Abstract
Objective
The expression pattern of exosomal miRNAs derived from parathyroid tumor is still unknown. In the present work, we aimed to examine the differences on microRNA (miRNA) expression, present in serum exosomes, by comparing parathyroid carcinoma (PC) and parathyroid adenoma (PA).
Methods
MiRNA expression profile of serum exosomes, derived from 4 PC patients and 4 PA patients, were analyzed by next-generation sequencing technology. The differential expressions of target miRNAs were further verified in both serum exosomes and tissues of PC/PA patients by quantitative reverse transcription polymerase chain reaction (qRT-PCR). Lastly, receiver operating characteristic (ROC) curves were plotted to investigate the efficiency of target exosomal miRNAs in distinguishing PC patients from PA controls.
Results
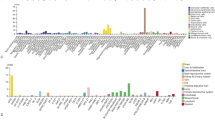
Multiple differentially expressed miRNAs of serum exosomes were screened out by sequencing. Based on this screening, hsa-miR-146b-5p (p = 0.0846), hsa-miR-27a-5p (p = 0.0412), hsa-miR-93-5p (p = 0.73), hsa-miR-381-3p (p = 0.1239) and hsa-miR-134-5p (p = 0.0694) were upregulated in the serum exosomes of PC patients. These results were validated by qPCR, where the trend on differential miRNA expression was consistent with the sequencing results. Specifically, the expression of exosomal hsa-miR-27a-5p was able to clearly distinguish PC patients from PA controls, and related analysis indicated that the area under the ROC curve was 0.8594 (p = 0.0157).
Conclusions
Here we present, for the first time, the miRNA expression profile of serum exosomes derived from PC patients. Based on this result, we presently suggest that the exosomal hsa-miR-27a-5p may serve as a putative tumor marker for preoperative identification of PC and PA subjects.




Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
F. Cetani, E. Pardi, C. Marcocci, Update on parathyroid carcinoma. J. Endocrinol. Invest. 39(6), 595–606 (2016)
Y. Hu et al. Circular RNA profile of parathyroid neoplasms: analysis of co-expression networks of circular RNAs and mRNAs. RNA Biol. 16(9), 1228–1236 (2019)
Y. Nakamura et al. Nonfunctional parathyroid carcinoma. Int. J. Clin. Oncol. 15(5), 500–503 (2010)
A. Harari et al. Parathyroid carcinoma: a 43-year outcome and survival analysis. J. Clin. Endocrinol. Metabol. 96(12), 3679–3686 (2011)
C.N. Patel et al. Clinical utility of ultrasound and 99mTc sestamibi SPECT/CT for preoperative localization of parathyroid adenoma in patients with primary hyperparathyroidism. Clin. Radiol. 65(4), 27a8–287a8 (2010)
W.T. Kassahun, S. Jonas, Focus on parathyroid carcinoma. Int. J. Surg. 9(1), 13–19 (2011)
S.E. Rodgers, N.D. Perrier, Parathyroid carcinoma. Curr. Opin. Oncol. 18(1), 16–22 (2006)
C. Sadler et al. Parathyroid carcinoma in more than 1,000 patients: a population-level analysis. Surgery 156(6), 1622–1630 (2014)
R. Kalluri, V.S. LeBleu, The biology, function, and biomedical applications of exosomes. Science 367(6478), eaau6977 (2020)
K.M. McAndrews, R. Kalluri, Mechanisms associated with biogenesis of exosomes in cancer. Mol. Cancer 18(1), 1–11 (2019)
K. Takahashi et al. Extracellular vesicle-mediated transfer of long non-coding RNA ROR modulates chemosensitivity in human hepatocellular cancer. FEBS Open Bio. 4(1), 458–467 (2014)
K. Li et al. Exosomes play roles in sequential processes of tumor metastasis. Int. J. Cancer 144(7), 1486–1495 (2019)
Q. Fan et al. The emerging role of exosome-derived non-coding RNAs in cancer biology. Cancer Lett. 1(414), 107–115 (2018)
R. Xu et al. Extracellular vesicles in cancer - implications for future improvements in cancer care. Nat. Rev. Clin. Oncol. 15(10), 617–638 (2018)
W. Wang, Y. Chen, Circulating miRNAs in cancer: from detection to therapy. J. Hematol. Oncol. 7(1), 86 (2014)
B. Givi, J.P. Shah, Parathyroid carcinoma. Clin. Oncol. 22(6), 498–507 (2010)
G. van Niel, G. D’Angelo, G. Raposo, Shedding light on the cell biology of extracellular vesicles. Nat. Rev. Mol. Cell Biol. 19(4), 213–228 (2018)
Y. Hu et al. Verification of candidate microRNA markers for parathyroid carcinoma. Endocrine. 60(2), 246–254 (2018)
J. Skog et al. Glioblastoma microvesicles transport RNA and proteins that promote tumour growth and provide diagnostic biomarkers. Nat. Cell Biol. 10(12), 1470–1476 (2008)
V. Armand-Labit, A. Pradines, Circulating cell-free microRNAs as clinical cancer biomarkers. Biomol. Concepts 8(2), 61–81 (2017)
R. Rahbari et al. Identification of differentially expressed microRNA in parathyroid tumors. Ann. Surg. Oncol. 18(4), 1158–1165 (2011)
Q. Fang, L. Sang, S. Du, Long noncoding RNA LINC00261 regulates endometrial carcinoma progression by modulating miRNA/FOXO1 expression. Cell Biochem. Funct. 36(6), 323–330 (2018)
M. Ashrafizadeh et al. Flaming the fight against cancer cells: the role of microRNA-93. Cancer Cell Int. 20, 277 (2020)
C. Wu et al. LINC00470 promotes tumour proliferation and invasion, and attenuates chemosensitivity through the LINC00470/miR-134/Myc/ABCC1 axis in glioma. J. Cell Mol. Med. 00, 1–13 (2020)
Y.Y. Luo et al. LncRNA-NEAT1 promotes proliferation of T-ALL cells via miR-146b-5p/NOTCH1 signaling pathway. Pathol. Res. Pract. 216(11), 153212 (2020)
H. Ma, G. Lv, D. Zhang, miR-381 mediates the development of head and neck squamous cell carcinoma via targeting STC2. Onco Targets Therapy 13, 4485–4493 (2020)
C.C. Juhlin et al. Loss of expression for the Wnt pathway components adenomatous polyposis coli and glycogen synthase kinase 3-beta in parathyroid carcinomas. Int. J. Oncol. 34(2), 481–492 (2009)
S. Shah et al. Ras and Rap1: a tale of two GTPases. Seminars Cancer Biol. 54, 29–39 (2019)
V. Hoskin et al. Ezrin regulates focal adhesion and invadopodia dynamics by altering calpain activity to promote breast cancer cell invasion. Mol. Biol. Cell 26(19), 3464–3479 (2015)
K. Leelahavanichkul et al. A role for p38 MAPK in head and neck cancer cell growth and tumor-induced angiogenesis and lymphangiogenesis. Mol. Oncol. 8(1), 105–118 (2014)
J. Zhang et al. MicroRNA-27a (miR-27a) in solid tumors: a review based on mechanisms and clinical observations. Front. Oncol. 9, 893 (2019)
J. Svedlund et al. Aberrant WNT/β-catenin signaling in parathyroid carcinoma. Mol. Cancer 9(1), 294 (2010)
Acknowledgements
We appreciated Thyroid and Neck Surgery of Beijing Chao-Yang Hospital for providing start-up funding for this study.
Author information
Authors and Affiliations
Contributions
J.W. analyzed and interpreted the experimental data, and was a major contributor in writing the manuscript. All authors read and approved the final manuscript
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
This study was approved by the Ethics Review Board of Beijing Chao-Yang Hospital, Capital Medical University.
Informed consent
All patients have signed a written informed consent to participate in this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Wang, J., Wang, Q., Zhao, T. et al. Expression profile of serum-related exosomal miRNAs from parathyroid tumor. Endocrine 72, 239–248 (2021). https://doi.org/10.1007/s12020-020-02535-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-020-02535-7