Abstract
Purpose
To evaluate the effect of cabergoline treatment on metabolic parameters including the Triglyceride-glucose (TyG) index in newly diagnosed patients with prolactinoma.
Methods
71 consecutive nondiabetic patients with prolactinoma were enrolled. Anthropometric and laboratory tests including TyG index were measured at baseline, 3 and 6 months visits. Treatment with cabergoline at the dose of 0.25 mg twice weekly was started and increased according to prolactin levels and continued for 6 months.
Results
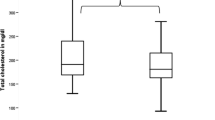
At the baseline examination, the mean (SD) age, body mass index (BMI), waist circumference (WC), systolic blood pressure (SBP), and diastolic blood pressure (DBP) of patients were 36.2 (10.5) years, 29.2 (5.0) kg/m2, 98.2 (13.7) cm, 115.3 (13.3) mmHg, and 71.4 (8.1) mmHg, respectively. Forty-one (57.7%) of patients were women and 46 (64.8%) had microadenoma. Cabergoline treatment significantly improved anthropometric and metabolic measures including BMI, WC, fasting plasma glucose, triglycerides, total cholesterol, low-density lipoprotein cholesterol, high-density lipoprotein cholesterol, uric acid (only among women), TyG index, and hypogonadism. Blood pressure (both systolic and diastolic) levels remained steady except for a significant decrease in DBP after 6 months of treatment only among women. A declining trend in MetS prevalence was found from baseline to a 3-month evaluation in both genders which was statistically significant among men.
Conclusion
Short-term treatment with cabergoline can significantly improve cardiovascular risk factors except for blood pressure. Moreover, the TyG index as a surrogate marker of insulin resistance decreased significantly after the reduction of prolactin by treatment. Generally, results were similar among both genders.
Similar content being viewed by others
Data availability
All data and materials are available upon request.
References
M.P. Gillam, M.E. Molitch, G. Lombardi, A. Colao. Advances in the treatment of prolactinomas. 27(5), 485–534 (2006)
B.A. Doğan, A. Arduç, M.M. Tuna, N.I. Nasıroğlu, S. Işık, D. Berker et al. Evaluation of atherosclerosis after cessation of cabergoline therapy in patients with prolactinoma. Anatol. J. Cardiol. 16(6), 440 (2016)
R. Haring, N. Friedrich, H. Völzke, R.S. Vasan, S.B. Felix, M. Dörr et al. Positive association of serum prolactin concentrations with all-cause and cardiovascular mortality. Eur. Heart J. 35(18), 1215–1221 (2014)
A. Jurek, P. Krzesiński, G. Gielerak, B. Uziębło-Życzkowska, P. Witek, G. Zieliński et al. Prolactinoma and cardiovascular diseases–an interdisciplinary problem. Pediatr. Med Rodz. 2021 17(4), 294–298 (2021). https://doi.org/10.15557/PiMR.2021.0046
O. Serri, L. Li, J.C. Mamputu, M.C. Beauchamp, F. Maingrette, G. Renier, The influences of hyperprolactinemia and obesity on cardiovascular risk markers: effects of cabergoline therapy. Clin. Endocrinol. 64(4), 366–370 (2006)
C.M. Silva, F.R. Barbosa, G.A. Lima, L. Warszawski, R. Fontes, R.C. Domingues et al. BMI and metabolic profile in patients with prolactinoma before and after treatment with dopamine agonists. Obes. (Silver Spring) 19(4), 800–805 (2011)
J. Krogh, C. Selmer, C. Torp-Pedersen, G.H. Gislason, C. Kistorp, Hyperprolactinemia and the association with all-cause mortality and cardiovascular mortality. Horm. Metab. Res. 49(06), 411–417 (2017)
K.E. Therkelsen, T.M. Abraham, A. Pedley, J.M. Massaro, P. Sutherland, U. Hoffmann et al. Association between prolactin and incidence of cardiovascular risk factors in the Framingham Heart Study. J. Am. Heart Assoc. 5(2), e002640 (2016)
S.S. Inancli, A. Usluogullari, Y. Ustu, S. Caner, A.A. Tam, R. Ersoy et al. Effect of cabergoline on insulin sensitivity, inflammation, and carotid intima media thickness in patients with prolactinoma. Endocrine 44(1), 193–199 (2013)
D. Yavuz, O. Deyneli, I. Akpinar, E. Yildiz, H. Gozu, O. Sezgin et al. Endothelial function, insulin sensitivity and inflammatory markers in hyperprolactinemic pre-menopausal women. Eur. J. Endocrinol. 149(3), 187–194 (2003)
S. Byberg, J. Futtrup, M. Andreassen, J. Krogh, Metabolic effects of dopamine agonists in patients with prolactinomas: a systematic review and meta-analysis. Endocr. Connect. 8(10), 1395–1404 (2019)
K.G. Alberti, R.H. Eckel, S.M. Grundy, P.Z. Zimmet, J.I. Cleeman, K.A. Donato et al. Harmonizing the metabolic syndrome: a joint interim statement of the international diabetes federation task force on epidemiology and prevention; national heart, lung, and blood institute; American heart association; world heart federation; international atherosclerosis society; and international association for the study of obesity. Circulation 120(16), 1640–1645 (2009)
F. Azizi, D. Khalili, H. Aghajani, A. Esteghamati, F. Hosseinpanah, A. Delavari et al. Appropriate waist circumference cut-off points among Iranian adults: the first report of the Iranian National Committee of Obesity. Archives of Iranian Medicine. 13(3) (2010)
S. Melmed, F.F. Casanueva, A.R. Hoffman, D.L. Kleinberg, V.M. Montori, J.A. Schlechte et al. Diagnosis and treatment of hyperprolactinemia: an Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 96(2), 273–288 (2011)
N.A. Pala, B.A. Laway, R.A. Misgar, R.A. Dar, Metabolic abnormalities in patients with prolactinoma: response to treatment with cabergoline. Diabetol. Metab. Syndr. 7(1), 1–6 (2015)
N.A. Pala, B.A. Laway, R.A. Misgar, Z.A. Shah, T.A. Gojwari, T.A. Dar, Profile of leptin, adiponectin, and body fat in patients with hyperprolactinemia: Response to treatment with cabergoline. Indian J. Endocrinol. Metab. 20(2), 177 (2016)
R.S. Auriemma, D. De Alcubierre, R. Pirchio, R. Pivonello, A. Colao, The effects of hyperprolactinemia and its control on metabolic diseases. Expert Rev. Endocrinol. Metab. 13(2), 99–106 (2018)
R.S. Auriemma, M. Galdiero, P. Vitale, L. Granieri, F.L. Calzo, C. Salzano et al. Effect of chronic cabergoline treatment and testosterone replacement on metabolism in male patients with prolactinomas. Neuroendocrinology 101(1), 66–81 (2015)
K. Berinder, T. Nyström, C. Höybye, K. Hall, A.-L. Hulting, Insulin sensitivity and lipid profile in prolactinoma patients before and after normalization of prolactin by dopamine agonist therapy. Pituitary 14(3), 199–207 (2011)
A. Ciresi, M.C. Amato, V. Guarnotta, F. Lo Castro, C. Giordano, Higher doses of cabergoline further improve metabolic parameters in patients with prolactinoma regardless of the degree of reduction in prolactin levels. Clin. Endocrinol. 79(6), 84–852 (2013)
V. Schwetz, R. Librizzi, C. Trummer, G. Theiler, C. Stiegler, T.R. Pieber et al. Treatment of hyperprolactinaemia reduces total cholesterol and LDL in patients with prolactinomas. Metab. Brain Dis. 32(1), 155–161 (2017)
R.S. Auriemma, L. Granieri, M. Galdiero, C. Simeoli, Y. Perone, P. Vitale et al. Effect of cabergoline on metabolism in prolactinomas. Neuroendocrinology 98(4), 299–310 (2013)
A.S. Posawetz, C. Trummer, M. Pandis, F. Aberer, T.R. Pieber, B. Obermayer-Pietsch et al. Adverse body composition and lipid parameters in patients with prolactinoma: a case-control study. BMC Endocr. Disord. 21(1), 1–9 (2021)
I.B. Andersen, M. Andreassen, J. Krogh, The effect of dopamine agonists on metabolic variables in adults with type 2 diabetes: a systematic review with meta analysis and trial sequential analysis of randomized clinical trials. Diabetes, Obes. Metab. 23(1), 58–67 (2021)
E. Lamos, D. Levitt, K. Munir, A review of dopamine agonist therapy in type 2 diabetes and effects on cardio-metabolic parameters. Prim. Care Diabetes 10(1), 60–65 (2016)
S.M. Taghavi, S.S. Fatemi, H. Rokni, Cabergoline effect on blood sugar in type 2 diabetic patients with oral agent failure. Med. J. Malays. 67(4), 390–392 (2012)
A. Bahar, Z. Kashi, E. Daneshpour, O. Akha, S. Ala, Effects of cabergoline on blood glucose levels in type 2 diabetic patients: a double-blind controlled clinical trial. Medicine 95(40), e4818 (2016). https://doi.org/10.1097/MD.0000000000004818
A. Aliasgarzadeh, S. Karimiavval, J. Houshyar, M. Gojazadeh, A. Hadi, Evaluating the effect of cabergoline on glycaemic control of patients with type 2 diabetes mellitus: a randomised controlled trial. J. Clin. Diagnostic Res. 14(2), OC01–OC04 (2020)
A.C.J. Vasques, F.S. Novaes, M.D.S. de Oliveira, J.R.M. Souza, A. Yamanaka, J.C. Pareja et al. TyG index performs better than HOMA in a Brazilian population: a hyperglycemic clamp validated study. Diabetes Res. Clin. Pract. 93(3), e98–e100 (2011)
K. Zhang, Y. Chen, L. Liu, M. Lu, J. Cheng, F. Gao et al. The Triglycerides and Glucose Index rather than HOMA-IR is more associated with Hypogonadism in Chinese men. Sci. Rep. 7(1), 1–7 (2017)
F. Guerrero-Romero, L.E. Simental-Mendía, M. González-Ortiz, E. Martínez-Abundis, M.G. Ramos-Zavala, S.O. Hernández-González et al. The product of triglycerides and glucose, a simple measure of insulin sensitivity. Comparison euglycemic-hyperinsulinemic clamp. J. Clin. Endocrinol. Metab. 95(7), 3347–3351 (2010)
L.E. Simental-Mendía, M. Rodríguez-Morán, F. Guerrero-Romero, The product of fasting glucose and triglycerides as surrogate for identifying insulin resistance in apparently healthy subjects. Metab. Syndr. Relat. Disord. 6(4), 299–304 (2008)
L. Sánchez‐Íñigo, D. Navarro‐González, A. Fernández‐Montero, J. Pastrana‐Delgado, J.A. Martínez, The TyG index may predict the development of cardiovascular events. Eur. J. Clin. Invest. 46(2), 189–197 (2016)
S. Li, B. Guo, H. Chen, Z. Shi, Y. Li, Q. Tian et al. The role of the triglyceride (triacylglycerol) glucose index in the development of cardiovascular events: a retrospective cohort analysis. Sci. Rep. 9(1), 1–9 (2019)
S.H. Khan, F. Sobia, N.K. Niazi, S.M. Manzoor, N. Fazal, F. Ahmad et al. Metabolic clustering of risk factors: evaluation of Triglyceride-glucose index (TyG index) for evaluation of insulin resistace. Diabetol. Metab. Syndr. 10(1), 1–8 (2018)
F. Azizi, F. Hadaegh, F. Hosseinpanah, P. Mirmiran, A. Amouzegar, H. Abdi et al. Metabolic health in the Middle East and North Africa. Lancet Diabetes Endocrinol. 7(11), 866–879 (2019)
K.G. Alberti, R.H. Eckel, S.M. Grundy, P.Z. Zimmet, J.I. Cleeman, K.A. Donato et al. Harmonizing the metabolic syndrome: a joint interim statement of the international diabetes federation task force on epidemiology and prevention; national heart, lung, and blood institute; American heart association; world heart federation; international atherosclerosis society; and international association for the study of obesity. Circulation 120(16), 1640–1645 (2009)
Author contributions
M.K., H.A, and F.G. planned the study, researched the data, and wrote the manuscript. S.M. and A.K analyzed the data and edited the manuscript. H.S. reviewed and edited the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical Approval
This study was approved by the Ethics Committee of the Golestan University of Medical Science (Protocol no. IR.GOUMS.REC.1398.300) and was performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent to publish
Informed consent for publication was obtained from patients.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kabootari, M., Shirmohammadli, H., Golgiri, F. et al. Metabolic effects of dopamine-agonists treatment among patients with prolactinomas. Endocrine 79, 537–544 (2023). https://doi.org/10.1007/s12020-022-03238-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-022-03238-x