Abstract
Purpose
Serum complement C3 has been shown to contribute to the incidence of type 2 diabetes (T2D), but how serum complement C3 affects islet β-cell function throughout the course of T2D is unclear. This study explored whether serum complement C3 is independently associated with changes in islet β-cell function over time in patients with T2D.
Methods
Serum complement C3 was measured, and endogenous β-cell function was evaluated by area under the C-peptide curve (AUCcp) during an oral glucose tolerance test (OGTT) in 411 patients with T2D at baseline from 2011 to 2015. Next, 347 of those patients with available data were pooled for a final follow-up analysis from 2014 to 2018. Changes in islet β-cell function at follow-up were evaluated by AUCcp percentage changes (ΔAUCcp%). In addition, other possible clinical risks for diabetes were also examined.
Results
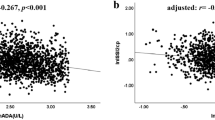
The 347 patients included in the analysis had a diabetes duration of 4.84 ± 3.63 years at baseline. Baseline serum complement C3 (baseline C3) levels were positively correlated with baseline natural logarithm of AUCcp (lnAUCcp) (n = 347, r = 0.288, p < 0.001), and baseline C3 was independently associated with baseline lnAUCcp (β = 0.17, t = 3.52, p < 0.001) after adjustment for baseline glycemic status and other clinical confounders by multivariate liner regression analysis. Compared with the baseline values, complement C3 changes (ΔC3) and ΔAUCcp% was –0.15 ± 0.28 mg/ml and –17.2 ± 18.4%, respectively, at a follow-up visit 4.57 ± 0.78 years later. Moreover, ΔC3 was positively correlated with ΔAUCcp% (n = 347, r = 0.302, p < 0.001). Furthermore, each 0.1 mg/ml increase in ΔC3 was associated with a higher ΔAUCcp% (1.41% [95% CI, 0.82–2.00%]) after adjusting for changes in glycemic status and other clinical confounders at follow-up.
Conclusions
In addition to serum complement C3 being independently associated with islet β-cell function at baseline, its changes were also independently associated with changes in islet β-cell function over time in patients with T2D.



Similar content being viewed by others
Abbreviations
- T2D:
-
type 2 diabetes
- AUCcp :
-
area under the C-peptide curve
- lnAUCcp :
-
natural logarithm of AUCcp
- ΔAUCcp%:
-
percentage AUCcp change
- AUCglu :
-
area under the glucose curve
- baseline C3:
-
complement C3 at baseline
- ΔC3:
-
changes in complement C3
- OGTT:
-
oral glucose tolerance test
- BMI:
-
body mass index
- SBP/DBP:
-
systolic/diastolic blood pressure
- serum UA:
-
serum uric acid
- TG:
-
triglyceride
- TC:
-
total cholesterol
- LDLC:
-
low-density lipoprotein cholesterol
- HDLC:
-
high-density lipoprotein cholesterol
- eGFR:
-
estimated glomerular filtration rate
- IScp :
-
insulin sensitivity from C-peptide
- HbA1c:
-
glycosylated hemoglobin A1c
References
B.L. Wajchenberg, beta-cell failure in diabetes and preservation by clinical treatment. Endocr. Rev. 28(2), 187–218 (2007). https://doi.org/10.1210/10.1210/er.2006-0038
R.C. Tubner, C.A. Cull, R.R. Holman, U.K. prospective diabetes study. Diabetes Care 19(2), 182–183 (1996)
M.Y. Donath, S.E. Shoelson, Type 2 diabetes as an inflammatory disease. Nat. Rev. Immunol. 11(2), 98–107 (2011). https://doi.org/10.1038/nri2925
J. Rojas, V. Bermudez, J. Palmar, M.S. Martinez, L.C. Olivar, M. Nava, D. Tomey, M. Rojas, J. Salazar, C. Garicano, M. Velasco, Pancreatic beta cell death: novel potential mechanisms in diabetes therapy. J. Diabetes Res. 2018, 9601801 (2018). https://doi.org/10.1155/2018/9601801
N.S. Merle, S.E. Church, V. Fremeaux-Bacchi, L.T. Roumenina, Complement system part I—molecular mechanisms of activation and regulation. Front. Immunol. 6, 262 (2015). https://doi.org/10.3389/fimmu.2015.00262
D. Ricklin, G. Hajishengallis, K. Yang, J.D. Lambris, Complement: a key system for immune surveillance and homeostasis. Nat. Immunol. 11(9), 785–797 (2010). https://doi.org/10.1038/ni.1923
J.M. Moreno-Navarrete, J.M. Fernandez-Real, The complement system is dysfunctional in metabolic disease: Evidences in plasma and adipose tissue from obese and insulin resistant subjects. Semin. Cell Dev. Biol. 85, 164–172 (2019). https://doi.org/10.1016/j.semcdb.2017.10.025
G. Bajic, S.E. Degn, S. Thiel, G.R. Andersen, Complement activation, regulation, and molecular basis for complement-related diseases. Embo. J. 34(22), 2735–2757 (2015). https://doi.org/10.15252/embj.201591881
E. Hertle, M.M. van Greevenbroek, C.D. Stehouwer, Complement C3: an emerging risk factor in cardiometabolic disease. Diabetologia 55(4), 881–884 (2012). https://doi.org/10.1007/s00125-012-2462-z
X. Bao, Y. Xia, Q. Zhang, H.M. Wu, H.M. Du, L. Liu, C.J. Wang, H.B. Shi, X.Y. Guo, X. Liu, C.L. Li, Q. Su, G. Meng, B. Yu, S.M. Sun, X. Wang, M. Zhou, Q.Y. Jia, K. Song, K.J. Niu, Elevated serum complement C3 levels are related to the development of prediabetes in an adult population: the Tianjin Chronic Low-Grade Systematic Inflammation and Health Cohort Study. Diabet. Med. 33(4), 446–453 (2016). https://doi.org/10.1111/dme.12827
G. Engstrom, B. Hedblad, K.F. Eriksson, L. Janzon, F. Lindgarde, Complement C3 is a risk factor for the development of diabetes: a population-based cohort study. Diabetes 54(2), 570–575 (2005)
J. Phieler, R. Garcia-Martin, J.D. Lambris, T. Chavakis, The role of the complement system in metabolic organs and metabolic diseases. Semin. Immunol. 25(1), 47–53 (2013). https://doi.org/10.1016/j.smim.2013.04.003
J. Sjolander, G.T. Westermark, E. Renstrom, A.M. Blom, Islet amyloid polypeptide triggers limited complement activation and binds complement inhibitor C4b-binding protein, which enhances fibril formation. J. Biol. Chem. 287(14), 10824–10833 (2012). https://doi.org/10.1074/jbc.M111.244285
S. He, C. Atkinson, F. Qiao, K. Cianflone, X. Chen, S. Tomlinson, A complement-dependent balance between hepatic ischemia/reperfusion injury and liver regeneration in mice. J. Clin. Investig. 119(8), 2304–2316 (2009). https://doi.org/10.1172/jci38289
American Diabetes Association, Diagnosis and classification of diabetes mellitus. Diabetes Care 34(Suppl 1), S62–S69 (2011). https://doi.org/10.2337/dc11-S062
T. Radaelli, K.A. Farrell, L. Huston-Presley, S.B. Amini, J.P. Kirwan, H.D. McIntyre, P.M. Catalano, Estimates of insulin sensitivity using glucose and C-Peptide from the hyperglycemia and adverse pregnancy outcome glucose tolerance test. Diabetes Care 33(3), 490–494 (2010). https://doi.org/10.2337/dc09-1463
L. Zhao, J. Ma, S. Wang, Y. Xie, Relationship between beta-cell function, metabolic control, and microvascular complications in type 2 diabetes mellitus. Diabetes Technol. Ther. 17(1), 29–34 (2015). https://doi.org/10.1089/dia.2014.0214
L.A. Inker, C.H. Schmid, H. Tighiouart, J.H. Eckfeldt, H.I. Feldman, T. Greene, J.W. Kusek, J. Manzi, F. Van Lente, Y.L. Zhang, J. Coresh, A.S. Levey, Estimating glomerular filtration rate from serum creatinine and cystatin C. N. Engl. J. Med. 367(1), 20–29 (2012). https://doi.org/10.1056/NEJMoa1114248
J.J. Meier, B.A. Menge, T.G. Breuer, C.A. Muller, A. Tannapfel, W. Uhl, W.E. Schmidt, H. Schrader, Functional assessment of pancreatic beta-cell area in humans. Diabetes 58(7), 1595–1603 (2009). https://doi.org/10.2337/db08-1611
J.M. Fernandez-Real, G. Penarroja, A. Castro, F. Garcia-Bragado, I. Hernandez-Aguado, W. Ricart, Blood letting in high-ferritin type 2 diabetes: effects on insulin sensitivity and beta-cell function. Diabetes 51(4), 1000–1004 (2002)
A. Mallipedhi, T. Min, S.L. Prior, C. MacIver, S.D. Luzio, G. Dunseath, R.M. Bracken, S. Islam, J.D. Barry, S. Caplin, J.W. Stephens, Association between the preoperative fasting and postprandial C-peptide AUC with resolution of type 2 diabetes 6 months following bariatric surgery. Metabolism 64(11), 1556–1563 (2015). https://doi.org/10.1016/j.metabol.2015.08.009
H. Tfayli, F. Bacha, N. Gungor, S. Arslanian, Islet cell antibody-positive versus -negative phenotypic type 2 diabetes in youth: does the oral glucose tolerance test distinguish between the two? Diabetes Care 33(3), 632–638 (2010). https://doi.org/10.2337/dc09-0305
P. Rowe, C. Wasserfall, B. Croker, M. Campbell-Thompson, A. Pugliese, M. Atkinson, D. Schatz, Increased complement activation in human type 1 diabetes pancreata. Diabetes Care 36(11), 3815–3817 (2013). https://doi.org/10.2337/dc13-0203
A. Klegeris, P.L. McGeer, Complement activation by islet amyloid polypeptide (IAPP) and alpha-synuclein 112. Biochem. Biophys. Res. Commun. 357(4), 1096–1099 (2007). https://doi.org/10.1016/j.bbrc.2007.04.055
Y. Mamane, C. Chung Chan, G. Lavallee, N. Morin, L.J. Xu, J. Huang, R. Gordon, W. Thomas, J. Lamb, E.E. Schadt, B.P. Kennedy, J.A. Mancini, The C3a anaphylatoxin receptor is a key mediator of insulin resistance and functions by modulating adipose tissue macrophage infiltration and activation. Diabetes 58(9), 2006–2017 (2009). https://doi.org/10.2337/db09-0323
J.C. McNelis, J.M. Olefsky, Macrophages, immunity, and metabolic disease. Immunity 41(1), 36–48 (2014). https://doi.org/10.1016/j.immuni.2014.05.010
W. Quan, E.K. Jo, M.S. Lee, Role of pancreatic beta-cell death and inflammation in diabetes. Diabetes Obes. Metab. 15(Suppl 3), 141–151 (2013). https://doi.org/10.1111/dom.12153
S.S. Rensen, Y. Slaats, A. Driessen, C.J. Peutz-Kootstra, J. Nijhuis, R. Steffensen, J.W. Greve, W.A. Buurman, Activation of the complement system in human nonalcoholic fatty liver disease. Hepatology 50(6), 1809–1817 (2009). https://doi.org/10.1002/hep.23228
H. Esterbauer, F. Krempler, H. Oberkofler, W. Patsch, The complement system: a pathway linking host defence and adipocyte biology. Eur. J. Clin. Investig. 29(8), 653–656 (1999)
Z. Liu, Q. Tang, J. Wen, Y. Tang, D. Huang, Y. Huang, J. Xie, Y. Luo, M. Liang, C. Wu, Z. Lu, A. Tan, Y. Gao, Q. Wang, Y. Jiang, Z. Yao, X. Lin, H. Zhang, Z. Mo, X. Yang, Elevated serum complement factors 3 and 4 are strong inflammatory markers of the metabolic syndrome development: a longitudinal cohort study. Sci. Rep. 6, 18713 (2016). https://doi.org/10.1038/srep18713
N. Wlazlo, M.M. van Greevenbroek, I. Ferreira, E.J. Feskens, C.J. van der Kallen, C.G. Schalkwijk, B. Bravenboer, C.D. Stehouwer, Complement factor 3 is associated with insulin resistance and with incident type 2 diabetes over a 7-year follow-up period: the CODAM Study. Diabetes Care 37(7), 1900–1909 (2014). https://doi.org/10.2337/dc13-2804
K.L. Rasmussen, B.G. Nordestgaard, S.F. Nielsen, Complement C3 and risk of diabetic microvascular disease: a cohort study of 95,202 individuals from the general population. Clin. Chem. 64(7), 1113–1124 (2018). https://doi.org/10.1373/clinchem.2018.287581
G. Engstrom, B. Hedblad, L. Janzon, F. Lindgarde, Complement C3 and C4 in plasma and incidence of myocardial infarction and stroke: a population-based cohort study. Eur. J. Cardiovasc. Prev. Rehabil. 14(3), 392–397 (2007). https://doi.org/10.1097/01.hjr.0000244582.30421.b2
I.U. Schraufstatter, R.G. Discipio, M. Zhao, S.K. Khaldoyanidi, C3a and C5a are chemotactic factors for human mesenchymal stem cells, which cause prolonged ERK1/2 phosphorylation. J. Immunol. 182(6), 3827–3836 (2009). https://doi.org/10.4049/jimmunol.0803055
N. Matsumoto, A. Satyam, M. Geha, P.H. Lapchak, J.J. Dalle Lucca, M.G. Tsokos, G.C. Tsokos, C3a enhances the formation of intestinal organoids through C3aR1. Front. Immunol. 8, 1046 (2017). https://doi.org/10.3389/fimmu.2017.01046
T. Haynes, A. Luz-Madrigal, E.S. Reis, N.P. Echeverri Ruiz, E. Grajales-Esquivel, A. Tzekou, P.A. Tsonis, J.D. Lambris, K. Del Rio-Tsonis, Complement anaphylatoxin C3a is a potent inducer of embryonic chick retina regeneration. Nat. Commun. 4, 2312 (2013). https://doi.org/10.1038/ncomms3312
C.W. Strey, M. Markiewski, D. Mastellos, R. Tudoran, L.A. Spruce, L.E. Greenbaum, J.D. Lambris, The proinflammatory mediators C3a and C5a are essential for liver regeneration. J. Exp. Med. 198(6), 913–923 (2003). https://doi.org/10.1084/jem.20030374
M.M. Markiewski, D. Mastellos, R. Tudoran, R.A. DeAngelis, C.W. Strey, S. Franchini, R.A. Wetsel, A. Erdei, J.D. Lambris, C3a and C3b activation products of the third component of complement (C3) are critical for normal liver recovery after toxic injury. J. Immunol. 173(2), 747–754 (2004). https://doi.org/10.4049/jimmunol.173.2.747
T.V. Fiorentino, M.L. Hribal, F. Andreozzi, M. Perticone, A. Sciacqua, F. Perticone, G. Sesti, Plasma complement C3 levels are associated with insulin secretion independently of adiposity measures in non-diabetic individuals. Nutr. Metab. Cardiovasc. Dis. 25(5), 510–517 (2015). https://doi.org/10.1016/j.numecd.2015.02.007
R.S. Dos Santos, L. Marroqui, F.A. Grieco, L. Marselli, M. Suleiman, S.R. Henz, P. Marchetti, R. Wernersson, D.L. Eizirik, Protective role of complement C3 against cytokine-mediated beta-cell apoptosis. Endocrinology 158(8), 2503–2521 (2017). https://doi.org/10.1210/en.2017-00104
B.C. King, K. Kulak, U. Krus, R. Rosberg, E. Golec, K. Wozniak, M.F. Gomez, E. Zhang, D.J. O’Connell, E. Renstrom, A.M. Blom, Complement component C3 is highly expressed in human pancreatic islets and prevents beta cell death via ATG16L1 interaction and autophagy regulation. Cell Metab. 29(1), 202–210.e206 (2019). https://doi.org/10.1016/j.cmet.2018.09.009
B. Ahren, P.J. Havel, G. Pacini, K. Cianflone, Acylation stimulating protein stimulates insulin secretion. Int. J. Obes. Relat. Metab. Disord. 27(9), 1037–1043 (2003). https://doi.org/10.1038/sj.ijo.0802369
J.C. Lo, S. Ljubicic, B. Leibiger, M. Kern, I.B. Leibiger, T. Moede, M.E. Kelly, D. Chatterjee Bhowmick, I. Murano, P. Cohen, A.S. Banks, M.J. Khandekar, A. Dietrich, J.S. Flier, S. Cinti, M. Bluher, N.N. Danial, P.O. Berggren, B.M. Spiegelman, Adipsin is an adipokine that improves beta cell function in diabetes. Cell 158(1), 41–53 (2014). https://doi.org/10.1016/j.cell.2014.06.005
M. Kolev, G. Le Friec, C. Kemper, Complement–tapping into new sites and effector systems. Nat. Rev. Immunol. 14(12), 811–820 (2014). https://doi.org/10.1038/nri3761
M. Wysoczynski, M. Solanki, S. Borkowska, P. van Hoose, K.R. Brittian, S.D. Prabhu, M.Z. Ratajczak, G. Rokosh, Complement component 3 is necessary to preserve myocardium and myocardial function in chronic myocardial infarction. Stem Cells 32(9), 2502–2515 (2014). https://doi.org/10.1002/stem.1743
Y. Rahpeymai, M.A. Hietala, U. Wilhelmsson, A. Fotheringham, I. Davies, A.K. Nilsson, J. Zwirner, R.A. Wetsel, C. Gerard, M. Pekny, M. Pekna, Complement: a novel factor in basal and ischemia-induced neurogenesis. Embo. J. 25(6), 1364–1374 (2006). https://doi.org/10.1038/sj.emboj.7601004
Y. Ma, Y. Liu, Z. Zhang, G.Y. Yang, Significance of complement system in ischemic stroke: a comprehensive review. Aging Dis. 10(2), 429–462 (2019). https://doi.org/10.14336/ad.2019.0119
Funding
This study was funded by the Social Development Projects of Nantong (HS2012028, MS22015065) and the Medical Research Project of Jiangsu Health Commission (QNRC2016408).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The study design was authorized by the ethical committee of Affiliated Hospital 2 of Nantong University in accordance with the principles of the Declaration of Helsinki.
Informed consent
Written informed consent was obtained from all patients.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
About this article
Cite this article
Su, Jb., Wu, YY., Xu, F. et al. Serum complement C3 and islet β-cell function in patients with type 2 diabetes: A 4.6-year prospective follow-up study. Endocrine 67, 321–330 (2020). https://doi.org/10.1007/s12020-019-02144-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-019-02144-z