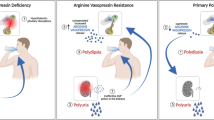
Abstract
In this mini-review, current evidence for how the vasopressin/V2-type receptor/aquaporin axis developed co-evolutionary as a crucial part of the urine-concentrating mechanism will be presented. The present-day human kidney, allowing the concentration of urine up to a maximal osmolality around 1200 mosmol kg−1—or urine to plasma osmolality ratio around 4—with essentially no sodium secreted is the result of up to 3 billion years evolution. Moving from aquatic to terrestrial habitats required profound changes in kidney morphology, most notable the loops of Henle modifying the kidneys from basically a water excretory system to a water conserving system. Vasopressin-like molecules has during the evolution played a significant role in body fluid homeostasis, more specifically, the osmolality of body liquids by controlling the elimination/reabsorption of fluid trough stimulating V2-type receptors to mobilize aquaporin water channels in the renal collector tubules. Recent evidence supports that all components of the vasopressin/V2-type receptor/aquaporin axis can be traced back to early precursors in evolutionary history. The potential clinical and pharmacological implications of a better phylogenetic understanding of these biological systems so essential for body fluid homeostasis relates to any pathological aspects of the urine-concentrating mechanism, in particular deficiencies of any part of the vasopressin-V2R-AQP2 axis causing central or nephrogenic diabetes insipidus—and for broader patient populations also in preventing and treating disturbances in human circadian regulation of urine volume and osmolality that may lead to enuresis and nocturia.

Similar content being viewed by others
References
H.W. Smith, From Fish to Philosopher (Summit, Ciba, 1959)
J.M. Sands, J.P. Kokko, Countercurrent system. Kidney Int. 38(4), 695–699 (1990)
C. Esteva-Font, J. Ballarin, P. Fernández-Llama, Molecular biology of water and salt regulation in the kidney. Cell Mol. Life Sci. 69(5), 683–695 (2012)
G.J. Roch, E.R. Busby, N.M. Sherwood, Evolution of GnRH: diving deeper. Gen. Comp. Endocrinol. 171(1), 1–16 (2011). Epub 2010 Dec 23
G.L. Robertson, Antidiuretic hormone: normal and disordered function. Endocrinol. Metab. Clin. North Am. 30, 671–694 (2001)
C.F. Ferris, Vasopressin/oxytocin and aggression. Novartis Found. Symp. 268, 190–8 (2005). discussion 198–200, 242–53
E.K. Jackson, Vasopressin and Other Agents Affecting the Renal Conservation of Water, in Goodman and Gilman’s The Pharmacological Basis of Therapeutics, 11th edn., ed. by L.L. Brunton, J.S. Lazo, K.L. Parker (McGraw-Hill, Inc., New York, 2001), pp. 771–788
C.H. Hoyle, Neuropeptide families: evolutionary perspectives. Regul. Pept. 73, 1–33 (1998)
R. Acher, J. Chauvet, The neurohypophysial endocrine regulatory cascade: precursors, mediators, receptors, and effectors. Front. Neuroendocrinol. 16(3), 237–289 (1995)
H. Minakata, Oxytocin/vasopressin and gonadotropin-releasing hormone from cephalopods to vertebrates. Ann. N. Y. Acad. Sci. 1200, 33–42 (2010)
R. Acher, Molecular evolution of biologically active polypeptides. Proc. R. Soc. Lond. B Biol. Sci. 210, 21–43 (1980)
S. Hyodo, Y. Kato, M. Ono, A. Urano, Cloning and sequence analyses of cDNAs encoding vasotocin and isotocin precursors of chum salmon, Oncorhynchus keta: evolutionary relationships of neurohypophysial hormone precursors. J. Comp. Physiol. B. 160, 601–608 (1991)
B.B. McEwen, General introduction to vasopressin and oxytocin: Structure/metabolism, evolutionary aspects, neural pathway/receptor distribution, and functional aspects relevant to memory processing. Adv. Pharmacol. 50, 1–50 (2004)
Y.V. Natochin, E.I. Shakhmatova, The origin of the hydroosmotic effect of arginine vasopressin: a hypothesis. Dokl. Biol. Sci. 389, 96–98 (2003)
Y. Takei, A. Kawakoshi, T. Tsukada, S. Yuge, M. Ogoshi, K. Inoue, S. Hyodo, H. Bannai, S. Miyano, Contribution of comparative fish studies to general endocrinology: structure and function of some osmoregulatory hormones. J Exp Zool A Comp Exp Biol. 305(9), 787–798 (2006)
L. Abrami, R. Gobin, V. Berthonaud, H.L. Thanh, J. Chevalier, P. Ripoche, J.M. Verbavatz, Localization of the FA-CHIP water channel in frog urinary bladder. Eur. J. Cell Biol. 73(3), 215–221 (1997)
S.O. Kim, S.H. Song, E.C. Hwang, K.J. Oh, K. Ahn, S.I. Jung, T.W. Kang, D. Kwon, K. Park, S.B. Ryu, Changes in aquaporin (AQP)2 and AQP3 expression in ovariectomized rat urinary bladder: potential implication of water permeability in urinary bladder. World J. Urol. (2011). doi:10.1007/s00345-011-0674-3
I. Böselt, H. Römpler, T. Hermsdorf, D. Thor, W. Busch, A. Schulz, T. Schöneberg, Involvement of the V2 vasopressin receptor in adaptation to limited water supply. PLoS One 4(5), e5573 (2009)
C. Barberis, B. Mouillac, T. Durroux, Structural bases of vasopressin/oxytocin receptor function. J. Endocrinol. 156, 223–229 (1998)
G. Heckel, S. Fink, Evolution of the arginine vasopressin 1a receptor and implications for mammalian social behaviour. Prog. Brain Res. 170, 321–330 (2008)
S. Nielsen, D. Marples, J. Frokiaer, M. Knepper, P. Agre, The aquaporin family of water channels in kidney: an update on physiology and pathophysiology of aquaporin-2. Kidney Int. 49, 1718–1723 (1996)
C.E. Gustafson, T. Katsura, M. McKee, R. Bouley, J.E. Casanova, D. Brown, Recycling of AQP2 occurs through a temperature- and bafilomycin-sensitive trans-Golgi-associated compartment. Am. J. Physiol. Renal Physiol. 278, F317–F326 (2000)
T.X. Sun, A. Van Hoek, Y. Huang, R. Bouley, M. McLaughlin, D. Brown, Aquaporin-2 localization in clathrin-coated pits: inhibition of endocytosis by dominant-negative dynamin. Am. J. Physiol. Renal Physiol. 282, F998–F1011 (2002)
H. Lu, T.X. Sun, R. Bouley, K. Blackburn, M. McLaughlin, D. Brown, Inhibition of endocytosis causes phosphorylation (S256)-independent plasma membrane accumulation of AQP2. Am. J. Physiol. Renal Physiol. 286, F233–F243 (2004)
G.L. Robertson, Disorders of water balance, in Clinical Pediatric Endocrinology, 4th edn., ed. by C.G.D. Brook, P.C. Hindmarsch (Blackwell Science, Oxford, 2001), pp. 193–221
K.V. Juul, D.G. Bichet, J.P. Nørgaard, Desmopressin duration of antidiuretic action in patients with central diabetes insipidus. Endocrine 40(1), 67–74 (2011)
N. Nakamura, Reduced aldehyde dehydrogenase activity and arginine vasopressin receptor 2 expression in the kidneys of male TALLYHO/JngJ mice of prediabetic age. Endocrine 40(3), 379–385 (2011)
T. Sahakitrungruang, S. Wacharasindhu, T. Sinthuwiwat, V. Supornsilchai, K. Suphapeetiporn, V. Shotelersuk, Identification of two novel aquaporin-2 mutations in a Thai girl with congenital nephrogenic diabetes insipidus. Endocrine 33(2), 210–214 (2008)
S. Nielsen, P. Agre, The aquaporin family of water channels in kidney. Kidney Int. 48(4), 1057–1068 (1995)
R.N. Finn, J. Cerdà, Aquaporin evolution in fishes. Front Physiol. 2, 44 (2011)
J.B. Heymann, A. Engel, Aquaporins: phylogeny, structure, and physiology of water channels. News Physiol. Sci. 14, 187–193 (1999)
K. Murata, K. Mitsuoka, T. Hirai, T. Walz, P. Agre, J.B. Heymann, A. Engel, Y. Fujiyoshi, Structural determinants of water permeation through aquaporin-1. Nature 407(6804), 599–605 (2000)
D.X. Fu, A. Libson, L.J.W. Miercke, C. Weitzman, P. Nollert, J. Krucinski, R.M. Stroud, Structure of a glycerol-conducting channel and the basis for its selectivity. Science 290, 481–486 (2000)
E. Kruse, N. Uehlein, R. Kaldenhoff, The aquaporins. Genome Biol. 7(2), 206 (2006). Epub 2006 Feb 28
B. Wu, C. Steinbronn, M. Alsterfjord, T. Zeuthen, E. Beitz, Concerted action of two cation filters in the aquaporin water channel. EMBO J. 28, 2188–2194 (2009)
L.S. King, D. Kozono, P. Agre, From structure to disease: the evolving tale of aquaporin biology. Natl. Rev. Mol. Cell. Biol. 5(9), 687–698 (2004)
K. Ishibashi, S. Kondo, S. Hara, Y. Morishita, The evolutionary aspects of aquaporin family. Am. J. Physiol. Regul. Integr. Comp. Physiol. 300(3), R566–R576 (2011). Epub 2010 Dec 9
D. Gomes, A. Agasse, P. Thiébaud, S. Delrot, H. Gerós, F. Chaumont, Aquaporins are multifunctional water and solute transporters highly divergent in living organisms. Biochim. Biophys. Acta 1788(6), 1213–1228 (2009). Epub 2009 Mar 25
E.M. Campbell, A. Ball, S. Hoppler, A.S. Bowman, Invertebrate aquaporins: a review. J. Comp. Physiol. 178, 935–955 (2008)
D. Gorelick, J. Praetorius, T. Tsunenari, S. Nielsen, P. Agre, Aquaporin-11: a channel protein lacking apparent transport function expressed in brain. BMC Biochem. 7, 14 (2006)
S. Chanprasertyothin, S. Saetung, R. Rajatanavin, B. Ongphiphadhanakul, Genetic variant in the aquaporin 9 gene is associated with bone mineral density in postmenopausal women. Endocrine 38(1), 83–86 (2010). Epub 2010 Jun 18
S. Nielsen, S.R. DiGiovanni, E.I. Christensen et al., Cellular and subcellular immunolocalization of vasopressin-regulated water channel in rat kidney. Proc. Natl. Acad. Sci. USA 90, 11663–11667 (1993)
N. Konno, S. Hyodo, Y. Yamaguchi, K. Matsuda, M. Uchiyama, Vasotocin/V2-type receptor/aquaporin axis exists in African lungfish kidney but is functional only in terrestrial condition. Endocrinology 151(3), 1089–1096 (2010)
Y. Kondo, T. Morimoto, T. Nishio, U.F. Aslanova, M. Nishino, E.I. Farajov, N. Sugawara, N. Kumagai, A. Ohsaga, Y. Maruyama, S. Takahashi, Phylogenetic, ontogenetic, and pathological aspects of the urine-concentrating mechanism. Clin. Exp. Nephrol. 10(3), 165–174 (2006)
Acknowledgments
The author would like to acknowledge and to express gratitude to my mentor, Professor Jens Peter Nørgaard, for encouraging this study and suggesting significant improvements of early drafts of this mini-review.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Juul, K.V. The evolutionary origin of the vasopressin/V2-type receptor/aquaporin axis and the urine-concentrating mechanism. Endocrine 42, 63–68 (2012). https://doi.org/10.1007/s12020-012-9634-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-012-9634-y