Abstract
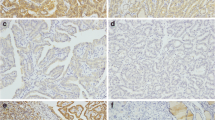
BRAF V600E mutation, usually performed by DNA techniques, is one of the most common diagnostic markers in papillary thyroid carcinoma. Few papers have demonstrated that plump cells (eosinophilic cytoplasms and papillary thyroid carcinoma nuclei) and peculiar sickle-shaped nuclei represent morphological features of BRAF V600E on papillary thyroid carcinomas. These features seem to be linked to glycolytic phenotype whereby monocarboxylate transporters 1–4 are hypothesized to have a dominant role as lactate transporters. We investigated the association between these morphological features and monocarboxylate transporters 1 and 4 in 48 cyto-histological samples diagnosed as “positive for malignancy-favoring papillary thyroid carcinoma”. These cases were processed with liquid-based cytology and underwent BRAF V600E mutational analysis (pyrosequencing) on liquid-based cytology and monocarboxylate transporters immunostaining on histology. The expression of monocarboxylate transporter 1, monocarboxylate transporter 4, glucose trasporter-1 and carbonic anhidrase were scored semi-quantitatively with expression from 0 to 3+ (strong positivity). The 33 mutated and 15 wild type cases showed 100 % cyto-histological concordance. The cytological evaluation revealed plump cells and sickle nuclear shape in 100 % mutated cases. Monocarboxylate transporter 1 yielded 76 % positivity in the mutated cases especially in both the plump cells and sickle-shaped nuclei, whereas the wild types showed 13.3 % positive monocarboxylate transporter 1 (p = 0.00013). Monocarboxylate transporter 4 resulted in 100 % positivity in mutated and 40 % in wild types (p < 0.005). Furthermore, 20 % of the wild types showed weak monocarboxylate transporter 1 nuclear expression associated to a less aggressive behavior. The analysis of glucose trasporter-1 and carbonic anhidrase did not highlight any statistical significance (p > 0.05). This is the first report analyzing the association between monocarboxylate transporter expression and the morphological features of BRAF V600E mutated papillary thyroid carcinomas suggesting the possible involvement of lactate in the morphological features.






Similar content being viewed by others
References
M. Xing, BRAF mutation in papillary thyroid cancer: pathogenic role, molecular bases and clinical implications. Endocr. Rev. 28, 742–762 (2007)
T.H. Kim, Y.J. Park, J.A. Lim, H.Y. Ahn et al., The association of the BRAF V600E mutation with prognostic factors and poor clinical outcome in papillary thyroid cancer. Cancer 118, 1764–1773 (2012)
M.N. Nikiforova, Y. Nikiforov, Molecular diagnostics and predictors in thyroid cancer. Thyroid 19, 1351–1361 (2009)
Y.E. Nikiforov, Molecular diagnostics of thyroid tumors. Arch. Pathol. Lab. Med. 135, 569–577 (2011)
V. Trovisco, I. Vieira de Castro, P. Soares et al., BRAF mutations are associated with some histological types of papillary thyroid carcinoma. J. Pathol. 202, 247–251 (2004)
E.D. Rossi, G. Fadda, F. Schmitt, The night mare of indeterminate follicular proliferations: When liquid based cytology and ancillary techniques area not a moon landing but a concrete plan. Acta Cytol. 58, 543–551 (2014)
H. Chen, I. Izevbaye, F. Chen, B. Weinstein, Recent advances in follicular variant of papillary thyroid carcinoma. N. A. J. Med. Sci. 5, 212–219 (2012)
Z. Wang, J.Q. Chen, J.L. Liu, X.G. Qin, Clinical impact of BRAF mutation in the diagnosis and prognosis of papillary thyroid carcinoma: a systematic review and meta-analysis. Eur. J. Clin. Invest. 46, 146–157 (2016)
C.A. Routhier, M.C. Mochel, K. Lynch, D. Dias-Santagata, D.N. Louis, M.P. Hoang, Comparison of 2monoclonal antibodies for immunohistochemical detection of BRAF V600E mutation in malignant melanoma, pulmonary carcinoma, gastrointestinal carcinoma, thyroid carcinoma and gliomas. Human Pathol. 44, 2563–2570 (2013)
A.K. Zimmermann, U. Camerisch, M.P. Rechsteiner, B. Bode-Lesniewska, M. Rossle, Value of immunohistochemistry in detection of BRAF V600E mutations in fine needle aspiration biopsies of papillary thyroid carcinoma. Cancer Cytopathol 122, 48–58 (2014)
E.D. Rossi, M. Martini, S. Capodimonti, B. Angrisani et al., Analysis of immunocytochemistry and molecular BRAF expression in thyroid carcinoma: Cyto-histological institutional experience. Cancer Cytopathol. 122, 527–535 (2014)
R.K. Virk, C.G.A. Theoharis, A. Prasad, D. Chhieng, M.L. Prasad, Morphology predicts BRAFR V600E mutation in papillary thyroid carcinoma: an interobserver reproducibility study. Virchow Arch 464, 435–442 (2014)
A. Finkelstein, G.H. Levy, P. Hui et al., Papillary thyroid carcinomas with and without BRAF V600E mutations are morphological distinct. Histopathology 60, 1052–1059 (2012)
E.D. Rossi, T. Bizzarro, M. Martini et al., Morphologic parameters able to predict BRAF V600E mutated malignancies on thyroid FNAC. Our institutional experience. Cancer Cytopathol. 122, 883–891 (2014)
E.D. Rossi, T. Bizzarro, G. Fadda, L.M. Larocca, F.S. Schmitt, Is morphology alone able to predict BRAF mutated malignancies on thyroid FNAC? Virchows Arch 465, 247–248 (2014)
I. Arozarena, B. Sanchez-Laorden, L. Packer, C. Hidalgo-Carcedo et al., Oncogenic BRAF induces melanoma cell invasion by downregulating the cGMP-specific phosphodiesterase PDE5A. Cancer Cell 19, 45–57 (2011)
A. Hall, K.D. Meyle, M.K. Lange et al., Dysfunctional oxidative phosphorylation makes malignant melanoma cells addicted to glycolysis driven by the V600E BRAF oncogene. Oncotarget 4, 584–599 (2013)
M.H. Lee, S.E. Lee, D.W. Kim et al., Mitochondrial localization and regulation of BRAF V600E in thyroid cancer: a clinically used RAF inhibitor is unable to block the mitochondrial activities of BRAF V600E. J. Clin. Endocrinol. Metab. 96, E19–E30 (2011)
E. White, Exploiting the bad eating habits of RAS-driven cancers. Genes and Develop. 27, 2065–2071 (2015)
A.M. Strohecker, E. White, Targeting mitochondrial metabolism by inhibiting autophagy in BRAF-driven cancers. Cancer Discov. 4, 766–772 (2014)
C. Pinheiro, V. Penna, F. Morais-Santos et al., Characterization of monocarboxylate transporters (MCTs) expression in soft tissue sarcomas:distinct prognostic impact of MCT1 subcellular localization. J. Transl. Med. 12, 118–128 (2014)
C. Pinheiro, A. Longatto-Filho, C. Scapulatempo et al., Increased expression of monocarboxylate transporters 1,2 and 4 in colorectal carcinomas. Virchows Arch 452, 139–146 (2008)
C. Pinheiro, A. Longatto-Filho, K. Simoes et al., The prognostic value of CD147/EMMPRIN is associated with monocarboxylate transporter 1 co-expression in gastric cancer. Eur. J. Cancer 45, 2418–2424 (2009)
C. Pinheiro, A. Albergaria, J. Paredes et al., Monocarboxylate transporter 1 is up-regulated in basal-like breast carcinoma. Histopathology 56, 860–867 (2010)
C. Pinheiro, A. Longatto-Filho, T. Soares et al., CD147 immunohistochemistry discriminates between reactive mesothelial cells and malignant mesothelioma. Diagn. Cytopathol. 40, 478–483 (2012)
C. Pinheiro, A. Longatto-Filho, S.M.M. Pereira et al., Monocarboxylate transporters 1 and 4 are associated with CD147 in cervical carcinoma. Dis. Markers 26, 97–103 (2009)
K. Narumi, A. Furugen, M. Kobayashi, S. Otake, S. Itagaki, K. Iseki, Regulation of monocarboxylate transporter 1 in skeletal muscle cells by intracellular signaling pathways. Biol. Pharm. Bull. 33, 1568–1573 (2010)
E.D. Rossi, M. Martini, S. Capodimonti et al., Diagnostic and prognostic value of immunocytochemistry and BRAF mutation analysis on liquid based biopsies of thyroid neoplasms suspicious for carcinoma. Eur. J. Endocrinol. 168, 853–859 (2013)
British Thyroid Association, Guidelines for the Management of Thyroid Cancer, 2nd edn. London (2007)
G. Fadda, F. Basolo, A. Bondi et al.; SIAPEC-IAP Italian Consensus Working Group, Cytological classification of thyroid nodules. Pathologica 102, 405–408 (2010)
E.S. Cibas, S.Z. Ali, The Bethesda system for reporting thyroid cytopathology. Thyroid 19, 1159–1165 (2009)
E.D. Rossi, M. Martini, S. Capodimonti, C.P. Lombardi et al., BRAF(V600E) mutation analysis on LBC-processed aspiration biopsies predicts bilaterality and nodal involvement in papillary thyroid microcarcinoma. Cancer Cytopathol. 121, 291–297 (2013)
American Joint Commission on Cancer (AJCC), Cancer Staging Atlas 2nd edn. Chicago (2013)
F. Morais-Santos, S. Granja, Miranda-Gonçalves V et al., Targeting lactate transport suppresses in vivo breast tumour growth. Oncotarget 6191, 77–89 (2015)
S. Granja, I. Marchiq, R. Le Floch, C.S. Moura, F. Baltazar, J. Pouysségur, Disruption of BASIGIN decreases lactic acid export and sensitizes non-small cell lung cancer to biguanides independently of the LKB1. Oncotarget 6, 6708–6721 (2015)
C. Pinheiro, A. Longatto-Filho, J. Azevedo-Silva, M. Casal, F.C. Schmitt, F. Baltazar, Role of monocarboxylate transporters in human cancers:state of the art. J. Bioenerg. Biomembr. 44, 127–139 (2012)
Funding
This research did not receive any specific grant from any funding agency in the public, commercial or not-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
The preliminary data of this project were presented as a poster at the 104th USCAP meeting in Boston, 21–27 March 2015
Luigi Maria Larocca, Fatima Baltazar and Fernando Schmitt shared senior authorship.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Rossi, E.D., Bizzarro, T., Granja, S. et al. The expression of monocarboxylate transporters in thyroid carcinoma can be associated with the morphological features of BRAF V600E mutation. Endocrine 56, 379–387 (2017). https://doi.org/10.1007/s12020-016-1044-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-016-1044-0