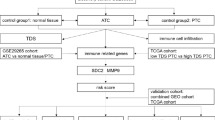
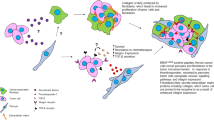
Abstract
Objective
The aim of this study was to construct a collagen-related prognostic model for thyroid cancer and to investigate prognostic value of collagen family genes for thyroid cancer.
Methods
A LASSO Cox regression model for thyroid cancer was developed based on the expression profiles of collagen-related genes. Kaplan–Meier survival analysis was performed for high and low risk groups. The ROC method was used to assess its predictive performance. Predictive independence was verified by multivariate Cox regression analysis. The relationship between this feature and immune cell infiltration was analyzed by tumor microenvironment. COL18A1 was validated by immunohistochemistry and RT-PCR in thyroid cancer tissues. The effect of COL18A1 on cell proliferation, migration and invasion ability of tumor cells were further valuated by CCK-8 assay and transwell assay. The effect of COL18A1 on the immune escape ability of tumor cells was further valuated by cytotoxicity assays.
Results
A model including 4 collagen family genes was developed to predict thyroid cancer prognosis. Patients with high-risk score had a poorer prognosis than those with low-risk scores for 1-, 2-, 3-, and 5- year survival. The model independently predicted prognosis after adjusting for other prognostic factors. A nomogram combining risk score and age was constructed with high sensitivity and specificity. This feature was significantly associated with immune cell infiltration. COL18A1 was aberrantly over-expressed in thyroid cancer compared with control tissues and significantly increased proliferative capacity, migration capacity, invasion capacity, and immune escape ability of tumor cells.
Conclusion
Our findings establish a signature associated with collagen family genes that can be a promising tool to predict the prognosis of thyroid cancer. High COL18A1 expression significantly correlates with the poor prognosis of patients and enhances the immune escape ability of tumor cells.







Similar content being viewed by others
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
C.D. Seib, J.A. Sosa, Evolving understanding of the epidemiology of thyroid cancer. Endocrinol. Metab. Clin. North Am., 48(1), 23-+ (2019)
R.C. Mould et al. Immune responses in the thyroid cancer microenvironment: making immunotherapy a possible mission. Endocr.-Relat. Cancer 24(12), T311–T329 (2017)
Y. Bai, K. Kakudo, Y. Li, Subclassification of non-solid-type papillary thyroid carcinoma identification of high-risk group in common type (vol 99, pg 1908, 2008). Cancer Sci. 99(12), 2548 (2008)
T.R. Cox, J.T. Erler, Remodeling and homeostasis of the extracellular matrix: implications for fibrotic diseases and cancer. Dis. Models Mechanisms 4(2), 165–178 (2011)
P. Friedl, K. Wolf, Tube travel: The role of proteases in individual and collective a cancer cell invasion. Cancer Res. 68(18), 7247–7249 (2008)
P.G. Gritsenko, O. Ilina, P. Friedl, Interstitial guidance of cancer invasion. J. Pathol. 226(2), 185–199 (2012)
R.V. Iozzo, M.A. Gubbiotti, Extracellular matrix: The driving force of mammalian diseases. Matrix Biol. 71–72, 1–9 (2018)
N.K. Karamanos et al. Matrix modeling and remodeling: A biological interplay regulating tissue homeostasis and diseases. Matrix Biol. 75–76, 1–11 (2019)
N.K. Karamanos et al. Proteoglycan chemical diversity drives multifunctional cell regulation and therapeutics. Chem. Rev. 118(18), 9152–9232 (2018)
N. Afratis et al. Glycosaminoglycans: key players in cancer cell biology and treatment. Febs J. 279(7), 1177–1197 (2012)
A.D. Theocharis, D. Manou, N.K. Karamanos, The extracellular matrix as a multitasking player in disease. Febs J. 286(15), 2830–2869 (2019)
P.P. Provenzano et al. Collagen density promotes mammary tumor initiation and progression. Bmc Med. 6, 11 (2008)
W.J. Han et al. Oriented collagen fibers direct tumor cell intravasation. Proc. Natl Acad. Sci. USA 113(40), 11208–11213 (2016)
M.W. Conklin et al. Collagen alignment as a predictor of recurrence after ductal carcinoma in situ. Cancer Epidemiol. Biomark. Prev. 27(2), 138–145 (2018)
M.W. Conklin et al. Aligned collagen is a prognostic signature for survival in human breast carcinoma. Am. J. Pathol. 178(3), 1221–1232 (2011)
K.B. Pointer et al. Association of collagen architecture with glioblastoma patient survival. J. Neurosurg. 126(6), 1812–1821 (2017)
M.F. Penet et al. Structure and function of a prostate cancer dissemination-permissive extracellular matrix. Clin. Cancer Res. 23(9), 2245–2254 (2017)
R. Hristu et al. Quantitative second harmonic generation microscopy for the structural characterization of capsular collagen in thyroid neoplasms. Biomed. Opt. Express 9(8), 3923–3936 (2018)
T. Li et al. CUL4 E3 ligase regulates the proliferation and apoptosis of lung squamous cell carcinoma and small cell lung carcinoma. Cancer Biol. Med 17(2), 357–370 (2020)
K.M. Mak, C.Y.M. Png, D.J. Lee, Type V collagen in health, disease, and fibrosis. Anat. Rec.-Adv. Integr. Anat. Evolut. Biol. 299(5), 613–629 (2016)
T. Nardo et al. Adhesion to type V collagen enhances staurosporine-induced apoptosis of adrenocortical cancer cells. Tumor Biol. 35(10), 9949–9955 (2014)
S.H. Barsky et al. Increased content of Type V Collagen in desmoplasia of human breast carcinoma. Am. J. Pathol. 108(3), 276–283 (1982)
H. Fischer et al. Colorectal carcinogenesis is associated with stromal expression of COL11A1 and COL5A2. Carcinogenesis 22(6), 875–878 (2001)
S. Berchtold et al. Collagen type V promotes the malignant phenotype of pancreatic ductal adenocarcinoma. Cancer Lett. 356(2), 721–732 (2015)
W.B. Liu et al. COL5A1 may contribute the metastasis of lung adenocarcinoma. Gene 665, 57–66 (2018)
G. Feng et al. Overexpression of COL5A1 promotes tumor progression and metastasis and correlates with poor survival of patients with clear cell renal cell carcinoma. Cancer Manag Res 11, 1263–1274 (2019)
W. Ren et al. Overexpression of collagen type V alpha1 chain in human breast invasive ductal carcinoma is mediated by TGF-beta1. Int J. Oncol. 52(5), 1694–1704 (2018)
M.S. O’Reilly et al. Endostatin: an endogenous inhibitor of angiogenesis and tumor growth. Cell 88(2), 277–285 (1997)
N. Yamaguchi et al. Endostatin inhibits VEGF-induced endothelial cell migration and tumor growth independently of zinc binding. EMBO J. 18(16), 4414–4423 (1999)
M. Dhanabal et al. Endostatin induces endothelial cell apoptosis. J. Biol. Chem. 274(17), 11721–11726 (1999)
D.K. Skovseth et al. Endostatin dramatically inhibits endothelial cell migration, vascular morphogenesis, and perivascular cell recruitment in vivo. Blood 105(3), 1044–1051 (2005)
G. Schuch et al. Endostatin inhibits the vascular endothelial growth factor-induced mobilization of endothelial progenitor cells. Cancer Res 63(23), 8345–8350 (2003)
P. Hagg et al. Type XIII collagen is identified as a plasma membrane protein. J. Biol. Chem. 273(25), 15590–15597 (1998)
P. Hagg et al. Type XIII collagen: a novel cell adhesion component present in a range of cell-matrix adhesions and in the intercalated discs between cardiac muscle cells. Matrix Biol. 19(8), 727–742 (2001)
M. Yi, J.E. Schnitzer, Impaired tumor growth, metastasis, angiogenesis and wound healing in annexin A1-null mice. Proc. Natl Acad. Sci. USA 106(42), 17886–17891 (2009)
S. Nallanthighal, J.P. Heiserman, and D.J. Cheon, Collagen Type XI Alpha 1 (COL11A1): A Novel Biomarker and a Key Player in Cancer. Cancers (Basel), 13(5), (2021)
F. Vazquez-Villa et al. COL11A1/(pro)collagen 11A1 expression is a remarkable biomarker of human invasive carcinoma-associated stromal cells and carcinoma progression. Tumor Biol. 36(4), 2213–2222 (2015)
Z. Raglow, S.M. Thomas, Tumor matrix protein collagen XI alpha 1 in cancer. Cancer Lett. 357(2), 448–453 (2015)
P.M. Angel, S.C. Zambrzycki, Predictive value of collagen in cancer. Adv. Cancer Res 154, 15–45 (2022)
A.C. Martins Cavaco et al. Collagen biology making inroads into prognosis and treatment of cancer progression and metastasis. Cancer Metastasis Rev. 39(3), 603–623 (2020)
X.P. Shen et al. Patient age-associated mortality risk is differentiated by BRAF V600E status in papillary thyroid cancer. J. Clin. Oncol. 36(5), 438-+ (2018)
T.R. Cox et al. The hypoxic cancer secretome induces pre-metastatic bone lesions through lysyl oxidase. Nature 522(7554), 106–110 (2015)
S. Kauppila et al. Aberrant type I and type III collagen gene expression in human breast cancer in vivo. J. Pathol. 186(3), 262–268 (1998)
M.A. Wozniak et al. ROCK-generated contractility regulates breast epithelial cell differentiation in response to the physical properties of a three-dimensional collagen matrix. J. Cell Biol. 163(3), 583–595 (2003)
G. Kaplan, G. Gaudernack, In vitro differentiation of human monocytes. Differences in monocyte phenotypes induced by cultivation on glass or on collagen. J. Exp. Med 156(4), 1101–14. (1982)
N.G. Nicolson et al. Immune cell infiltrate-associated dysregulation of DNA repair machinery may predispose to papillary thyroid carcinogenesis. Surgery 167(1), 66–72 (2020)
Funding
This work was partially supported by grants from National Natural Science Foundation of China (Grant No. 81872079), Clinical Research Project of the Second Hospital of Tianjin Medical University (Grant No. KY2019K086-02).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Data collection and analysis were performed by J.N., W.G., and N.J. The experiments in vitro were performed by J.N. and Y.C. The first draft of the manuscript was written by J.N. and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval
The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. And this study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of the Second Hospital of Tianjin Medical University.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Niu, J., Guo, W., Chen, YZ. et al. Identification of the collagen family as prognostic biomarkers in papillary thyroid carcinoma. Endocrine 78, 491–506 (2022). https://doi.org/10.1007/s12020-022-03175-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-022-03175-9