Abstract
Purpose
Patients with anaplastic thyroid cancer (ATC) have a very poor prognosis. Immunotherapy is a potential treatment, while the current outcome is limited which may be due to the complicated tumor microenvironment (TME). Tumor associated macrophages (TAMs) is the most abundant cell in the TME of ATC. We aimed to clarify the novel indicators based on TAM in ATC.
Methods
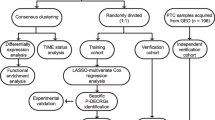
Transcriptome files were downloaded from the Gene Expression Omnibus (GEO) dataset. Weighted gene co-expression network analysis, cox regression, support vector machine, and random forest were utilized to identify TAM-related prognostic genes. Consensus clustering and principal component analysis were performed for integrated analysis. Moreover, external validation (Fudan University Shanghai Cancer Center cohort) was conducted in 23 ATC samples via immunohistochemistry.
Results
ATC patients with an abundance of TAMs had a poorer prognosis. Four TAM related genes (FZD6, RBBP8, PREX1, HSD3B7) were identified and a TAM-related prognostic index (TAMRPI) was constructed with high area under the curve (AUC). Next, high TAMRPI was related to the higher level of TAM infiltration and upregulation of several pathways, such as E2F targets, IL6-JAK-STAT3, and G2M checkpoint. Immune checkpoint TIM-3 and CSF1R were positively associated with TAMRPI, and dysfunction of T cells was increased in high TAMRPI subset. Moreover, in the external validation of protein level, strong expression of TAM related genes was related to poorer prognosis, which was further supported by time-dependent AUC analysis.
Conclusion
TAM is negatively correlated to the prognosis of ATC. FZD6, RBBP8, PREX1, and HSD3B7 are potential biomarkers of ATC.







Similar content being viewed by others
References
G. Nagaiah, A. Hossain, C.J. Mooney, J. Parmentier, S.C. Remick, Anaplastic thyroid cancer: A review of epidemiology, pathogenesis, and treatment. J. Oncol. 2011, 542358 (2011)
R.I. Haddad, W.M. Lydiatt, D.W. Ball, N.L. Busaidy, D. Byrd, G. Callender, P. Dickson, Q.Y. Duh, H. Ehya, M. Haymart, C. Hoh, J.P. Hunt, A. Iagaru, F. Kandeel, P. Kopp, D.M. Lamonica, J.C. McCaffrey, J.F. Moley, L. Parks, C.D. Raeburn, J.A. Ridge, M.D. Ringel, R.P. Scheri, J.P. Shah, R.C. Smallridge, C. Sturgeon, T.N. Wang, L.J. Wirth, K.G. Hoffmann, M. Hughes, Anaplastic Thyroid Carcinoma, Version 2.2015. J. Natl Compr. Canc Netw. 13, 1140–1150 (2015)
P. D.M., The blockade of immune checkpoints in cancer immunotherapy. Nat. Rev. Cancer 12, 252–264 (2012)
C.G. Drake, E.J. Lipson, J.R. Brahmer, Breathing new life into immunotherapy: Review of melanoma, lung and kidney cancer. Nat. Rev. Clin. Oncol. 11, 24–37 (2014)
M. Nishino, N.H. Ramaiya, H. Hatabu, F.S. Hodi, Monitoring immune-checkpoint blockade: Response evaluation and biomarker development. Nat. Rev. Clin. Oncol. 14, 655–668 (2017)
M. Ryder, R.A. Ghossein, J.C.M. Ricarte-Filho, J.A. Knauf, J.A. Fagin, Increased density of tumor-associated macrophages is associated with decreased survival in advanced thyroid cancer. Endocr.-Relat. Cancer 15, 1069–1074 (2008)
A. Mantovani, A. Sica, Macrophages, innate immunity and cancer: balance, tolerance, and diversity. Curr. Opin. Immunol. 22, 231–237 (2010)
S. Gordon, Alternative activation of macrophages. Nat. Rev. Immunol. 3, 23–35 (2003)
A.M. Newman, C.L. Liu, M.R. Green, A.J. Gentles, W. Feng, Y. Xu, C.D. Hoang, M. Diehn, A.A. Alizadeh, Robust enumeration of cell subsets from tissue expression profiles. Nat. Methods 12, 453–457 (2015)
D. Aran, Z. Hu, A.J. Butte, xCell: digitally portraying the tissue cellular heterogeneity landscape. Genome Biol. 18, 220 (2017)
L. Zhang, Z. Ren, Z. Su, Y. Liu, T. Yang, M. Cao, Y. Jiang, Y. Tang, H. Chen, W. Zhang, R. Gong, T. Wei, Y. Peng, B. Liu, W. Zhang, L. Yang, Y. Hu, Z. Li, J. Zhu, H. Xu, Y. Shu, H. Luo, Novel recurrent altered genes in Chinese patients with anaplastic thyroid cancer. J. Clin. Endocrinol. Metabol. 106, 988–998 (2021)
I. Abe, A.K. Lam, Anaplastic thyroid carcinoma: Updates on WHO classification, clinicopathological features and staging. Histol Histopathol. 36, 239–248 (2020)
A.M. Mirabile, L. Biafora, G. Giordano, G. Arrigoni, M.G. Cangi, I. Dell'Oca, D. Lira Luce, D. Di Santo, A. Galli, M. Tulli, R. Mellone, D. Valsecchi, V. Gregorc, M. Bussi, Uncommon Site of Metastasis and Prolonged Survival in Patients with Anaplastic Thyroid Carcinoma: A Systematic Review of the Literature. Cancers (Basel) 12, 2585 (2020)
D. Hanahan, A. Robert, Weinberg, hallmarks of cancer: The next generation. Cell 144, 646–674 (2011)
D. Chen, X. Zhang, Z. Li, B. Zhu, Metabolic regulatory crosstalk between tumor microenvironment and tumor-associated macrophages. Theranostics 11, 1016–1030 (2021)
P.J. Murray, Macrophage polarization. Annu Rev. Physiol. 79, 541–566 (2017)
X.-T. Fu, Z. Dai, K. Song, Z.-J. Zhang, Z.-J. Zhou, S.-L. Zhou, Y.-M. Zhao, Y.-S. Xiao, Q.-M. Sun, Z.-B. Ding, J. Fan, Macrophage-secreted IL-8 induces epithelial-mesenchymal transition in hepatocellular carcinoma cells by activating the JAK2/STAT3/Snail pathway. Int J. Oncol. 46, 587–596 (2015)
M.L. Pinto, E. Rios, C. Durães, R. Ribeiro, J.C. Machado, A. Mantovani, M.A. Barbosa, F. Carneiro, M.J. Oliveira, The Two Faces of Tumor-Associated Macrophages and Their Clinical Significance in Colorectal Cancer. Front Immunol. 10, 1875 (2019)
C.M. Schürch, M.A. Roelli, S. Forster, M.-H. Wasmer, F. Brühl, R.S. Maire, S. Di Pancrazio, M.-D. Ruepp, R. Giger, A. Perren, A.M. Schmitt, P. Krebs, R.-P. Charles, M.S. Dettmer, Targeting CD47 in anaplastic thyroid carcinoma enhances tumor phagocytosis by macrophages and is a promising therapeutic strategy. Thyroid 29, 979–992 (2019)
A. Mantovani, S. Sozzani, M. Locati, P. Allavena, A. Sica, Macrophage polarization: Tumor-associated macrophages as a paradigm for polarized M2 mononuclear phagocytes. Trends Immunol. 23, 549–555 (2002)
H. Ye, Q. Zhou, S. Zheng, G. Li, Q. Lin, L. Wei, Z. Fu, B. Zhang, Y. Liu, Z. Li, R. Chen, Tumor-associated macrophages promote progression and the Warburg effect via CCL18/NF-kB/VCAM-1 pathway in pancreatic ductal adenocarcinoma. Cell Death Dis. 9, 453 (2018)
M. Shen, Y. Chen, L. Xu, R. Zhu, X. Xue, Y. Tsai, P.C. Keng, S.O. Lee, Y. Chen, Increased infiltration of macrophages to radioresistant lung cancer cells contributes to the development of the additional resistance of tumor cells to the cytotoxic effects of NK cells. Int J. Oncol. 53, 317–328 (2018)
Y. Chen, Y. Song, W. Du, L. Gong, H. Chang, Z. Zou, Tumor-associated macrophages: An accomplice in solid tumor progression. J. Biomed. Sci. 26, 78 (2019)
D.I. Kim, E. Kim, Y.A. Kim, S.W. Cho, J.A. Lim, Y.J. Park, Macrophage densities correlated with CXC chemokine receptor 4 expression and related with poor survival in anaplastic thyroid cancer. Endocrinol. Metab. (Seoul.) 31, 469–475 (2016)
J. Espinal-Enríquez, S. Muñoz-Montero, I. Imaz-Rosshandler, A. Huerta-Verde, C. Mejía, E. Hernández-Lemus, Genome-wide expression analysis suggests a crucial role of dysregulation of matrix metalloproteinases pathway in undifferentiated thyroid carcinoma. BMC Genomics 16, 207 (2015)
S. Ahn, T.H. Kim, S.W. Kim, C.S. Ki, H.W. Jang, J.S. Kim, J.H. Kim, J.H. Choe, J.H. Shin, S.Y. Hahn, Y.L. Oh, J.H. C., Comprehensive screening for PD-L1 expression in thyroid cancer. Endocr. Relat. Cancer 24, 97–106 (2017)
M. Camus, M. Tosolini, B. Mlecnik, F. Pages, A. Kirilovsky, A. Berger, A. Costes, G. Bindea, P. Charoentong, P. Bruneval, Z. Trajanoski, W.H. Fridman, J. Galon, Coordination of intratumoral immune reaction and human colorectal cancer recurrence. Cancer Res 69, 2685–2693 (2009)
R. Giannini, S. Moretti, C. Ugolini, E. Macerola, E. Menicali, N. Nucci, S. Morelli, R. Colella, M. Mandarano, A. Sidoni, M. Panfili, F. Basolo, E. Puxeddu, Immune profiling of thyroid carcinomas suggests the existence of two major phenotypes: An ATC-Like and a PDTC-Like. J. Clin. Endocrinol. Metab. 104, 3557–3575 (2019)
B. Caillou, M. Talbot, U. Weyemi, C. Pioche-Durieu, A. Al Ghuzlan, J.M. Bidart, S. Chouaib, M. Schlumberger, C. Dupuy, Tumor-associated macrophages (TAMs) form an interconnected cellular supportive network in anaplastic thyroid carcinoma. PLoS One 6, e22567 (2011)
P.A. Oldenborg, A. Zheleznyak, Y.F. Fang, C.F. Lagenaur, H.D. Gresham, F.P. Lindberg, Role of CD47 as a marker of self on red blood cells. Science 288, 2051–2054 (2000)
C.M. Schürch, S. Forster, F. Brühl, S.H. Yang, E. Felley-Bosco, E. Hewer, The “don’t eat me” signal CD47 is a novel diagnostic biomarker and potential therapeutic target for diffuse malignant mesothelioma. Oncoimmunology 7, e1373235 (2017)
Acknowledgements
We thank the colleagues for their useful comments for the improvement and modification of the study.
Author contributions
Conceptualization: Y.L.; methodology: Y.L. and Y.Y.; analysis of data: Y.L., W.X., and B.M.; writing: Y.L.; supervision, project administration: T.L. and Y.W.
Funding
This work was supported by the National Natural Science Foundation of China (82072951).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares no competing interests.
Ethical approval
The research ethics committee of FUSCC has confirmed that no ethical approval is required (050432-4-1911D).
Informed consent
Informed consent was obtained from all individual participants.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Luo, Y., Yang, YC., Ma, B. et al. Integrated analysis of novel macrophage related signature in anaplastic thyroid cancer. Endocrine 78, 517–530 (2022). https://doi.org/10.1007/s12020-022-03179-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-022-03179-5