Abstract
Aim
This study aimed to determine the association of decreased muscle mass with reduced bone mineral density in patients with Graves’ disease.
Methods
A total of 758 patients with Graves’ disease at diagnosis (mean age 41.2 years) were enrolled for a cross-sectional study; of these, 287 were enrolled for a cohort study with a median follow-up of 24 months. Meanwhile, 1164 age- and sex-matched healthy controls were recruited. All participants underwent dual-energy x-ray absorptiometry and muscle mass index (ASMI) measurements. The changes in ASMI and bone mineral density (BMD) were calculated from the measurements made at a gap of 2 years.
Results
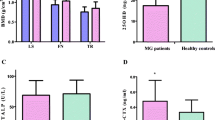
The BMD of patients with Graves’ disease was still significantly lower after normalizing serum thyroid hormone levels compared with that of healthy controls. ASMI positively correlated with BMD in patients with Graves’ disease (lumbar BMD, r = 0.210; femoral neck BMD, r = 0.259; hip BMD, r = 0.235; P < 0.001), and this relationship persisted after successful anti-thyroid therapy (lumbar BMD, r = 0.169; femoral neck BMD, r = 0.281; hip BMD, r = 0.394; P < 0.001). Low muscle mass was associated with low BMD (OR, 1.436; 95% CI, 1.026–2.010). Improving the muscle mass led to changes in the bone mass of the femoral neck (OR, 0.420; 95% CI, 0.194–0.911) and hip (OR, 0.217; 95% CI, 0.092–0.511) during the follow-up. However, this phenomenon was not observed in lumbar and bone turnover markers.
Conclusions
The recovery of bone mass might be related to the recovery of the muscle mass. Patients with Graves’ disease should be helped to regain their muscle mass and thus accelerate the recovery of bone mass while administering anti-thyroid therapy.




Similar content being viewed by others
References
D.S. Ross, H.B. Burch, D.S. Cooper, M.C. Greenlee, P. Laurberg, A.L. Maia et al. 2016 American Thyroid Association Guidelines for Diagnosis and Management of Hyperthyroidism and Other Causes of Thyrotoxicosis. Thyroid. Off. J. Am. Thyroid. Assoc. 26, 1343–1421 (2016). https://doi.org/10.1089/thy.2016.0229
A.P. Delitala, A. Scuteri, C. Doria, Thyroid hormone diseases and osteoporosis. J. Clin. Med. 2020;9. https://doi.org/10.3390/jcm9041034
P. Vestergaard, L. Mosekilde, Hyperthyroidism, bone mineral, and fracture risk-a meta-analysis. Thyroid. Off. J. Am. Thyroid. Assoc. 13, 585–593 (2003). https://doi.org/10.1089/105072503322238854
K. Takedani, M. Notsu, Graves’ disease and vertebral fracture: possible pathogenic link in postmenopausal women. Clin. Endocrinol. 93, 204–211 (2020). https://doi.org/10.1111/cen.14208
S.R. Cummings, M.C. Nevitt, W.S. Browner, K. Stone, K.M. Fox, K.E. Ensrud et al. Risk factors for hip fracture in white women. Study of Osteoporotic Fractures Research Group. N. Engl. J. Med. 332, 767–773 (1995). https://doi.org/10.1056/nejm199503233321202
G.R. Williams, J.H.D. Bassett, Thyroid diseases and bone health. J. Endocrinological Investig. 41, 99–109 (2018). https://doi.org/10.1007/s40618-017-0753-4
G.C. Isaia, C. Roggia, D. Gola, M.D. Stefano, G. Gallone, G. Aimo et al. Bone turnover in hyperthyroidism before and after thyrostatic management. J. Endocrinological Investig. 23, 727–731 (2000). https://doi.org/10.1007/bf03345061
C. Szlejf, C.K. Suemoto, C. Janovsky, Thyroid function and sarcopenia: results from the ELSA-Brasil study. J. Am. Geriatrics Soc. 68, 1545–1553 (2020). https://doi.org/10.1111/jgs.16416
K. Agbaht, M.F. Erdogan, R. Emral, N. Baskal, S. Gullu, Circulating glucagon to ghrelin ratio as a determinant of insulin resistance in hyperthyroidism. Endocrine 45, 106–113 (2014). https://doi.org/10.1007/s12020-013-9951-9
L. Cianferotti, M.L. Brandi, Muscle-bone interactions: basic and clinical aspects. Endocrine 45, 165–177 (2014). https://doi.org/10.1007/s12020-013-0026-8
M.D. Brennan, C. Powell, K.R. Kaufman, P.C. Sun, R.S. Bahn, K.S. Nair, The impact of overt and subclinical hyperthyroidism on skeletal muscle. Thyroid. Off. J. Am. Thyroid. Assoc. 16, 375–380 (2006). https://doi.org/10.1089/thy.2006.16.375
A.J. Cruz-Jentoft, G. Bahat, J. Bauer, Y. Boirie, O. Bruyère, T. Cederholm et al. Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing 48, 16–31 (2019). https://doi.org/10.1093/ageing/afy169
F. Cosman, S.J. de Beur, M.S. LeBoff, E.M. Lewiecki, B. Tanner, S. Randall et al. Clinician’s guide to prevention and treatment of osteoporosis. Osteoporos. Int. 25, 2359–2381 (2014). https://doi.org/10.1007/s00198-014-2794-2
S.M. de la Monte, J.J. Kril, Human alcohol-related neuropathology. Acta Neuropathologica 127, 71–90 (2014). https://doi.org/10.1007/s00401-013-1233-3
F. Du, D. Birong, H. Changquan, W. Hongmei, Z. Yanling, Z. Wen et al. Association of osteoporotic fracture with smoking, alcohol consumption, tea consumption and exercise among Chinese nonagenarians/centenarians. J. Nutr. Health Aging 15, 327–331 (2011). https://doi.org/10.1007/s12603-010-0270-z
G.J. Cho, J.H. Shin, K.W. Yi, H.T. Park, T. Kim, J.Y. Hur et al. Serum ferritin levels are associated with metabolic syndrome in postmenopausal women but not in premenopausal women. Menopause 18, 1120–1124 (2011). https://doi.org/10.1097/gme.0b013e318217e172
J.S. Meengs, L.S. Roe, B.J. Rolls, Vegetable variety: an effective strategy to increase vegetable intake in adults. J. Acad. Nutr. Dietetics 112, 1211–1215 (2012). https://doi.org/10.1016/j.jand.2012.05.013
E.Z. Movassagh, A.D.G. Baxter-Jones, S. Kontulainen, S. Whiting, M. Szafron, H. Vatanparast, Vegetarian-style dietary pattern during adolescence has long-term positive impact on bone from adolescence to young adulthood: a longitudinal study. Nutr J. 17, 36 (2018). https://doi.org/10.1186/s12937-018-0324-3
H.R. Kim, Y.S. Hong, S.H. Park, J.H. Ju, K.Y. Kang, Low bone mineral density predicts the formation of new syndesmophytes in patients with axial spondyloarthritis. Arthritis Res. Ther. 20, 231 (2018). https://doi.org/10.1186/s13075-018-1731-8
I.K. Blom-Høgestøl, S. Hewitt, M. Chahal-Kummen, C. Brunborg, H.L. Gulseth, J.A. Kristinsson et al. Bone metabolism, bone mineral density and low-energy fractures 10 years after Roux-en-Y gastric bypass. Bone 127, 436–445 (2019). https://doi.org/10.1016/j.bone.2019.07.014
R.A. Fielding, B. Vellas, W.J. Evans, S. Bhasin, J.E. Morley, A.B. Newman et al. Sarcopenia: an undiagnosed condition in older adults. Current consensus definition: prevalence, etiology, and consequences. International working group on sarcopenia. J. Am. Med. Dir. Assoc. 12, 249–256 (2011). https://doi.org/10.1016/j.jamda.2011.01.003
H.K. Kim, T. Suzuki, K. Saito, H. Yoshida, H. Kobayashi, H. Kato et al. Effects of exercise and amino acid supplementation on body composition and physical function in community-dwelling elderly Japanese sarcopenic women: a randomized controlled trial. J. Am. Geriatrics Soc. 60, 16–23 (2012). https://doi.org/10.1111/j.1532-5415.2011.03776.x
Y. Zhang, X. Shen, L. He, F. Zhao, S. Yan, Association of sarcopenia and muscle mass with both peripheral neuropathy and nerve function in patients with type 2 diabetes. Diabetes Res. Clin. Pract. 162, 108096 (2020). https://doi.org/10.1016/j.diabres.2020.108096
M.L. Bouxsein, R. Eastell, L.Y. Lui, L.A. Wu, A.E. de Papp, A. Grauer et al. Change in bone density and reduction in fracture risk: a meta-regression of published trials. J. Bone Miner. Res. 34, 632–642 (2019). https://doi.org/10.1002/jbmr.3641
R. Eastell, P. Szulc, Use of bone turnover markers in postmenopausal osteoporosis. lancet Diabetes Endocrinol. 5, 908–923 (2017). https://doi.org/10.1016/s2213-8587(17)30184-5
K. Maratova, O. Soucek, J. Matyskova, Z. Hlavka, L. Petruzelkova, B. Obermannova et al. Muscle functions and bone strength are impaired in adolescents with type 1 diabetes. Bone 106, 22–27 (2018). https://doi.org/10.1016/j.bone.2017.10.005
M. Hayashi, K. Abe, M. Fujita, K. Okai, A. Takahashi, H. Ohira, Association between sarcopenia and osteoporosis in chronic liver disease. Hepatol. Res. 48, 893–904 (2018). https://doi.org/10.1111/hepr.13192
Apostu D., Lucaciu O., Oltean-Dan D., Mureșan A.D., Moisescu-Pop C., Maxim A., et al. The influence of thyroid pathology on osteoporosis and fracture risk: a review. Diagnostics. 2020;10: https://doi.org/10.3390/diagnostics10030149
T. Grofte, T. Wolthers, N. Moller, J.O. Jorgensen, A. Flyvbjerg, H. Orskov et al. Moderate hyperthyroidism reduces liver amino nitrogen conversion, muscle nitrogen contents and overall nitrogen balance in rats. Eur. J. Clin. Investig. 27, 85–92 (1997). https://doi.org/10.1046/j.1365-2362.1997.00101.x
P. Iglesias, J.J. Díez, Influence of thyroid dysfunction on serum concentrations of adipocytokines. Cytokine 40, 61–70 (2007). https://doi.org/10.1016/j.cyto.2007.10.001
N. Kubota, W. Yano, T. Kubota, T. Yamauchi, S. Itoh, H. Kumagai et al. Adiponectin stimulates AMP-activated protein kinase in the hypothalamus and increases food intake. Cell Metab. 6, 55–68 (2007). https://doi.org/10.1016/j.cmet.2007.06.003
C. Di Munno, R.A. Busiello, J. Calonne, A.M. Salzano, J. Miles-Chan, A. Scaloni et al. Adaptive thermogenesis driving catch-up fat is associated with increased muscle type 3 and decreased hepatic type 1 iodothyronine deiodinase activities: a functional and proteomic study. Front. Endocrinol. 12, 631176 (2021). https://doi.org/10.3389/fendo.2021.631176
S. Judex, K.J. Carlson, Is bone’s response to mechanical signals dominated by gravitational loading? Med. Sci. Sports Exerc. 41, 2037–2043 (2009). https://doi.org/10.1249/MSS.0b013e3181a8c6e5
R.J. Wetzsteon, B.S. Zemel, J. Shults, K.M. Howard, L.W. Kibe, M.B. Leonard, Mechanical loads and cortical bone geometry in healthy children and young adults. Bone 48, 1103–1108 (2011). https://doi.org/10.1016/j.bone.2011.01.005
G. Karsenty, E.N. Olson, Bone and muscle endocrine functions: unexpected paradigms of inter-organ communication. Cell 164, 1248–1256 (2016). https://doi.org/10.1016/j.cell.2016.02.043
L.H. Bogl, A. Latvala, J. Kaprio, O. Sovijärvi, A. Rissanen, K.H. Pietiläinen, An investigation into the relationship between soft tissue body composition and bone mineral density in a young adult twin sample. J. Bone Miner. Res. 26, 79–87 (2011). https://doi.org/10.1002/jbmr.192
G. Battafarano, M. Rossi, F. Marampon, S. Minisola, A. Del Fattore, Bone control of muscle function. Int. J. Mol. Sci. 2020;21, https://doi.org/10.1016/bs.acc.2019.07.010
K. Tsang, H. Liu, Y. Yang, J.F. Charles, J. Ermann, Defective circadian control in mesenchymal cells reduces adult bone mass in mice by promoting osteoclast function. Bone 121, 172–180 (2019). https://doi.org/10.1016/j.bone.2019.01.016
S.C. Manolagas, R.L. Jilka, Bone marrow, cytokines, and bone remodeling. Emerging insights into the pathophysiology of osteoporosis. N. Engl. J. Med. 332, 305–311 (1995). https://doi.org/10.1056/nejm199502023320506
K.M. Kim, E.Y. Lee, S. Lim, H.C. Jang, C.O. Kim, Favorable effects of skeletal muscle on bone are distinguished according to gender and skeletal sites. Osteoporos. Sarcopenia 3, 32–36 (2017). https://doi.org/10.1016/j.afos.2016.11.001
B.J. Ausk, P. Huber, S. Srinivasan, S.D. Bain, R.Y. Kwon, E.A. McNamara et al. Metaphyseal and diaphyseal bone loss in the tibia following transient muscle paralysis are spatiotemporally distinct resorption events. Bone 57, 413–422 (2013). https://doi.org/10.1016/j.bone.2013.09.009
M.K. Kwak, B.J. Kim, J.S. Kim, S.H. Lee, J.M. Koh, The local and systemic interactions between muscle and bone in postmenopausal Korean women. Calcif. Tissue Int. 105, 373–382 (2019). https://doi.org/10.1007/s00223-019-00585-2
C. Suetta, S.P. Magnusson, A. Rosted, P. Aagaard, A.K. Jakobsen, L.H. Larsen et al. Resistance training in the early postoperative phase reduces hospitalization and leads to muscle hypertrophy in elderly hip surgery patients-a controlled, randomized study. J. Am. Geriatrics Soc. 52, 2016–2022 (2004). https://doi.org/10.1111/j.1532-5415.2004.52557.x
J.T. Cramer, A.J. Cruz-Jentoft, F. Landi, M. Hickson, M. Zamboni, S.L. Pereira et al. Impacts of high-protein oral nutritional supplements among malnourished men and women with sarcopenia: a multicenter, randomized, double-blinded, controlled trial. J. Am. Med. Dir. Assoc. 17, 1044–1055 (2016). https://doi.org/10.1016/j.jamda.2016.08.009
R.W. Lau, L.R. Liao, F. Yu, T. Teo, R.C. Chung, M.Y. Pang, The effects of whole body vibration therapy on bone mineral density and leg muscle strength in older adults: a systematic review and meta-analysis. Clin. Rehabil. 25, 975–988 (2011). https://doi.org/10.1177/0269215511405078
Acknowledgements
We are grateful to the patients for their help and willingness to participate in the study.
Funding
This work was financially supported by Joint Funds for the Innovation of Science and Technology, Fujian Province (No. 2019Y9110), Youth Scientific Research Project of Fujian Provincial Health Commission (No. 2020QNA057) and Natural Science Foundation of Fujian Province (No. 2018J01171) financially supported this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008 (5).
Informed consent
Informed consent was obtained from all patients for being included in the study. This study was approved by the Ethics Committee of the First Affiliated Hospital of Fujian Medical University, and the participants gave informed consent.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, Y., Ke, Y., Huang, L. et al. Association of decreased muscle mass with reduced bone mineral density in patients with Graves’ disease. Endocrine 75, 916–926 (2022). https://doi.org/10.1007/s12020-021-02960-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-021-02960-2