Abstract
Purpose
The potential mechanisms of endocrine therapy for thrombosis remain currently unclear, and more studies are warranted for further investigation and elucidation. However, high plasma concentration of lipoprotein(a) (Lp(a)) is a recognized prothrombotic factor. The aim of our study was to systematically evaluate the effect of different anti-oestrogen therapy on plasma Lp(a) level in postmenopausal women.
Methods
A systematic literature search was conducted in multiple electronic databases to identify the randomized, double-blind, placebo-controlled clinical studies on this topic. Effect size for changes in Lp(a) was expressed as mean difference (MD) and 95% confidence intervals (CI).
Results
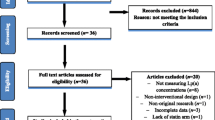
Data were pooled from 10 clinical trials comprising 24 treatment arms, which included 2049 women (1128 women in the active-treated arms and 921 women in the control arms). Meta-analysis of data suggested that anti-oestrogen therapy in women significantly reduced Lp(a) [MD = −5.92% (95%CI: −9.05%,−2.8%)].
Conclusions
This observation is of both clinical and pathophysiological relevance, also in view that the identification of molecular determinants and cellular pathways implicated in Lp(a) synthesis and metabolism is still of concern as a critical issue in lipidology and CV prevention.




Similar content being viewed by others
Data availability
Data supporting study’s findings are available from the Corresponding Author.
References
S. Coassin, F. Kronenberg, Lipoprotein(a) beyond the kringle IV repeat polymorphism: the complexity of genetic variation in the LPA gene. Atherosclerosis 349, 17–35 (2022). https://doi.org/10.1016/j.atherosclerosis.2022.04.003
D. Boffelli, D.A. Zajchowski, Z. Yang, R.M. Lawn, Estrogen modulation of apolipoprotein(a) expression. Identif. Regul. Elem. J. Biol. Chem. 274, 15569–15574 (1999). https://doi.org/10.1074/jbc.274.22.15569
F. Nakagami, H. Nakagami, M.K. Osako, M. Iwabayashi, Y. Taniyama, T. Doi, H. Shimizu, M. Shimamura, H. Rakugi, R. Morishita, Estrogen attenuates vascular remodeling in Lp(a) transgenic mice. Atherosclerosis 211, 41–47 (2010). https://doi.org/10.1016/j.atherosclerosis.2010.01.016
G. Ferretti, T. Bacchetti, T.P. Johnston, M. Banach, M. Pirro, A. Sahebkar, Lipoprotein(a): A missing culprit in the management of athero-thrombosis. J. Cell. Physiol. 233, 2966–2981 (2018). https://doi.org/10.1002/jcp.26050
M.J. Page, J.E. McKenzie, P.M. Bossuyt, I. Boutron, T.C. Hoffmann, C.D. Mulrow, L. Shamseer, J.M. Tetzlaff, E.A. Akl, S.E. Brennan et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 372, n71 (2021). https://doi.org/10.1136/bmj.n71
J. Higgins, S. Green, Cochrane Handbook for Systematic Reviews of Interventions. Version 5.0. 2. 2009. (John Wiley and Sons Ltd., Chichester, UK, Ref Type: Report, 2010)
F. Fogacci, D. Grassi, M. Rizzo, A.F.G. Cicero, Metabolic effect of berberine-silymarin association: A meta-analysis of randomized, double-blind, placebo-controlled clinical trials. Phytother. Res. 33, 862–870 (2019). https://doi.org/10.1002/ptr.6282
M. Borenstein, L. Hedges, J. Higgins, H. Rothstein, Comprehensive meta-analysis version 3. 104 (Biostat, Englewood, NJ, 2005)
D. Follmann, P. Elliott, I. Suh, J. Cutler, Variance imputation for overviews of clinical trials with continuous response. J. Clin. Epidemiol. 45, 769–773 (1992)
X. Wan, W. Wang, J. Liu, T. Tong, Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med. Res. Methodol. 14, 135 (2014)
W.G. Melsen, M.C. Bootsma, M.M. Rovers, M.J. Bonten, The effects of clinical and statistical heterogeneity on the predictive values of results from meta-analyses. Clin. Microbiol. Infect. 20, 123–129 (2014)
M.J. Bown, A.J. Sutton, Quality control in systematic reviews and meta-analyses. Eur. J. Vasc. Endovasc. Surg. 40, 669–677 (2010)
A.F.G. Cicero, R. Pontremoli, F. Fogacci, F. Viazzi, C. Borghi, Effect of Bempedoic Acid on Serum Uric Acid and Related Outcomes: A Systematic Review and Meta-analysis of the available Phase 2 and Phase 3 Clinical Studies. Drug. Saf. 43, 727–736 (2020). https://doi.org/10.1007/s40264-020-00931-6
C.B. Begg, M. Mazumdar, Operating characteristics of a rank correlation test for publication bias. Biometrics 50, 1088–1101 (1994)
J.A. Sterne, D. Gavaghan, M. Egger, Publication and related bias in meta-analysis: power of statistical tests and prevalence in the literature. J. Clin. Epidemiol. 53, 1119–1129 (2000)
S. Duval, R. Tweedie, Trim and fill: a simple funnel plot–based method of testing and adjusting for publication bias in meta-analysis. Biometrics 56, 455–463 (2000)
R. Rosenthal, The file drawer problem and tolerance for null results. Psychol. Bull. 86, 638–664 (1979). https://doi.org/10.1037/0033-2909.86.3.638
J.V. Selby, M.A. Austin, C. Sandholzer, C.P. Quesenberry Jr, D. Zhang, E. Mayer, G. Utermann, Environmental and behavioral influences on plasma lipoprotein(a) concentration in women twins. Prev. Med. 23, 345–353 (1994). https://doi.org/10.1006/pmed.1994.1048
J. Suk Danik, N. Rifai, J.E. Buring, P.M. Ridker, Lipoprotein(a), hormone replacement therapy, and risk of future cardiovascular events. J. Am. Coll. Cardiol. 52, 124–131 (2008). https://doi.org/10.1016/j.jacc.2008.04.009
B.V. Howard, J.E. Rossouw, Estrogens and cardiovascular disease risk revisited: the Women’s Health Initiative. Curr. Opin. Lipido. 24, 493–499 (2013)
A. Sahebkar, M.C. Serban, P. Penson, C. Gurban, S. Ursoniu, P.P. Toth, S.R. Jones, G. Lippi, K. Kotani, K. Kostner et al. Lipid and Blood Pressure Meta-analysis Collaboration (LBPMC) Group. The Effects of Tamoxifen on Plasma Lipoprotein(a) Concentrations: Systematic Review and Meta-Analysis. Drugs 77, 1187–1197 (2017). https://doi.org/10.1007/s40265-017-0767-4
G. Ferretti, T. Bacchetti, L.E. Simental-Mendía, Ž. Reiner, M. Banach, A. Sahebkar, Raloxifene Lowers Plasma Lipoprotein(a) Concentrations: a Systematic Review and Meta-analysis of Randomized Placebo-Controlled Trials. Cardiovasc. Drugs Ther. 31, 197–208 (2017). https://doi.org/10.1007/s10557-017-6721-6
F. Fogacci, A.F. Cicero, S. D’Addato, L. D’Agostini, M. Rosticci, M. Giovannini, E. Bertagnin, C. Borghi; Brisighella Heart Study Group, Serum lipoprotein(a) level as long-term predictor of cardiovascular mortality in a large sample of subjects in primary cardiovascular prevention: data from the Brisighella Heart Study. Eur. J. Intern. Med. 37, 49–55 (2017). https://doi.org/10.1016/j.ejim.2016.08.018
F. Kronenberg, S. Mora, E.S.G. Stroes, B.A. Ference, B.J. Arsenault, L. Berglund, M.R. Dweck, M. Koschinsky, G. Lambert, F. Mach, et al. Lipoprotein(a) in atherosclerotic cardiovascular disease and aortic stenosis: a European Atherosclerosis Society consensus statement. Eur. Heart. J. ehac361 (2022). https://doi.org/10.1093/eurheartj/ehac361. Epub ahead of print
A. Cicero, F. Fogacci, G. Derosa, A. D’Angelo, F. Ventura, E. Rizzoli, S. D’Addato, C. Borghi, On Behalf Of The Brisighella Heart Study Group. Lipoprotein(a) Serum Levels Predict Pulse Wave Velocity in Subjects in Primary Prevention for Cardiovascular Disease with Large Apo(a) Isoforms: Data from the Brisighella Heart Study. Biomedicines 10, 656 (2022). https://doi.org/10.3390/biomedicines10030656
F. Fogacci, A. Cicero, S. D’Addato, M. Giovannini, C. Borghi; Brisighella Heart Study Group, Effect of spontaneous changes in dietary components and lipoprotein(a) levels: Data from the Brisighella Heart Study. Atherosclerosis 262, 202–204 (2017). https://doi.org/10.1016/j.atherosclerosis.2017.03.036
L. Strilchuk, F. Fogacci, A.F. Cicero, Safety and tolerability of injectable lipid-lowering drugs: an update of clinical data. Expert Opin. Drug Saf. 18, 611–621 (2019). https://doi.org/10.1080/14740338.2019.1620730
N.S. Nurmohamed, J.M. Kraaijenhof, E.S.G. Stroes, Lp(a): a New Pathway to Target? Curr. Atheroscler. Rep. (2022) https://doi.org/10.1007/s11883-022-01060-4. Epub ahead of print
M.J. Borrelli, A. Youssef, M.B. Boffa, M.L. Koschinsky, New Frontiers in Lp(a)-Targeted Therapies. Trends Pharmacol. Sci. 40, 212–225 (2019). https://doi.org/10.1016/j.tips.2019.01.004
S.P.A. McCormick, W.J. Schneider, Lipoprotein(a) catabolism: a case of multiple receptors. Pathology 51, 155–164 (2019). https://doi.org/10.1016/j.pathol.2018.11.003
D.J. Rader, W. Cain, L.A. Zech, D. Usher, H.B. Brewer Jr., Variation in lipoprotein(a) concentrations among individuals with the same apolipoprotein (a) isoform is determined by the rate of lipoprotein(a) production. J. Clin. Investig. 91, 443–447 (1993). https://doi.org/10.1172/JCI116221
B.H. Mitlak, F.J. Cohen, In search of optimal long-term female hormone replacement: the potential of selective estrogen receptor modulators. Horm. Res. 48, 155–163 (1997). https://doi.org/10.1159/000185507
M.L. Snyder, R.V. Hay, P.F. Whitington, A.M. Scanu, G.M. Fless, Binding and degradation of lipoprotein(a) and LDL by primary cultures of human hepatocytes. Comparison with cultured human monocyte-macrophages and fibroblasts. Arterioscler. Thromb. 14, 770–779 (1994). https://doi.org/10.1161/01.atv.14.5.770
R. Romagnuolo, C.A. Scipione, M.B. Boffa, S.M. Marcovina, N.G. Seidah, M.L. Koschinsky, Lipoprotein(a) catabolism is regulated by proprotein convertase subtilisin/kexin type 9 through the low density lipoprotein receptor. J. Biol. Chem. 290, 11649–11662 (2015). https://doi.org/10.1074/jbc.M114.611988
F. Cerrato, M.E. Fernández-Suárez, R. Alonso, M. Alonso, C. Vázquez, O. Pastor, P. Mata, M.A. Lasunción, D. Gómez-Coronado, Clinically used selective oestrogen receptor modulators increase LDL receptor activity in primary human lymphocytes. Br. J. Pharmacol. 172, 1379–1394 (2015). https://doi.org/10.1111/bph.13016
M.C. Verwer, F. Waissi, J.M. Mekke, M. Dekker, E.S.G. Stroes, G.J. de Borst, J. Kroon, C.E.V.B. Hazenberg, D.P.V. de Kleijn, High lipoprotein(a) is associated with major adverse limb events after femoral artery endarterectomy. Atherosclerosis 349, 196–203 (2022). https://doi.org/10.1016/j.atherosclerosis.2021.11.019
G. Reyes-Soffer, H.N. Ginsberg, L. Berglund, P.B. Duell, S.P. Heffron, P.R. Kamstrup, D.M. Lloyd-Jones, S.M. Marcovina, C. Yeang, M.L. Koschinsky; American Heart Association Council on Arteriosclerosis, Thrombosis and Vascular Biology; Council on Cardiovascular Radiology and Intervention; and Council on Peripheral Vascular Disease, Lipoprotein(a): A Genetically Determined, Causal, and Prevalent Risk Factor for Atherosclerotic Cardiovascular Disease: A Scientific Statement From the American Heart Association. Arterioscler. Thromb. Vasc. Biol. 42, e48–e60 (2022). https://doi.org/10.1161/ATV.0000000000000147
M.B. Boffa, Beyond fibrinolysis: The confounding role of Lp(a) in thrombosis. Atherosclerosis 349, 72–81 (2022). https://doi.org/10.1016/j.atherosclerosis.2022.04.009
F. Dentali, V. Gessi, R. Marcucci, M. Gianni, A.M. Grandi, M. Franchini, Lipoprotein(a) as a Risk Factor for Venous Thromboembolism: A Systematic Review and Meta-analysis of the Literature. Semin. Thrombosis Hemost. 43, 614–620 (2017). https://doi.org/10.1055/s-0036-1598002
F. Paciullo, D. Giannandrea, G. Virgili, C. Cagini, P. Gresele, Role of Increased Lipoprotein (a) in Retinal Vein Occlusion: A Systematic Review and Meta-analysis. TH Open.: Companion J. Thrombosis Haemost. 5, e295–e302 (2021). https://doi.org/10.1055/s-0041-1732803
M.N. Mirzabeigi, J.A. Nelson, J.P. Fischer, S.J. Kovach, J.M. Serletti, L.C. Wu, S. Kanchwala, Tamoxifen (selective estrogen-receptor modulators) and aromatase inhibitors as potential perioperative thrombotic risk factors in free flap breast reconstruction. Plast. Reconstr. Surg. 135, 670e–679e (2015). https://doi.org/10.1097/PRS.0000000000001127
H. Shi, H.O. Santos, I.G.O. de Souza, G.J. Hoilat, C.E.C. Martins, H.K. Varkaneh, J.A. Alkhwildi, A.T. Hejji, F. Almuqayyid et al. The Effect of Raloxifene Treatment on Lipid Profile in Elderly Individuals: a Systematic Review and Meta-analysis of Randomized Clinical Trials. Clin. Ther. 43, 297–317 (2021). https://doi.org/10.1016/j.clinthera.2021.07.017
S.A. Alomar, M.A. Găman, K. Prabahar, O.A. Arafah, F. Almarshood, S. Baradwan, S.A.S. Aboudi, M. Abuzaid, A.A.M.A. Almubarki, O. Alomar et al. The effect of tamoxifen on the lipid profile in women: a systematic review and meta-analysis of randomized controlled trials. Exp. Gerontol. 159, 111680 (2022). https://doi.org/10.1016/j.exger.2021.111680
V.A. Korneva, T.Y. Kuznetsova, U. Julius, Modern Approaches to Lower Lipoprotein(a) Concentrations and Consequences for Cardiovascular Diseases. Biomedicines 9, 1271 (2021). https://doi.org/10.3390/biomedicines9091271
B. Andersson, G. Johannsson, G. Holm, B.A. Bengtsson, A. Sashegyi, I. Pavo, T. Mason, P.W. Anderson, Raloxifene does not affect insulin sensitivity or glycemic control in postmenopausal women with type 2 diabetes mellitus: a randomized clinical trial. J. Clin. Endocrinol. Metab. 87, 122–128 (2002). https://doi.org/10.1210/jcem.87.1.8168
T. Cigler, H. Richardson, M.J. Yaffe, C.J. Fabian, D. Johnston, J.N. Ingle, E. Nassif, R.L. Brunner, M.E. Wood, J.L. Pater et al. A randomized, placebo-controlled trial (NCIC CTG MAP.2) examining the effects of exemestane on mammographic breast density, bone density, markers of bone metabolism and serum lipid levels in postmenopausal women. Breast Cancer Res. Treat. 126, 453–461 (2011). https://doi.org/10.1007/s10549-010-1322-0
A. Decensi, B. Bonanni, A. Guerrieri-Gonzaga, S. Gandini, C. Robertson, H. Johansson, R. Travaglini, M.T. Sandri, A. Tessadrelli, G. Farante et al. Biologic activity of tamoxifen at low doses in healthy women. J. Natl Cancer Inst. 90, 1461–1467 (1998). https://doi.org/10.1093/jnci/90.19.1461
R.A. Lobo, J.V. Pinkerton, M.L.S. Gass, M.H. Dorin, S. Ronkin, J.H. Pickar, G. Constantine, Evaluation of bazedoxifene/conjugated estrogens for the treatment of menopausal symptoms and effects on metabolic parameters and overall safety profile. Fertil. Steril. 92, 1025–1038 (2009). https://doi.org/10.1016/j.fertnstert.2009.03.113
P.E. Lønning, J. Geisler, L.E. Krag, B. Erikstein, Y. Bremnes, A.I. Hagen, E. Schlichting, E.A. Lien, E.S. Ofjord, J. Paolini et al. Effects of exemestane administered for 2 years versus placebo on bone mineral density, bone biomarkers, and plasma lipids in patients with surgically resected early breast cancer. J. Clin. Oncol. 23, 5126–5137 (2005). https://doi.org/10.1200/JCO.2005.07.097
D.A. Shewmon, J.L. Stock, C.J. Rosen, K.M. Heiniluoma, M.M. Hogue, A. Morrison, E.M. Doyle, T. Ukena, V. Weale, S. Baker, Tamoxifen and estrogen lower circulating lipoprotein(a) concentrations in healthy postmenopausal women. Arterioscler. Thromb. 14, 1586–1593 (1994). https://doi.org/10.1161/01.atv.14.10.1586
R.G. Smolders, T.E. Vogelvang, V. Mijatovic, W.M. van Baal, S.J. Neele, J.C. Netelenbos, P. Kenemans, M.J. van der Mooren, A 2-year, randomized, comparative, placebo-controlled study on the effects of raloxifene on lipoprotein(a) and homocysteine. Maturitas 41, 105–114 (2002). https://doi.org/10.1016/s0378-5122(01)00280-8
T.E. Vogelvang, V. Mijatovic, P. Kenemans, T. Teerlink, M.J. van der Mooren, HMR 3339, a novel selective estrogen receptor modulator, reduces total cholesterol, low-density lipoprotein cholesterol, and homocysteine in healthy postmenopausal women. Fertil. Steril. 82, 1540–1549 (2004). https://doi.org/10.1016/j.fertnstert.2004.05.093
B.W. Walsh, L.H. Kuller, R.A. Wild, S. Paul, M. Farmer, J.B. Lawrence, A.S. Shah, P.W. Anderson, Effects of raloxifene on serum lipids and coagulation factors in healthy postmenopausal women. JAMA 279, 1445–1451 (1998). https://doi.org/10.1001/jama.279.18.1445
K.M. Wasan, P.E. Goss, P.H. Pritchard, L. Shepherd, M.J. Palmer, S. Liu, D. Tu, J.N. Ingle, M. Heath, D. Deangelis et al. The influence of letrozole on serum lipid concentrations in postmenopausal women with primary breast cancer who have completed 5 years of adjuvant tamoxifen (NCIC CTG MA.17L). Ann. Oncol. 16, 707–715 (2005). https://doi.org/10.1093/annonc/mdi158
Author information
Authors and Affiliations
Contributions
Conceptualization, F.F. and A.F.G.C.; Methodology, F.F. and A.F.G.C.; Formal analysis, F.F.; Investigation, F.F., C.B., S.D., G.S., and A.F.G.C.; Data curation, F.F., C.B., and A.F.G.C.; Writing—original draft preparation, F.F. and A.F.G.C.; Writing—review and editing, C.B., S.D., and G.S.; Visualization, F.F.; Supervision, A.F.G.C.; Project administration, A.F.G.C. All authors have read and agreed to the published version of the paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethics approval
Ethical review and approval were waived for this study due to its design.
Informed consent
Patient consent was waived due to the study design.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fogacci, F., Borghi, C., Davinelli, S. et al. Impact of anti-oestrogen therapy on lipoprotein(a) in postmenopausal women: a systematic review and meta-analysis of double-blind placebo-controlled clinical studies. Endocrine 80, 292–302 (2023). https://doi.org/10.1007/s12020-022-03287-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-022-03287-2