Abstract
Background
Long non-coding RNAs (lncRNAs) are an emerging class of regulators in cancer. A lncRNA, MCM3AP-AS1, has been demonstrated as a versatile mediator in many cancers, except papillary thyroid cancer. The aim of this study is to investigate the role and mechanism of MCM3AP-AS1 in papillary thyroid cancer.
Methods
Quantitative real-time PCR was used to assess the level of MCM3AP-AS1 and miR-211-5p in papillary thyroid cancer tissues and cells. Western blot was used to detect E-cadherin and secreted protein acidic and cysteine rich (SPARC) protein levels. CCK-8, scratch wound assay, and transwell assay were used to evaluate papillary thyroid cancer cell proliferation, migration, and invasion, respectively. BLAST alignment and luciferase assay were used to explore the interaction among MCM3AP-AS1, mi/r-211, and SPARC.
Results
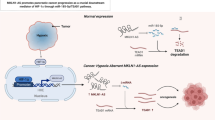
In papillary thyroid cancer, MCM3AP-AS1 was upregulated, while miR-211 was downregulated. MCM3AP-AS1 overexpression promoted papillary thyroid cancer proliferation, migration, and invasion. Further, MCM3AP-AS1 was shown to be negatively correlated with miR-211-5p. We next validated that miR-211-5p overexpression could reverse the promoting role of MCM3AP-AS1 in papillary thyroid cancer, whereby SPARC plays an important regulating role. In vivo, we confirmed the anti-tumor role of MCM3AP-AS1 silencing and the close relation among MCM3AP-AS1, miR-211-5p, and SPARC.
Conclusions
MCM3AP-AS1 promotes papillary thyroid cancer by regulating the MCM3AP-AS1/miR-211-5p/SPARC axis, which could potentially be a therapeutic target in papillary thyroid cancer.





Similar content being viewed by others
Data availability
The analyzed data sets generated during the study are available from the corresponding author on reasonable request.
Abbreviations
- PTC:
-
Papillary thyroid cancer
- lncRNA:
-
Long non-coding RNA
- miRNAs:
-
microRNAs
- GECs:
-
glioma-associated endothelial cells
- GBM:
-
Glioblastoma
- GFP:
-
green fluorescent protein
References
Z. Liu, T. Lv, C. Xie, Z. Di, BRAF V600E gene mutation is associated with bilateral malignancy of papillary thyroid cancer. Am. J. Med. Sci. 356(2), 130–134 (2018)
R.L. Siegel, K.D. Miller, A. Jemal, Cancer statistics, 2017. CA: A Cancer J. Clin. 67(1), 7–30 (2017)
W. Chen, R. Zheng, P.D. Baade, S. Zhang, H. Zeng, F. Bray, A. Jemal, X.Q. Yu, J. He, Cancer statistics in China, 2015. CA: A Cancer J. Clin. 66(2), 115–132 (2016)
F. Sui, M. Ji, P. Hou, Long non-coding RNAs in thyroid cancer: Biological functions and clinical significance. Mol. Cell. Endocrinol. 469, 11–22 (2018)
Y. Jin, W. Jin, Z. Zheng, E. Chen, Q. Wang, Y. Wang, O. Wang, X. Zhang, GABRB2 plays an important role in the lymph node metastasis of papillary thyroid cancer. Biochem. Biophys. Res. Commun. 492(3), 323–330 (2017)
P. Zhu, Y. Wang, J. Wu, G. Huang, B. Liu, B. Ye, Y. Du, G. Gao, Y. Tian, L. He et al. LncBRM initiates YAP1 signalling activation to drive self-renewal of liver cancer stem cells. Nat. Commun. 7, 13608 (2016)
X. Wu, Y. Yan, H. Li, N. Ji, T. Yu, Y. Huang, W. Shi, L. Gao, L. Ma, Y. Hu, DNA copy number gain-mediated lncRNA LINC01061 upregulation predicts poor prognosis and promotes papillary thyroid cancer progression. Biochem. Biophys. Res. Commun. 503(3), 1247–1253 (2018)
Y. He, X.M. Meng, C. Huang, B.M. Wu, L. Zhang, X.W. Lv, J. Li, Long noncoding RNAs: Novel insights into hepatocelluar carcinoma. Cancer Lett. 344(1), 20–27 (2014)
J.S. Mattick, I.V. Makunin, Non-coding RNA. Hum. Mol. Genet. 15, R17–R29 (2006). Spec No 1
E. Xia, A. Bhandari, Y. Shen, X. Zhou, N. Sindan, J. Xiang, Y. Guan, F. Yang, O. Wang, LncRNA CCND2-AS1 promotes proliferation, migration, and invasion in papillary thyroid carcinoma. Biochem. Biophys. Res. Commun. 496(2), 628–632 (2018)
N. Liu, Q. Zhou, Y. -H. Qi, H. Wang, L. Yang, Q. -Y. Fan, Effects of long non-coding RNA H19 and microRNA let7a expression on thyroid cancer prognosis. Exp. Mol. Pathol. 103(1), 71–77 (2017)
D. Kim, W.K. Lee, S. Jeong, M. -Y. Seol, H. Kim, K. -S. Kim, E.J. Lee, J. Lee, Y.S. Jo, Upregulation of long noncoding RNA LOC100507661 promotes tumor aggressiveness in thyroid cancer. Mol. Cell. Endocrinol. 431, 36–45 (2016)
W. Dai, Y. Tian, B. Jiang, W. Chen, Down-regulation of long non-coding RNA AFAP1-AS1 inhibits tumor growth, promotes apoptosis and decreases metastasis in thyroid cancer. Biomed. Pharmacother. 99, 191–197 (2018)
Y. Liu, P. Yue, T. Zhou, F. Zhang, H. Wang, X. Chen, LncRNA MEG3 enhances 131I sensitivity in thyroid carcinoma via sponging miR-182. Biomed. Pharmacother. 105, 1232–1239 (2018)
Q. Yuan, Y. Liu, Y. Fan, Z. Liu, X. Wang, M. Jia, Z. Geng, J. Zhang, X. Lu, LncRNA HOTTIP promotes papillary thyroid carcinoma cell proliferation, invasion and migration by regulating miR-637. Int. J. Biochem. Cell Biol. 98, 1–9 (2018)
C. Yang, J. Zheng, Y. Xue, H. Yu, X. Liu, J. Ma, L. Liu, P. Wang, Z. Li, H. Cai et al. The effect of MCM3AP-AS1/miR-211/KLF5/AGGF1 axis regulating glioblastoma angiogenesis. Front. Mol. Neurosci. 10(437), 437 (2018)
X. Li, S. Wang, Z. Li, X. Long, Z. Guo, G. Zhang, J. Zu, Y. Chen, L. Wen, The lncRNA NEAT1 facilitates cell growth and invasion via the miR-211/HMGA2 axis in breast cancer. Int. J. Biol. Macromol. 105, 346–353 (2017)
M. Boufraqech, J. Klubo-Gwiezdzinska, E. Kebebew, MicroRNAs in the thyroid. Best Pract. Res. Clin. Endocrinol. Metab. 30(5), 603–619 (2016)
P. Makhdoumi, A. Roohbakhsh, G. Karimi, MicroRNAs regulate mitochondrial apoptotic pathway in myocardial ischemia-reperfusion-injury. Biomed. Pharmacother. 84, 1635–1644 (2016)
V. Rosa, R. Lucia, P. Pierlorenzo, M. Ivana De, F. Angelo, L. Vincenza, B. Eleonora, P. Fabio, A. Hansjuerg, C. Carlo Maria et al. MicroRNAs (miR)-221 and miR-222, both overexpressed in human thyroid papillary carcinomas, regulate p27Kip1 protein levels and cell cycle. Endocr.-Relat. Cancer Endocr. Relat. Cancer 14(3), 791–798 (2007)
H. He, K. Jazdzewski, W. Li, S. Liyanarachchi, R. Nagy, S. Volinia, G.A. Calin, Liu C-g, K. Franssila, S. Suster et al. The role of microRNA genes in papillary thyroid carcinoma. Proc. Natl Acad. Sci. USA 102(52), 19075 (2005)
L. Liang, X. Zheng, M. Hu, Y. Cui, Q. Zhong, S. Wang, F. Huang, MiRNA-221/222 in thyroid cancer: A meta-analysis. Clin. Chim. Acta 484, 284–292 (2018)
L. Luo, L. Xia, B. Zha, C. Zuo, D. Deng, M. Chen, L. Hu, Y. He, F. Dai, J. Wu et al. miR-335-5p targeting ICAM-1 inhibits invasion and metastasis of thyroid cancer cells. Biomed. Pharmacother. 106, 983–990 (2018)
S. Wang, J. Wu, J. Ren, A.C. Vlantis, Li M-y, S.Y.W. Liu, E.K.W. Ng, A.B.W. Chan, D.-C. Luo, Z. Liu et al. MicroRNA-125b Interacts with Foxp3 to Induce Autophagy in Thyroid Cancer. Mol. Ther. 26(9), 2295–2303 (2018)
X. Li, Y. Tian, Y. Hu, Z. Yang, L. Zhang, J. Luo. CircNUP214 sponges miR-145 to promote the expression of ZEB2 in thyroid cancer cells. Biochem. Biophys. Res. Commun. 168–172 (2018)
A.A. Svoronos, D.M. Engelman, F.J. Slack. OncomiR or tumor suppressor? The duplicity of microRNAs in cancer. Cancer Res. 3666–3670 (2016)
R.E. Bell, M. Khaled, D. Netanely, S. Schubert, T. Golan, A. Buxbaum, M.M. Janas, B. Postolsky, M.S. Goldberg, R. Shamir et al. Transcription factor/microRNA axis blocks melanoma invasion program by miR-211 targeting NUAK1. J. Invest. Dermatol. 134(2), 441–451 (2014)
D. Xu, S. Liu, L. Zhang, L. Song, MiR-211 inhibits invasion and epithelial-to-mesenchymal transition (EMT) of cervical cancer cells via targeting MUC4. Biochem. Biophys. Res. Commun. 485(2), 556–562 (2017)
T.-H. Chu, C.-C. Yang, C.-J. Liu, M.-T. Lui, S.-C. Lin, K.-W. Chang, miR-211 promotes the progression of head and neck carcinomas by targeting TGFβRII. Cancer Lett. 337(1), 115–124 (2013)
G. Jiang, L. Wen, W. Deng, Z. Jian, H. Zheng, Regulatory role of miR-211-5p in hepatocellular carcinoma metastasis by targeting ZEB2. Biomed. Pharmacother. 90, 806–812 (2017)
J. Feng, L. Tang, SPARC in tumor pathophysiology and as a potential therapeutic target. Curr. Pharm. Des. 20(39), 6182–6190 (2014)
P. Liao, W. Li, R.Z. Liu, J.K. Teer, B.B. Xu, W. Zhang, X. Li, H.L. Mcleod, Y.J. He, Genome-scale analysis identifies SERPINE1 and SPARC as diagnostic and prognostic biomarkers in gastric cancer. Oncotargets Ther. 11, 6969–6980 (2018)
J. Ma, S. Gao, X. Xie, E. Sun, M. Zhang, Q. Zhou, C. Lu, SPARC inhibits breast cancer bone metastasis and may be a clinical therapeutic target. Oncol. Lett. 14(5), 5876–5882 (2017)
L. Wang, Y.-f. Shen, Z.-m. Shi, X.-j. Shang, D.-I. Jin, F. Xi. Overexpression miR-211-5p hinders the proliferation, migration, and invasion of thyroid tumor cells by downregulating SOX11. J. Clin. Lab Anal. 32, e22293 (2017)
H. Yu, W. Yang, MiR-211 is epigenetically regulated by DNMT1 mediated methylation and inhibits EMT of melanoma cells by targeting RAB22A. Biochem. Biophys. Res. Commun. 476(4), 400–405 (2016)
X. Qu, D. Gao, Q. Ren, X. Jiang, J. Bai, L. Sheng, miR-211 inhibits proliferation, invasion and migration of cervical cancer via targeting SPARC. Oncol. Lett. 16(1), 853–860 (2018)
Y. Wang, L. Yang, T. Chen, X. Liu, Y. Guo, Q. Zhu, X. Tong, W. Yang, Q. Xu, D. Huang, A novel lncRNA MCM3AP-AS1 promotes the growth of hepatocellular carcinoma by targeting miR-194-5p/FOXA1 axis. Mol. Cancer 18(1), 28 (2019)
Author contributions
All authors have read and approved the final manuscript.
Funding
This work was supported by the Petrel Science Foundation of Harbin Medical University Cancer Hospital (Grant No. JJLX2014-01).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The present study was approved by the Ethics Committee of Harbin Medical University Cancer Hospital.
Informed consent
All patients and healthy volunteers provided written informed consent prior to their inclusion within the study.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liang, M., Jia, J., Chen, L. et al. LncRNA MCM3AP-AS1 promotes proliferation and invasion through regulating miR-211-5p/SPARC axis in papillary thyroid cancer. Endocrine 65, 318–326 (2019). https://doi.org/10.1007/s12020-019-01939-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-019-01939-4