Abstract
Purpose
To describe the case of a 9-year-old male patient with neurofibromatosis type 1 (NF1), partial empty sella (PES), transient central adrenal insufficiency (CAI) and growth hormone (GH) deficiency (GHD) treated with recombinant GH (rGH).
Methods
The diagnosis of GHD was established upon peak GH response <10 ng/mL following glucagon and clonidine stimulation tests. CAI was diagnosed when peak cortisol response was <18 μg/dL following 1 μg Synacthen test (ST) with normal ACTH levels.
Results
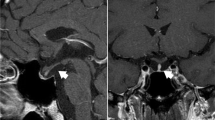
The diagnosis of NF1 was made at the age of 1.5 year. The patient first attended our Department at the age of 4.5 years. He presented with short stature (height: 95 cm < 3rd percentile), macrocephaly, frontal bossing, café-au-lait spots and bilateral proptosis. His growth rate (GR) initially was 5.3 cm/year. Brain/pituitary MRI showed T2-hyperintensities typical for NF1 and PES with reduced pituitary gland height (3 mm). The pituitary function tests revealed GHD. During follow-up his imaging findings remained unchanged, while his GR decelerated. He was started on rGH at the age of 8.5 years. Within the following year he grew 8.7 cm in height and could preserve a normal GR thereafter. At the age of 10.3 years, he was diagnosed with CAI (maximum cortisol response post-1 μg ST: 13.1 μg/dL). Ηe received hydrocortisone for 1 year. A repeat 1 μg ST off hydrocortisone showed normal cortisol response. During follow-up, brain MRI findings remained stable, while his pituitary demonstrated normal size and signal intensity.
Conclusion
Empty sella and hypopituitarism may occur in the context of NF1. Short stature may be associated with GHD in the absence of intrasellar masses in affected individuals. Lifelong endocrine follow-up is recommended for all NF1 patients.


Similar content being viewed by others
References
A.C. Hirbe, D.H. Gutmann, Neurofibromatosis type 1: a multidisciplinary approach to care. Lancet Neurol. 13(8), 834–843 (2014)
V.C. Williams, J. Lucas, M.A. Babcock, D.H. Gutmann, B. Korf, B.L. Maria, Neurofibromatosis type 1 revisited. Pediatrics 123(1), 124–133 (2009)
B. Hegedus, T.H. Yeh, D.Y. Lee, R.J. Emnett, J. Li, D.H. Gutmann, Neurofibromin regulates somatic growth through the hypothalamic-pituitary axis. Hum. Mol. Genet. 17(19), 2956–2966 (2008)
Y.S. Yap, J.R. McPherson, C.K. Ong, S.G. Rozen, B.T. Teh, A.S. Lee, D.F. Callen, The NF1 gene revisited—from bench to bedside. Oncotarget 5(15), 5873–5892 (2014)
S. El-Watidy, A. Jamjoom, Z.A. Jamjoom, W. Selen, Cranio-orbital anomalies in Von Recklinghausen neurofibromatosis simulating an intraorbital space-occupying lesion. Ann. Saudi. Med. 15(4), 399–401 (1995)
E.A. Soucy, D. van Oppen, N.L. Nejedly, F. Gao, D.H. Gutmann, A.S. Hollander, Height assessments in children with neurofibromatosis type 1. J. Child. Neurol. 28(3), 303–307 (2013)
M. Clementi, S. Milani, I. Mammi, S. Boni, C. Monciotti, R. Tenconi, Neurofibromatosis type 1 growth charts. Am. J. Med. Genet. 87(4), 317–323 (1999)
J. Szudek, P. Birch, J.M. Friedman, Growth in North American white children with neurofibromatosis 1 (NF1). J. Med. Genet. 37(12), 933–938 (2000)
R. Vassilopoulou-Sellin, M.J. Klein, J.K. Slopis, Growth hormone deficiency in children with neurofibromatosis type 1 without suprasellar lesions. Pediatr. Neurol. 22(5), 355–358 (2000)
S.J. Howell, P. Wilton, A. Lindberg, S.M. Shalet, Growth hormone replacement and the risk of malignancy in children with neurofibromatosis. J. Pediatr. 133(2), 201–205 (1998)
W. Waheed, M.H. Nathan, G.B. Allen, N.M. Borden, M.A. Babi, R. Tandan, Neurofibromatosis 1-associated panhypopituitarism presenting as hypoglycaemic seizures and stroke-like symptoms. BMJ Case Rep. (2015). https://doi.org/10.1136/bcr-2015-210816
S. Raman, A. Grimberg, S.G. Waguespack, B.S. Miller, C.A. Sklar, L.R. Meacham, B.C. Patterson, Risk of neoplasia in pediatric patients receiving growth hormone therapy–a report from the Pediatric Endocrine Society Drug and Therapeutics Committee. J. Clin. Endocrinol. Metab. 100(6), 2192–2203 (2015)
C. Bizzarri, G. Bottaro, Endocrine implications of neurofibromatosis 1 in childhood. Horm. Res. Paediatr. 83(4), 232–241 (2015)
I. Sani, A. Albanese, Endocrine long-term follow-up of children with neurofibromatosis type 1 and optic pathway glioma. Horm. Res. Paediatr. 87(3), 179–188 (2017)
D. Carmi, M. Shohat, A. Metzker, Z. Dickerman, Growth, puberty, and endocrine functions in patients with sporadic or familial neurofibromatosis type 1: a longitudinal study. Pediatrics. 103(6 Pt 1), 1257–1262 (1999)
R. Virdis, M.E. Street, M.A. Bandello, C. Tripodi, A. Donadio, A.R. Villani, L. Cagozzi, L. Garavelli, S. Bernasconi, Growth and pubertal disorders in neurofibromatosis type 1. J. Pediatr. Endocrinol. Metab. 16(Suppl 2), 289–292 (2003)
M.L. Iezzi, G. Varriale, L. Zagaroli, M. Greco, S. Lasorella, A. Verrotti di Pianella, Growth hormone deficit as a first clinical manifestation of early diffuse cerebral glioma in patient with neurofibromatosis. Ann. Clin. Lab. Res. 5, 1 (2017)
M.H. Cnossen, E.N. Stam, L.C. Cooiman, H.J. Simonsz, H. Stroink, A.P. Oranje, D.J. Halley, A. de Goede-Bolder, M.F. Niermeijer, S.M.de Muinck Keizer-Schrama, Endocrinologic disorders and optic pathway gliomas in children with neurofibromatosis type 1. Pediatrics 100(4), 667–670 (1997).
A.R. Shepard, W. Zhang, N.L. Eberhardt, Two CGTCA motifs and a GHF1/Pit1 binding site mediate cAMP-dependent protein kinase a regulation of human growth hormone gene expression in rat anterior pituitary GC cells. J. Biol. Chem. 269(3), 1804–1814 (1994)
B. Dasgupta, L.L. Dugan, D.H. Gutmann, The neurofibromatosis 1 gene product neurofibromin regulates pituitary adenylate cyclase-activating polypeptide-mediated signaling in astrocytes. J. Neurosci. 23(26), 8949–8954 (2003)
E.H. Burrows, Bone changes in orbital neurofibromatosis. Br. J. Radiol. 36, 549–561 (1963)
S.K. Gupta, H.V. Nema, P.L. Bhatia, K. Sasibabu, R. Kesharwani, The radiological features of craniofacial neurofibromatosis. Clin. Radiol. 30(5), 553–557 (1979)
R.L. Agrawal, S. Bhargava, A.H. Samma, A.K. Kothari, H.K. Bedi, R.L. Shrimali, Empty sella syndrome with neurofibromatosis. Indian J. Ophthalmol. 24(4), 38–40 (1977)
D. Miljic, S. Pekic, V. Popovic, Empty sella. In Endotext [Internet], eds by K.R. Feingold, B. Anawalt, A. Boyce, G. Chrousos, K. Dungan, A. Grossman, J.M. Hershman, G. Kaltsas, C. Koch, P. Kopp, M. Korbonits, R. McLachlan, J.E. Morley, M. New, L. Perreault, J. Purnell, R. Rebar, F. Singer, D.L. Trence, A. Vinik, D.P. Wilson (MDText.com, Inc, South Dartmouth, MA, 2018)
L. De Marinis, S. Bonadonna, A. Bianchi, G. Maira, A. Giustina, Primary empty sella. J. Clin. Endocrinol. Metab. 90(9), 5471–5477 (2005)
S. Chiloiro, A. Giampietro, A. Bianchi, T. Tartaglione, A. Capobianco, C. Anile, L. De Marinis, Diagnosis of endocrine disease: primary empty sella: a comprehensive review. Eur. J. Endocrinol. 177(6), R275–R285 (2017)
A.M. Lenz, A.W. Root, Empty sella syndrome. Pediatr. Endocrinol. Rev. 9(4), 710–715 (2012)
D. Rath, R.K. Sahoo, J. Choudhury, D.K. Dash, A. Mohapatra, Empty sella syndrome in a male child with failure to thrive. J. Pediatr. Neurosci. 10(1), 45–47 (2015)
R.E. Ferner, D.H. Gutmann, Neurofibromatosis type 1 (NF1): diagnosis and management. Handb. Clin. Neurol. 115, 939–955 (2013)
P. Julayanont, A. Karukote, D. Ruthirago, D. Panikkath, R. Panikkath, Idiopathic intracranial hypertension: ongoing clinical challenges and future prospects. J. Pain. Res. 9, 87–99 (2016)
A.L. Gilbert, G. Heidary, Update on the evaluation of pediatric idiopathic intracranial hypertension. Curr. Opin. Ophthalmol. 27(6), 493–497 (2016)
B. Shofty, L. Ben-Sira, S. Constantini, S. Freedman, A. Kesler, Optic nerve sheath diameter on MR imaging: establishment of norms and comparison of pediatric patients with idiopathic intracranial hypertension with healthy controls. AJNR Am. J. Neuroradiol. 33(2), 366–369 (2012)
T. Mushtaq, F. Shakur, J.K. Wales, N.P. Wright, Reliability of the low dose synacthen test in children undergoing pituitary function testing. J. Pediatr. Endocrinol. Metab. 21(12), 1129–1132 (2008)
M.C. Raux Demay, J.P. Magny, N. Idrès, A. Grimfeld, Y. Le Bouc, Use of the low-dose corticotropin stimulation test for the monitoring of children with asthma treated with inhaled corticosteroids. Horm. Res. 66(2), 51–60 (2006)
L.M. Mongioì, R.A. Condorelli, F. Barbagallo, R. Cannarella, S. La Vignera, A.E. Calogero, Accuracy of the low-dose ACTH stimulation test for adrenal insufficiency diagnosis: a re-assessment of the cut-off value. J. Clin. Med. 8(6), pii: E806 (2019)
M. Wade, S. Baid, K. Calis, H. Raff, N. Sinaii, L. Nieman, Technical details influence the diagnostic accuracy of the 1 microg ACTH stimulation test. Eur. J. Endocrinol. 162(1), 109–113 (2010)
A. Ciresi, S. Radellini, E. Vigneri, V. Guarnotta, J. Bianco, M.G. Mineo, C. Giordano, Correlation between adrenal function, growth hormone secretion, and insulin sensitivity in children with idiopathic growth hormone deficiency. J. Endocrinol. Invest. 41(3), 333–342 (2018)
C. Giavoli, R. Libé, S. Corbetta, E. Ferrante, A. Lania, M. Arosio, A. Spada, P. Beck-Peccoz, Effect of recombinant human growth hormone (GH) replacement on the hypothalamic-pituitary-adrenal axis in adult GH-deficient patients. J. Clin. Endocrinol. Metab. 89(11), 5397–5401 (2004)
C. Giavoli, Unmasking other pituitary deficits during growth hormone replacement therapy. Ann. Endocrinol. 68(4), 237–240 (2007)
M. Tschöp, H. Lahner, H. Feldmeier, H. Grasberger, K.M. Morrison, O.E. Janssen, A.F. Attanasio, C.J. Strasburger, Effects of growth hormone replacement therapy on levels of cortisol and cortisol-binding globulin in hypopituitary adults. Eur. J. Endocrinol. 143(6), 769–773 (2000)
L. De Paepe, R. Abs, J. Verlooy, A. Clemens, R.M. Smets, P. Louis, J.J. Martin, Benign intracranial hypertension as a cause of transient partial pituitary deficiency. J. Neurol. Sci. 114(2), 152–155 (1993)
M.T. Zagardo, W.S. Cail, S.E. Kelman, M.I. Rothman, Reversible empty sella in idiopathic intracranial hypertension: an indicator of successful therapy? AJNR Am. J. Neuroradiol. 17(10), 1953–1956 (1996)
V. Triggiani, V.A. Giagulli, M. Moschetta, E. Guastamacchia, An unusual case of reversible empty sella. Endocr. Metab. Immune Disord. Drug Targets. 16(2), 154–156 (2016)
A. Giustina, G. Aimaretti, M. Bondanelli, F. Buzi, S. Cannavò, S. Cirillo, A. Colao, L. De Marinis, D. Ferone, M. Gasperi, S. Grottoli, T. Porcelli, E. Ghigo, E. degli Uberti, Primary empty sella: why and when to investigate hypothalamic-pituitary function. J. Endocrinol. Invest. 33(5), 343–346 (2010)
R.E. Ferner, S.M. Huson, N. Thomas, C. Moss, H. Willshaw, D.G. Evans, M. Upadhyaya, R. Towers, M. Gleeson, C. Steiger, A. Kirby, Guidelines for the diagnosis and management of individuals with neurofibromatosis 1. J. Med. Genet. 44(2), 81–88 (2007)
K.S. Cunha, E.P. Barboza, E.C. Fonseca, Identification of growth hormone receptor in plexiform neurofibromas of patients with neurofibromatosis type 1. Clinics 63(1), 39–42 (2008)
U. Dagalakis, M. Lodish, E. Dombi, N. Sinaii, J. Sabo, A. Baldwin, S.M. Steinberg, C.A. Stratakis, B.C. Widemann, Puberty and plexiform neurofibroma tumor growth in patients with neurofibromatosis type I. J. Pediatr. 164(3), 620–624 (2014)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The study was approved by the Ethics Committee of “Aghia Sophia” Children’s Hospital and all procedures performed were in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from the patient’s parents before including the results of his investigations in this case report.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kyritsi, E.M., Hasiotou, M. & Kanaka-Gantenbein, C. Partial empty sella syndrome, GH deficiency and transient central adrenal insufficiency in a patient with NF1. Endocrine 69, 377–385 (2020). https://doi.org/10.1007/s12020-020-02351-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-020-02351-z