Abstract
Purpose
Neuroendocrine neoplasms (NENs) differ in their biological behavior and growth potential in a way that can be predicted using histological classification and grading systems. A subset of pancreatic NENs (pNENs) may develop a more aggressive phenotype during the course of the disease, associated with an increase in the Ki-67 proliferation index (PI). The purpose of the study was to present the clinical characteristics of these patients.
Methods
Using re-biopsy of growing lesions, we investigated the increase in Ki-67 PI sufficient to change initial grading (G).
Results
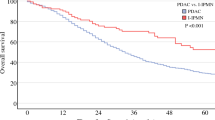
Of 264 patients with well differentiated (WD) pNENs who showed progressive disease during follow-up, 15 (6%) exhibited an increase in Ki-67 PI at a median time 36.8 (9.3–255.8) months. All neoplasms had WD-morphology: five had G1 (Ki-67 median value 1%), nine G2 (median value 5%), one G3 (25%) grades. Upon change of Ki-67 PI, 3 patients had G2 (8%) and 12 G3 (57.5%) NENs, while all retained their WD-morphology. At last follow-up, eight patients were alive with a median overall survival (OS) of 52.5 (9.5–264.3) months. Μedian OS was shorter in patients who had a change in Ki-67 PI before 36 months compared to those who had a change of Ki-67 PI at a later stage (27.5 95%CI: 11.88–43.06 vs. 120.87 95%CI: 96.05–145.69; log-rank p = 0.018).
Conclusions
During the course of their disease, 6% patients with progressive pNENs develop an increase in Ki-67 PI resulting in an increase in grading status while maintaining their morphology. This process is associated with worse OS when it occurs at an early stage.



Similar content being viewed by others
Availability of data and material
Available on request.
Code availability
Available on request.
References
A. Dasari, C. Shen, D. Halperin, B. Zhao, S. Zhou, Y. Xu, T. Shih, J.C. Yao, Trends in the incidence, prevalence, and survival outcomes in patients with neuroendocrine tumors in the United States. AMA Oncol. 3, 1335–1342 (2017)
J.C. Yao, M. Hassan, A. Phan, C. Dagohoy, C. Leary, J.E. Mares, E.K. Abdalla, J.B. Fleming, J.N. Vauthey, A. Rashid, D.B. Evans, One hundred years after “carcinoid”: epidemiology of and prognostic factors for neuroendocrine tumors in 35,825 cases in the United States. J. Clin. Oncol. 26, 3063–3072 (2008)
S. Singh, J. Hallet, C. Rowsell, C.H. Law, Variability of Ki67 labeling index in multiple neuroendocrine tumors specimens over the course of the disease. Eur. J. Surg. Oncol. 40, 1517–1522 (2014)
S. Richards-Taylor, C. Tilley, E. Jaynes, H. Hu, T. Armstrong, N.W. Pearce, R. Plant, J. Cave, Clinically significant differences in Ki-67 proliferation index between primary and metastases in resected pancreatic neuroendocrine tumors. Pancreas 46, 1354–1358 (2016)
C. Shi, R.S. Gonzalez, Z. Zhao, T. Koyama, T.C. Cornish, K.R. Hande, R. Walker, M. Sandler, J. Berlin, E.H. Liu, Liver metastases of small intestine neuroendocrine tumors: Ki-67 heterogeneity and World Health Organization grade discordance with primary tumors. Am. J. Clin. Pathol. 143, 398–404 (2015)
H.C. Miller, P. Drymousis, R. Flora, R. Goldin, D. Spalding, A. Frilling, Role of Ki-67 proliferation index in the assessment of patients with neuroendocrine neoplasias regarding the stage of disease. World J. Surg. 38, 1353–1361 (2014)
D. Paul, V. Ostwal, S. Bose, S. Basu, S. Gupta, Personalized treatment approach to gastroenteropancreatic neuroendocrine tumors: a medical oncologist’s perspective. Eur. J. Gastroenterol. Hepatol. 28, 985–990 (2016)
L. Carideo, D. Prosperi, F. Panzuto, L. Magi, M.S. Pratesi, M. Rinzivillo, B. Annibale, A. Signore, Role of combined [68Ga]Ga-DOTA-SST analogues and [18F]FDG PET/CT in the management of GEP-NENs: a systematic review. J. Clin. Med. 8, 1032 (2019)
D.L.H. Chan, N. Pavlakis, G.P. Schembri, E.J. Bernard, E. Hsiao, A. Hayes, T. Barnes, C. Diakos, M. Khasraw, J. Samra, E. Eslick, P.J. Roach, S.J. Clarke, D.L. Bailey, Dual somatostatin receptor/FDG PET/CT imaging in metastatic neuroendocrine tumours: proposal for a novel grading scheme with prognostic significance. Theranostics 7, 1149–1158 (2017)
O.A. Hentic, J.A. Cros, V.A. Rebours, M.A. Zappa, N.A. Muller, F.A. Maire, P.A. Levy, P.A. Hammel, A.A. Couvelard, P.A. Levy, Well-differentiated G1/G2 pancreatic NETs can evolve towards G3 tumors, Proceedings of the 14th Annual ENETS Conference, 8–10 March 2017, Barcelona, Spain, Neuroendocrinology. 105 (suppl 1), 1–338, Abstract G13 (2017)
F. Panzuto, N. Cicchese, S. Partelli, M. Rinzivillo, G. Capurso, E. Merola, M. Manzoni, E. Pucci, E. Iannicelli, E. Pilozzi, M. Rossi, C. Doglioni, M. Falconi, G. Delle Fave, Impact of Ki67 re-assessment at time of disease progression in patients with pancreatic neuroendocrine neoplasms. PLoS ONE 12, e0179445 (2017)
F. Grillo, M. Albertelli, M.P. Brisigotti, T. Borra, M. Boschetti, R. Fiocca, D. Ferone, L. Mastracci, Grade increases in gastroenteropancreatic neuroendocrine tumor metastases compared to the primary tumor. Neuroendocrinology 103, 452–59 (2016)
H. Shi, Q. Zhang, C. Han, D. Zhen, R. Lin, Variability of the Ki-67 proliferation index in gastroenteropancreatic neuroendocrine neoplasms—a single-center retrospective study. BMC Endocr. Disord. 18, 51 (2018)
J. Botling, A. Lamarca, D. Bajic, O. Norlén, V. Lönngren, J. Kjaer, B. Eriksson, S. Welin, P. Hellman, G. Rindi, B. Skogseid, J. Crona, High-grade progression confers poor survival in pancreatic neuroendocrine tumors. Neuroendocrinology 110, 891–898 (2020)
R.V. Lloyd, R.Y. Osamura, G. Kloppel, J. Rosai. WHO Classification of Tumors of Endocrine organs. 4th ed. International. Agency for Research on Cancer, Lyon, (2017) 211–214
P. Yu, C. Sa, F. Xiaobing, Z. Andong, p53: the barrier or guardian for cell dedifferentiation? BioScience 64, 883–892 (2014)
T. Cuny, W. de Herder, A. Barlier, L.J. Hofland, Role of the tumor microenvironment in digestive neuroendocrine tumors. Endocr. Relat. Cancer 25, R519–R544 (2018)
V. Fendrich, K. Maschuw, J. Waldmann, M. Buchholz, J. Rehm, T.M. Gress, D.K. Bartsch, A. König, Epithelial-mesenchymal transition is a critical step in tumorgenesis of pancreatic neuroendocrine tumors. Cancers 4, 281–294 (2012)
D. Friedmann-Morvinski, I.M. Verma, Dedifferentiation and reprogramming: origins of cancer stem cells. Embo. Rep. 15, 244–253 (2014)
WHO Classification of Tumors Editorial Board. Digestive System Tumors WHO Classification of Tumors, Volume 1. 5th ed. International Agency for Research on Cancer, Lyon, (2019) 17–19
B. Konukiewitz, A.M. Schlitter, M. Jesinghaus, D. Pfister, K. Steiger, A. Segler, A. Agaimy, B. Sipos, G. Zamboni, W. Weichert, I. Esposito, N. Pfarr, G. Klöppel, Somatostatin receptor expression related to TP53 and RB1 alterations in pancreatic and extrapancreatic neuroendocrine neoplasms with a Ki67-index above 20. Mod. Pathol. 30, 587–598 (2017)
G. Kyriakopoulos, V. Mavroeidi, E. Chatzellis, G.A. Kaltsas, K.I. Alexandraki, Histopathological, immunohistochemical, genetic and molecular markers of neuroendocrine neoplasms. Ann. Transl. Med. 6, 252 (2018)
S. Basu, V. Ostwal, Observation on enhanced avidity on somatostatin receptor targeted 68Ga-DOTATATE PET-CT following therapy with everolimus and capecitabine-temozolamide: is redifferentiation akin phenomenon a reality in neuroendocrine tumors? Nucl. Med. Commun. 37, 669–671 (2016)
K. Daskalakis, M. Tsoli, R. Srirajaskanthan, E. Chatzellis, K. Alexandraki, A. Angelousi, M. Pizanias, H. Randeva, G. Kaltsas, M.O. Weickert, Lung metastases in patients with well-differentiated gastroenteropancreatic neuroendocrine neoplasms: an appraisal of the validity of thoracic imaging surveillance. Neuroendocrinology 108, 308–316 (2019)
K.I. Alexandraki, M. Pizanias, I. Uri, D. Thomas, T. Page, D. Kolomodi, C.S. Low, O. Adesanya, M. Tsoli, D.J. Gross, H. Randeva, R. Srirajaskanthan, S. Grozinsky-Glasberg, G. Kaltsas, M.O. Weickert, The prognosis and management of neuroendocrine neoplasms-related metastatic bone disease: lessons from clinical practice. Endocrine 64, 690–701 (2019)
K.I. Alexandraki, A. Philippou, G. Boutzios, I. Theohari, M. Koutsilieris, I.K. Delladetsima, G.A. Kaltsas, IGF-IEc expression is increased in secondary compared to primary foci in neuroendocrine neoplasms. Oncotarget 8, 79003–79011 (2017)
M. Kaklamanos, I. Karoumpalis, C. Salla, D. Thomas, G. Kanakis, K. Alexandraki, S. Sougioultzis, E. Diakatou, G. Kontogeorgos, G. Kaltsas, Diagnostic accuracy and clinical significance of the fine needle aspiration Ki-67 labelling index in pancreatic endocrine tumours. Endocr. Relat. Cancer 18, L1–3 (2011)
L. Laskiewicz, S. Jamshed, Y. Gong, S. Ainechi, J. LaFemina, X. Wang, The diagnostic value of FNA biopsy in grading pancreatic neuroendocrine tumors. Cancer Cytopathol. 126, 170–178 (2018)
Author information
Authors and Affiliations
Contributions
Conceptualization, K.I.A., G.A.K.; Design/methodology, K.I.A., S.G.-G., G.R., G.A.K.; Analysis, K.I.A. M.K., G.K., V.M., A.K., K.A., S.T.; Investigation, K.I.A. G.K., S.T.; Data curation, K.I.A. M.K., G.K., V.M., A.K., K.A., S.T., G.R., S.G.-G.; Writing—original draft preparation, K.I.A.; Writing—review and editing, K.I.A., S.G.-G., and G.A.K.; Visualization, G.K., S.T.; Validation, S.T., G.R., A.B.G.; Supervision, S.G.-G., A.B.G., G.A.K.; Project administration, K.I.A.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Alexandraki, K.I., Kaltsatou, M., Kyriakopoulos, G. et al. Distinctive features of pancreatic neuroendocrine neoplasms exhibiting an increment in proliferative activity during the course of the disease. Endocrine 72, 279–286 (2021). https://doi.org/10.1007/s12020-020-02540-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-020-02540-w