).
Methods
A total of 179 T2DM patients with CKD were retrospectively included. CKD progression was defined as a doubling of baseline serum creatinine or onset of end-stage kidney disease (ESKD). Internal validation was performed by the Kidney Failure Risk Equation (KFRE) model and Net reclassification improvement (NRI).
Results
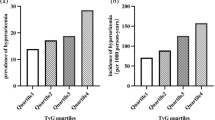
The optimal cut-off value of the TyG index was 9.17. The cumulative incidence of kidney outcomes was significantly higher in the high-TyG group (v.s low-TyG group, P = 0.019). In addition, the high-TyG index was associated with a greater risk of CKD progression (HR 1.794, 95% CI 1.026–3.137, P = 0.040). And reclassification analyses confirmed the final adjusted model improved NRI (61.90% v.s model 2, 43.80% v.s model 1). The further RCS curves presented an inverted S-shaped relationship between the TyG index and the risk of CKD progression. Internal validation verified that a higher TyG index was associated with 2.10-fold increased odds of 2-year ESKD risk >10% (95% CI 1.82–8.21). Moreover, subgroup analysis suggested that the association was more pronounced in those at relatively early CKD stages (higher than stage 2) and with no medication history of oral hypoglycemic agents.
Conclusion
An elevated TyG index was associated with a higher risk of CKD progression in T2DM patients. Our findings suggested that timely targeting insulin sensitivity at the early stages of T2DM might be associated with declined future risk of CKD development.





Similar content being viewed by others
Data availability
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
References
Z. Kleinaki et al. Type 2 diabetes mellitus management in patients with chronic kidney disease: an update. Hormones (Athens) 19(4), 467–476 (2020)
N.H. Cho et al. IDF Diabetes Atlas: Global estimates of diabetes prevalence for 2017 and projections for 2045. Diabetes Res. Clin. Pr. 138, 271–281 (2018)
R.J. Macisaac, E.I. Ekinci, G. Jerums, Markers of and risk factors for the development and progression of diabetic kidney disease. Am. J. Kidney Dis. 63(2 Suppl 2), S39–S62 (2014)
C. Yang et al. CKD in China: Evolving Spectrum and Public Health Implications. Am. J. Kidney Dis. 76(2), 258–264 (2020)
L. Zhang et al. Trends in Chronic Kidney Disease in China. N. Engl. J. Med. 375(9), 905–906 (2016)
K.R. Tuttle et al. Diabetic kidney disease: a report from an ADA Consensus Conference. Diabetes Care 37(10), 2864–2883 (2014)
E.W. Gregg et al. Changes in diabetes-related complications in the United States, 1990-2010. N. Engl. J. Med. 370(16), 1514–1523 (2014)
M. Kato, R. Natarajan, Epigenetics and epigenomics in diabetic kidney disease and metabolic memory. Nat. Rev. Nephrol. 15(6), 327–345 (2019)
L. Liu et al. Association between the triglyceride-glucose index and diabetic nephropathy in patients with type 2 diabetes: A cross-sectional study. J. Diabetes Investig. 12(4), 557–565 (2021)
M. Akhtar et al. Diabetic Kidney Disease: Past and Present. Adv. Anat. Pathol. 27(2), 87–97 (2020)
B.C. Martin et al. Role of glucose and insulin resistance in development of type 2 diabetes mellitus: results of a 25-year follow-up study. Lancet 340(8825), 925–929 (1992)
B. Spoto, A. Pisano, C. Zoccali, Insulin resistance in chronic kidney disease: a systematic review. Am. J. Physiol. Ren. Physiol. 311(6), F1087–F1108 (2016)
A. Mitrofanova, M.A. Sosa, A. Fornoni, Lipid mediators of insulin signaling in diabetic kidney disease. Am. J. Physiol. Ren. Physiol. 317(5), F1241–F1252 (2019)
G.I. Welsh et al. Insulin signaling to the glomerular podocyte is critical for normal kidney function. Cell Metab. 12(4), 329–340 (2010)
D.M. Maahs et al. Higher prevalence of elevated albumin excretion in youth with type 2 than type 1 diabetes: the SEARCH for Diabetes in Youth study. Diabetes Care 30(10), 2593–2598 (2007)
E. Ahlqvist et al. Novel subgroups of adult-onset diabetes and their association with outcomes: a data-driven cluster analysis of six variables. Lancet Diabetes Endocrinol. 6(5), 361–369 (2018)
E. Nerpin et al. Insulin sensitivity measured with euglycemic clamp is independently associated with glomerular filtration rate in a community-based cohort. Diabetes Care 31(8), 1550–1555 (2008)
A.I. Parvanova et al. Insulin resistance and microalbuminuria: a cross-sectional, case-control study of 158 patients with type 2 diabetes and different degrees of urinary albumin excretion. Diabetes 55(5), 1456–1462 (2006)
H. Pham et al. Chronic kidney disease, insulin resistance, and incident diabetes in older adults. Clin. J. Am. Soc. Nephrol. 7(4), 588–594 (2012)
S. Zhao et al. Association between macro- and microvascular damage and the triglyceride glucose index in community-dwelling elderly individuals: the Northern Shanghai Study. Cardiovasc. Diabetol. 18(1), 95 (2019)
L. Lv et al. Relationship Between the TyG Index and Diabetic Kidney Disease in Patients with Type-2 Diabetes Mellitus. Diabetes Metab. Syndr. Obes. 14, 3299–3306 (2021)
Y.L. Ou et al. Obesity-related indices are associated with albuminuria and advanced kidney disease in type 2 diabetes mellitus. Ren. Fail 43(1), 1250–1258 (2021)
Y. Pan et al. Association between Diabetes Complications and the Triglyceride-Glucose Index in Hospitalized Patients with Type 2 Diabetes. J. Diabetes Res. 2021, 8757996 (2021)
S. Zou et al. Association of the triglyceride-glucose index and major adverse cardiac and cerebrovascular events in female patients undergoing percutaneous coronary intervention with drug-eluting stents: A retrospective study. Diabetes Res. Clin. Pr. 181, 109073 (2021)
J. Fritz et al. The Triglyceride-Glucose Index and Obesity-Related Risk of End-Stage Kidney Disease in Austrian Adults. JAMA Netw. Open 4(3), e212612 (2021)
M. Zhang et al. Cumulative increased risk of incident type 2 diabetes mellitus with increasing triglyceride glucose index in normal-weight people: The Rural Chinese Cohort Study. Cardiovasc. Diabetol. 16(1), 30 (2017)
L. Sánchez-Íñigo et al. The TyG index may predict the development of cardiovascular events. Eur. J. Clin. Investig. 46(2), 189–197 (2016)
K.G. Alberti, P.Z. Zimmet, Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: diagnosis and classification of diabetes mellitus provisional report of a WHO consultation. Diabet. Med. 15(7), 539–553 (1998)
Z.N.K.Z. Zhi, [National guidelines for the prevention and control of diabetes in primary care(2018)]. Zhonghua Nei Ke Za Zhi. 57(12), 885–893 (2018)
A.K. Cheung et al. KDIGO 2021 Clinical Practice Guideline for the Management of Blood Pressure in Chronic Kidney Disease. Kidney Int. 99(3s), S1–S87 (2021)
B.H. Rovin et al. Executive summary of the KDIGO 2021 Guideline for the Management of Glomerular Diseases. Kidney Int. 100(4), 753–779 (2021)
F. Guerrero-Romero et al. The product of triglycerides and glucose, a simple measure of insulin sensitivity. Comparison with the euglycemic-hyperinsulinemic clamp. J. Clin. Endocrinol. Metab 95(7), 3347–3351 (2010)
L.E. Simental-Mendía, M. Rodríguez-Morán, F. Guerrero-Romero, The product of fasting glucose and triglycerides as surrogate for identifying insulin resistance in apparently healthy subjects. Metab. Syndr. Relat. Disord. 6(4), 299–304 (2008)
A.S. Levey et al. A new equation to estimate glomerular filtration rate. Ann. Intern. Med. 150(9), 604–612 (2009)
N. Tangri et al. A predictive model for progression of chronic kidney disease to kidney failure. Jama 305(15), 1553–1559 (2011)
C.L. Ramspek et al. Kidney Failure Prediction Models: A Comprehensive External Validation Study in Patients with Advanced CKD. J. Am. Soc. Nephrol. 32(5), 1174–1186 (2021)
C.S. Lennartz et al. External Validation of the Kidney Failure Risk Equation and Re-Calibration with Addition of Ultrasound Parameters. Clin. J. Am. Soc. Nephrol. 11(4), 609–615 (2016)
A. Sánchez-García, R. Rodríguez-Gutiérrez, Diagnostic Accuracy of the Triglyceride and Glucose Index for Insulin Resistance: A Systematic Review. Int. J. Endocrinol. 2020, 4678526 (2020)
A. Borai, C. Livingstone, G.A. Ferns, The biochemical assessment of insulin resistance. Ann. Clin. Biochem. 44(Pt 4), 324–342 (2007)
D.R. Matthews et al. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 28(7), 412–419 (1985)
M. Amatruda, G. Gembillo, The Aggressive Diabetic Kidney Disease in Youth-Onset Type 2 Diabetes: Pathogenetic Mechanisms and Potential Therapies. Medicina (Kaunas) 57(9), 868 (2021)
L. Hinden et al. Pathogenesis of diabesity-induced kidney disease: role of kidney nutrient sensing. FEBS J. 289(4), 901–921 (2022)
S. Tiwari et al. Deletion of the insulin receptor in the proximal tubule promotes hyperglycemia. J. Am. Soc. Nephrol. 24(8), 1209–1214 (2013)
E. Ahlqvist, R.B. Prasad, L. Groop, 100 YEARS OF INSULIN: Towards improved precision and a new classification of diabetes mellitus. J. Endocrinol. 252(3), R59–R70 (2021)
C.C. Hsu et al. Association between insulin resistance and development of microalbuminuria in type 2 diabetes: a prospective cohort study. Diabetes Care 34(4), 982–987 (2011)
M. Vedovato et al. Effect of sodium intake on blood pressure and albuminuria in Type 2 diabetic patients: the role of insulin resistance. Diabetologia 47(2), 300–303 (2004)
P. Bjornstad et al. Insulin sensitivity is an important determinant of renal health in adolescents with type 2 diabetes. Diabetes Care 37(11), 3033–3039 (2014)
S.H. Khan et al. Metabolic clustering of risk factors: evaluation of Triglyceride-glucose index (TyG index) for evaluation of insulin resistance. Diabetol. Metab. Syndr. 10, 74 (2018)
S. Duan et al. Current Challenges and Future Perspectives of Renal Tubular Dysfunction in Diabetic Kidney Disease. Front. Endocrinol. (Lausanne) 12, 661185 (2021)
L. Opazo-Ríos, S. Mas, Lipotoxicity and Diabetic Nephropathy: Novel Mechanistic Insights and Therapeutic Opportunities. Int. J. Mol. Sci. 21(7), 2632 (2020)
E. Ejerblad et al. Obesity and risk for chronic renal failure. J. Am. Soc. Nephrol. 17(6), 1695–1702 (2006)
R. Nakamichi, K. Hayashi, H. Itoh, Effects of High Glucose and Lipotoxicity on Diabetic Podocytes. Nutrients 13(1), 241 (2021)
H.M. Colhoun et al. Effects of atorvastatin on kidney outcomes and cardiovascular disease in patients with diabetes: an analysis from the Collaborative Atorvastatin Diabetes Study (CARDS). Am. J. Kidney Dis. 54(5), 810–819 (2009)
A. Keech et al. Effects of long-term fenofibrate therapy on cardiovascular events in 9795 people with type 2 diabetes mellitus (the FIELD study): randomised controlled trial. Lancet 366(9500), 1849–1861 (2005)
N.D. Vaziri, Disorders of lipid metabolism in nephrotic syndrome: mechanisms and consequences. Kidney Int. 90(1), 41–52 (2016)
L. Zeni et al. A more tubulocentric view of diabetic kidney disease. J. Nephrol. 30(6), 701–717 (2017)
Acknowledgements
The authors thank the patients for participation in our study and the staffs of the Department of Nephrology at the First Affiliated Hospital of Nanjing Medical University for assistance.
Author information
Authors and Affiliations
Contributions
S.D. designed the research and contributed to the writing. M.Z. and F.L. analyzed the data and performed statistical analysis. F.L., C.C., S.C., and L.G. conducted the research, C.Z., H.G., M.Z., B.S., and H.M. reviewed the paper. C.X. and B.Z. conceived, coordinated the study, and had responsibility for its final content. Y.Y. is the guarantor of this work, who had complete access to all the data in the study and takes ultimate responsibility for the study design and integrity of data analysis. All authors have read the final paper and approved the submission.
Funding
This work was supported by grants from the National Natural Science Foundation of China (82170699, 81870469 to Y.Y., No. 82100767 to S.D.), the Natural Science Foundation of Jiangsu Province (BK20191075 to S.D.), “PRO•Run” Fund of the Nephrology Group of CEBM (KYJ202206-0003-6 to Y.Y., KYS2021-03-02-14 to B.Z.), the “333 Project” of Jiangsu Province, the Six Talent Peaks Project in Jiangsu Province (WSN-010 to Y.Y.), “Yiluqihang·Shenmingyuanyang” medical development And Scientific Research Fund project on Kidney Diseases (SMYY20220301001 to Y.Y.), Project of clinical capability improvement of the First Affiliated Hospital of Nanjing Medical University (JSPH-MC-2021-14 to S.D.), Project of Bethune PuAi Medical Research Fund (PAYJ-058 to S.D.), Postgraduate Research & Practice Innovation Program of Jiangsu Province (JX10213856 to M.Z.), and the Priority Academic Program Development (PAPD) of Jiangsu Higher Education Institution.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethics approval and consent to participate
All procedures and methods were performed in accordance with relevant guidelines and regulations. This study protocol was reviewed and approved by the Ethics Committee of the First Affiliated Hospital of Nanjing Medical University. Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Duan, S., Zhou, M., Lu, F. et al. Triglyceride-glucose index is associated with the risk of chronic kidney disease progression in type 2 diabetes. Endocrine 81, 77–89 (2023). https://doi.org/10.1007/s12020-023-03357-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-023-03357-z