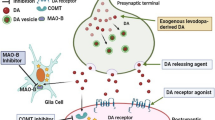
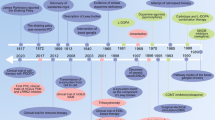
Abstract
Research advances have shed new insight into cellular pathways contributing to PD pathogenesis and offer increasingly compelling therapeutic targets. In this review, we made a broad survey of the published literature that report possible disease-modifying effects on PD. While there are many studies that demonstrate benefits for various therapies for PD in animal and human studies, we confined our search to human “randomised controlled trials” and with the key words “neuroprotection” or “disease-modifying”. It is hoped that through studying the results of these trials, we might clarify possible mechanisms that underlie idiopathic PD. This contrasts with studying the effect of pathophysiology of familial PD, which could be carried out by gene knockouts and animal models. Randomised controlled trials indicate promising effects of MAO-B inhibitors, dopamine agonists, NMDA receptor antagonists, metabotropic glutamate receptor antagonists, therapies related to improving glucose utilization and energy production, therapies related to reduction of excitotoxicity and oxidative stress, statin use, therapies related to iron chelation, therapies related to the use of phytochemicals, and therapies related to physical exercise and brain reward pathway on slowing PD progression. Cumulatively, these approaches fall into two categories: direct enhancement of dopaminergic signalling, and reduction of neurodegeneration. Overlaps between the two categories result in challenges in distinguishing between symptomatic versus disease-modifying effects with current clinical trial designs. Nevertheless, a broad-based approach allows us to consider all possible therapeutic avenues which may be neuroprotective. While the traditional standard of care focuses on symptomatic management with dopaminergic drugs, more recent approaches suggest ways to preserve dopaminergic neurons by attenuating excitotoxicity and oxidative stress.


Similar content being viewed by others
References
Athauda, D., Maclagan, K., Skene, S. S., Bajwa-Joseph, M., Letchford, D., Chowdhury, K., et al. (2017). Exenatide once weekly versus placebo in Parkinson’s disease: A randomised, double-blind, placebo-controlled trial. Lancet, 390(10103), 1664–1675. https://doi.org/10.1016/S0140-6736(17)31585-4
Athauda, D., Maclagan, K., Budnik, N., Zampedri, L., Hibbert, S., Skene, S. S., et al. (2018). What effects might exenatide have on non-motor symptoms in parkinson’s disease: a post hoc analysis. Journal of Parkinson’s Disease, 8(2), 247–258. https://doi.org/10.3233/JPD-181329
Aviles-Olmos, I., Limousin, P., Lees, A., & Foltynie, T. (2013). Parkinson’s disease, insulin resistance and novel agents of neuroprotection. Brain, 136(Pt 2), 374–384. https://doi.org/10.1093/brain/aws009
Aviles-Olmos, I., Dickson, J., Kefalopoulou, Z., Djamshidian, A., Kahan, J., Ell, P., et al. (2014). Motor and cognitive advantages persist 12 months after exenatide exposure in Parkinson’s disease. Journal of Parkinson’s Disease, 4(3), 337–344. https://doi.org/10.3233/JPD-140364
Bartzokis, G., Cummings, J. L., Markham, C. H., Marmarelis, P. Z., Treciokas, L. J., Tishler, T. A., et al. (1999). MRI evaluation of brain iron in earlier- and later-onset Parkinson’s disease and normal subjects. Magnetic Resonance Imaging, 17(2), 213–222. https://doi.org/10.1016/s0730-725x(98)00155-6
Berg, D., Godau, J., Trenkwalder, C., Eggert, K., Csoti, I., Storch, A., et al. (2011). AFQ056 treatment of levodopa-induced dyskinesias: Results of 2 randomized controlled trials. Movement Disorders, 26(7), 1243–1250. https://doi.org/10.1002/mds.23616
Bilgic, B., Pfefferbaum, A., Rohlfing, T., Sullivan, E. V., & Adalsteinsson, E. (2012). MRI estimates of brain iron concentration in normal aging using quantitative susceptibility mapping. NeuroImage, 59(3), 2625–2635. https://doi.org/10.1016/j.neuroimage.2011.08.077
Burke, R. E., & O’Malley, K. (2013). Axon degeneration in Parkinson’s disease. Experimental Neurology, 246, 72–83. https://doi.org/10.1016/j.expneurol.2012.01.011
Carroll, C. B., & Wyse, R. K. H. (2017). Simvastatin as a potential disease-modifying therapy for patients with parkinson’s disease: Rationale for clinical trial, and current progress. Journal of Parkinson’s Disease, 7(4), 545–568. https://doi.org/10.3233/JPD-171203
Chen, H., Huang, X., Guo, X., Mailman, R. B., Park, Y., Kamel, F., et al. (2010). Smoking duration, intensity, and risk of Parkinson disease. Neurology, 74(11), 878–884. https://doi.org/10.1212/WNL.0b013e3181d55f38
Chen, R., Hu, Z., Wei, L., Ma, Y., Liu, Z., & Copeland, J. R. (2011). Incident dementia in a defined older Chinese population. PLoS ONE, 6(9), e24817. https://doi.org/10.1371/journal.pone.0024817
Chergui, K., Akaoka, H., Charlety, P. J., Saunier, C. F., Buda, M., & Chouvet, G. (1994). Subthalamic nucleus modulates burst firing of nigral dopamine neurones via NMDA receptors. NeuroReport, 5(10), 1185–1188. https://doi.org/10.1097/00001756-199406020-00006
Corcos, D. M., Robichaud, J. A., David, F. J., Leurgans, S. E., Vaillancourt, D. E., Poon, C., et al. (2013). A two-year randomized controlled trial of progressive resistance exercise for Parkinson’s disease. Movement Disorders, 28(9), 1230–1240. https://doi.org/10.1002/mds.25380
Costa, J., Lunet, N., Santos, C., Santos, J., & Vaz-Carneiro, A. (2010). Caffeine exposure and the risk of Parkinson’s disease: A systematic review and meta-analysis of observational studies. Journal of Alzheimer’s Disease, 20(Suppl 1), S221-238. https://doi.org/10.3233/JAD-2010-091525
Damier, P., Hirsch, E. C., Agid, Y., & Graybiel, A. M. (1999). The substantia nigra of the human brain. II. Patterns of loss of dopamine-containing neurons in Parkinson’s disease. Brain, 122, 1437–1448. https://doi.org/10.1093/brain/122.8.1437
Daneshvar Kakhaki, R., Ostadmohammadi, V., Kouchaki, E., Aghadavod, E., Bahmani, F., Tamtaji, O. R., et al. (2020). Melatonin supplementation and the effects on clinical and metabolic status in Parkinson’s disease: A randomized, double-blind, placebo-controlled trial. Clinical Neurology and Neurosurgery, 195, 105878. https://doi.org/10.1016/j.clineuro.2020.105878
Daugherty, A., & Raz, N. (2013). Age-related differences in iron content of subcortical nuclei observed in vivo: A meta-analysis. NeuroImage, 70, 113–121. https://doi.org/10.1016/j.neuroimage.2012.12.040
David, F. J., Robichaud, J. A., Leurgans, S. E., Poon, C., Kohrt, W. M., Goldman, J. G., et al. (2015). Exercise improves cognition in Parkinson’s disease: The PRET-PD randomized, clinical trial. Movement Disorders, 30(12), 1657–1663. https://doi.org/10.1002/mds.26291
Dawson, T. M., & Dawson, V. L. (2003). Molecular pathways of neurodegeneration in Parkinson’s disease. Science, 302(5646), 819–822. https://doi.org/10.1126/science.1087753
Del Dotto, P., Pavese, N., Gambaccini, G., Bernardini, S., Metman, L. V., Chase, T. N., et al. (2001). Intravenous amantadine improves levadopa-induced dyskinesias: An acute double-blind placebo-controlled study. Movement Disorders, 16(3), 515–520. https://doi.org/10.1002/mds.1112
Devi, L., Raghavendran, V., Prabhu, B. M., Avadhani, N. G., & Anandatheerthavarada, H. K. (2008). Mitochondrial import and accumulation of alpha-synuclein impair complex I in human dopaminergic neuronal cultures and Parkinson disease brain. Journal of Biological Chemistry, 283(14), 9089–9100. https://doi.org/10.1074/jbc.M710012200
Devos, D., Moreau, C., Devedjian, J. C., Kluza, J., Petrault, M., Laloux, C., et al. (2014). Targeting chelatable iron as a therapeutic modality in Parkinson’s disease. Antioxidants & Redox Signaling, 21(2), 195–210. https://doi.org/10.1089/ars.2013.5593
Dias, V., Junn, E., & Mouradian, M. M. (2013). The role of oxidative stress in Parkinson’s disease. Journal of Parkinson’s Disease, 3(4), 461–491. https://doi.org/10.3233/JPD-130230
DiPiro, J., Talbert, R. L., Yee, G. C., Matzke, G. R., Wells, B. G., & Posey, L. M. (2017). Pharmacotherapy. A Pathophysiological Approach (10th (Edition). McGraw Hill.
Duncan, R. P., & Earhart, G. M. (2012). Randomized controlled trial of community-based dancing to modify disease progression in Parkinson disease. Neurorehabilitation and Neural Repair, 26(2), 132–143. https://doi.org/10.1177/1545968311421614
Fahn, S., Oakes, D., Shoulson, I., Kieburtz, K., Rudolph, A., Lang, A., et al. (2004). Levodopa and the progression of Parkinson’s disease. New England Journal of Medicine, 351(24), 2498–2508. https://doi.org/10.1056/NEJMoa033447
Farooqui, A. A. (2012) Therapeutic Potentials of Curcumin for Alzheimer Disease: Springer Science-Business Media
Fox, S. H., Katzenschlager, R., Lim, S. Y., Ravina, B., Seppi, K., Coelho, M., et al. (2011). The movement disorder society evidence-based medicine review update: Treatments for the motor symptoms of Parkinson’s disease. Movement Disorders, 26(Suppl 3), S2-41. https://doi.org/10.1002/mds.23829
Frazzitta, G., Maestri, R., Bertotti, G., Riboldazzi, G., Boveri, N., Perini, M., et al. (2015). Intensive rehabilitation treatment in early Parkinson’s disease: A randomized pilot study with a 2-year follow-up. Neurorehabilitation and Neural Repair, 29(2), 123–131. https://doi.org/10.1177/1545968314542981
Grolez, G., Moreau, C., Sablonniere, B., Garcon, G., Devedjian, J. C., Meguig, S., et al. (2015). Ceruloplasmin activity and iron chelation treatment of patients with Parkinson’s disease. BMC Neurology, 15, 74. https://doi.org/10.1186/s12883-015-0331-3
Haber, S. N., & Gdowski, M. J. (2004). The Basal Ganglia. In G. Paxinos & J. K. Mai (Eds.), The Human Nervous System (2nd (Edition, pp. 676–738). Elsevier.
Hauser, R. A., Lew, M. F., Hurtig, H. I., Ondo, W. G., Wojcieszek, J., Fitzer-Attas, C. J., et al. (2009). Long-term outcome of early versus delayed rasagiline treatment in early Parkinson’s disease. Movement Disorders, 24(4), 564–573. https://doi.org/10.1002/mds.22402
Hauser, R. A., Li, R., Perez, A., Ren, X., Weintraub, D., Elm, J., et al. (2017). Longer Duration of MAO-B Inhibitor Exposure is Associated with Less Clinical Decline in Parkinson’ Disease: An Analysis of NET-PD LS1. Journal of Parkinson’s Disease, 7(1), 117–127. https://doi.org/10.3233/JPD-160965
He, X., Jenner, A. M., Ong, W. Y., Farooqui, A. A., & Patel, S. C. (2006). Lovastatin modulates increased cholesterol and oxysterol levels and has a neuroprotective effect on rat hippocampal neurons after kainate injury. Journal of Neuropathology and Experimental Neurology, 65(7), 652–663. https://doi.org/10.1097/01.jnen.0000225906.82428.69
Hollmann, M., Hartley, M., & Heinemann, S. (1991). Ca2+ permeability of KA-AMPA–gated glutamate receptor channels depends on subunit composition. Science, 252(5007), 851–853. https://doi.org/10.1126/science.1709304
Jellinger, K. A. (2014). The pathomechanisms underlying Parkinson’s disease. Expert Review of Neurotherapeutics, 14(2), 199–215. https://doi.org/10.1586/14737175.2014.877842
Johnson, K. A., Conn, P. J., & Niswender, C. M. (2009). Glutamate receptors as therapeutic targets for Parkinson’s disease. CNS & Neurological Disorders: Drug Targets, 8(6), 475–491. https://doi.org/10.2174/187152709789824606
Kandel, E. R., Schwartz, J. H., Jessell, T. M., Siegelbaum, S. A., & Hudspeth, A. J. (2013) Principles of Neural Science (5th Edition ed.) McGraw-Hill
Keath, J. R., Iacoviello, M. P., Barrett, L. E., Mansvelder, H. D., & McGehee, D. S. (2007). Differential modulation by nicotine of substantia nigra versus ventral tegmental area dopamine neurons. Journal of Neurophysiology, 98(6), 3388–3396. https://doi.org/10.1152/jn.00760.2007
Koob, G. F., & Volkow, N. D. (2016). Neurobiology of addiction: A neurocircuitry analysis. Lancet Psychiatry, 3(8), 760–773. https://doi.org/10.1016/S2215-0366(16)00104-8
Landers, M. R., Navalta, J. W., Murtishaw, A. S., Kinney, J. W., & Pirio Richardson, S. (2019). A high-intensity exercise boot camp for persons with parkinson disease: A phase II, pragmatic, randomized clinical trial of feasibility, safety, signal of efficacy, and disease mechanisms. Journal of Neurologic Physical Therapy, 43(1), 12–25. https://doi.org/10.1097/NPT.0000000000000249
Lavoie, B., & Parent, A. (1991). Dopaminergic neurons expressing calbindin in normal and parkinsonian monkeys. NeuroReport, 2(10), 601–604. https://doi.org/10.1097/00001756-199110000-00012
Lee, Y. C., Lin, C. H., Wu, R. M., Lin, M. S., Lin, J. W., Chang, C. H., et al. (2013). Discontinuation of statin therapy associates with Parkinson disease: A population-based study. Neurology, 81(5), 410–416. https://doi.org/10.1212/WNL.0b013e31829d873c
Leehey, M., Luo, S., Sharma, S., Wills, A. A., Bainbridge, J. L., Wong, P. S., et al. (2017). Association of metabolic syndrome and change in unified parkinson’s disease rating scale scores. Neurology, 89(17), 1789–1794. https://doi.org/10.1212/WNL.0000000000004572
Lees, A., Fahn, S., Eggert, K. M., Jankovic, J., Lang, A., Micheli, F., et al. (2012). Perampanel, an AMPA antagonist, found to have no benefit in reducing “off” time in Parkinson’s disease. Movement Disorders, 27(2), 284–288. https://doi.org/10.1002/mds.23983
Li, M., Shi, A., Pang, H., Xue, W., Li, Y., Cao, G., et al. (2014). Safety, tolerability, and pharmacokinetics of a single ascending dose of baicalein chewable tablets in healthy subjects. Journal of Ethnopharmacology, 156, 210–215. https://doi.org/10.1016/j.jep.2014.08.031
Li, Z., Wang, P., Yu, Z., Cong, Y., Sun, H., Zhang, J., et al. (2015). The effect of creatine and coenzyme q10 combination therapy on mild cognitive impairment in Parkinson’s disease. European Neurology, 73(3–4), 205–211. https://doi.org/10.1159/000377676
Lipton, S. A. (2004). Paradigm shift in NMDA receptor antagonist drug development: Molecular mechanism of uncompetitive inhibition by memantine in the treatment of Alzheimer’s disease and other neurologic disorders. Journal of Alzheimer’s Disease, 6(6 Suppl), S61-74. https://doi.org/10.3233/jad-2004-6s610
Miyai, I., Fujimoto, Y., Ueda, Y., Yamamoto, H., Nozaki, S., Saito, T., et al. (2000). Treadmill training with body weight support: Its effect on Parkinson’s disease. Archives of Physical Medicine and Rehabilitation, 81(7), 849–852. https://doi.org/10.1053/apmr.2000.4439
Monti, D. A., Zabrecky, G., Kremens, D., Liang, T. W., Wintering, N. A., Cai, J., et al. (2016). N-acetyl cysteine may support dopamine neurons in parkinson’s disease: preliminary clinical and cell line data. PLoS ONE, 11(6), e0157602. https://doi.org/10.1371/journal.pone.0157602
Monti, D. A., Zabrecky, G., Kremens, D., Liang, T. W., Wintering, N. A., Bazzan, A. J., et al. (2019). N-Acetyl cysteine is associated with dopaminergic improvement in parkinson’s disease. Clinical Pharmacology and Therapeutics, 106(4), 884–890. https://doi.org/10.1002/cpt.1548
Moreau, C., Delval, A., Tiffreau, V., Defebvre, L., Dujardin, K., Duhamel, A., et al. (2013). Memantine for axial signs in Parkinson’s disease: A randomised, double-blind, placebo-controlled pilot study. Journal of Neurology, Neurosurgery and Psychiatry, 84(5), 552–555. https://doi.org/10.1136/jnnp-2012-303182
Novak, P., Pimentel Maldonado, D. A., & Novak, V. (2019). Safety and preliminary efficacy of intranasal insulin for cognitive impairment in Parkinson disease and multiple system atrophy: A double-blinded placebo-controlled pilot study. PLoS ONE, 14(4), e0214364. https://doi.org/10.1371/journal.pone.0214364
Oertel, W., Eggert, K., Pahwa, R., Tanner, C. M., Hauser, R. A., Trenkwalder, C., et al. (2017). Randomized, placebo-controlled trial of ADS-5102 (amantadine) extended-release capsules for levodopa-induced dyskinesia in Parkinson’s disease (EASE LID 3). Movement Disorders, 32(12), 1701–1709. https://doi.org/10.1002/mds.27131
Olanow, C. W., Rascol, O., Hauser, R., Feigin, P. D., Jankovic, J., Lang, A., et al. (2009). A double-blind, delayed-start trial of rasagiline in Parkinson’s disease. New England Journal of Medicine, 361(13), 1268–1278. https://doi.org/10.1056/NEJMoa0809335
Ong, W. Y., He, Y., & Garey, L. J. (1997). Localisation of glutamate receptors in the substantia nigra pars compacta of the monkey. Journal Fur Hirnforschung, 38(3), 291–298.
Ong, W. Y., Tanaka, K., Dawe, G. S., Ittner, L. M., & Farooqui, A. A. (2013). Slow excitotoxicity in Alzheimer’s disease. Journal of Alzheimer’s Disease, 35(4), 643–668. https://doi.org/10.3233/JAD-121990
Ong, W. Y., Wu, Y. J., Farooqui, T., & Farooqui, A. A. (2018). Qi Fu Yin-a Ming dynasty prescription for the treatment of dementia. Molecular Neurobiology, 55(9), 7389–7400. https://doi.org/10.1007/s12035-018-0908-0
Ong, W. Y., Farooqui, T., Ho, C. F. Y., Ng, Y. K., & Farooqui, A. A. (2017) Use of Phytochemicals against Neuroinflammation. In Neuroprotective Effects of Phytochemicals in Neurological Disorders: Wiley
Pahwa, R., Factor, S. A., Lyons, K. E., Ondo, W. G., Gronseth, G., Bronte-Stewart, H., et al. (2006). Practice Parameter: Treatment of Parkinson disease with motor fluctuations and dyskinesia (an evidence-based review): report of the quality standards subcommittee of the american academy of neurology. Neurology, 66(7), 983–995. https://doi.org/10.1212/01.wnl.0000215250.82576.87
Palhagen, S., Heinonen, E., Hagglund, J., Kaugesaar, T., Maki-Ikola, O., Palm, R., et al. (2006). Selegiline slows the progression of the symptoms of Parkinson disease. Neurology, 66(8), 1200–1206. https://doi.org/10.1212/01.wnl.0000204007.46190.54
Paquet, M., Tremblay, M., Soghomonian, J. J., & Smith, Y. (1997). AMPA and NMDA glutamate receptor subunits in midbrain dopaminergic neurons in the squirrel monkey: An immunohistochemical and in situ hybridization study. Journal of Neuroscience, 17(4), 1377–1396. https://doi.org/10.1523/jneurosci.17-04-01377.1997
Petramfar, P., Hajari, F., Yousefi, G., Azadi, S., & Hamedi, A. (2020). Efficacy of oral administration of licorice as an adjunct therapy on improving the symptoms of patients with Parkinson’s disease, A randomized double blinded clinical trial. Journal of Ethnopharmacology, 247, 112226. https://doi.org/10.1016/j.jep.2019.112226
Pringsheim, T., Jette, N., Frolkis, A., & Steeves, T. D. (2014). The prevalence of Parkinson’s disease: A systematic review and meta-analysis. Movement Disorders, 29(13), 1583–1590. https://doi.org/10.1002/mds.25945
Rascol, O., Fitzer-Attas, C. J., Hauser, R., Jankovic, J., Lang, A., Langston, J. W., et al. (2011). A double-blind, delayed-start trial of rasagiline in Parkinson’s disease (the ADAGIO study): Prespecified and post-hoc analyses of the need for additional therapies, changes in UPDRS scores, and non-motor outcomes. Lancet Neurology, 10(5), 415–423. https://doi.org/10.1016/s1474-4422(11)70073-4
Ren, M. Q., Ong, W. Y., Wang, X. S., & Watt, F. (2003). A nuclear microscopic and histochemical study of iron concentrations and distribution in the midbrain of two age groups of monkeys unilaterally injected with MPTP. Experimental Neurology, 184(2), 947–954. https://doi.org/10.1016/S0014-4886(03)00341-8
Ricciardi, L., De Nigris, F., Specchia, A., & Fasano, A. (2015). Homotaurine in Parkinson’s disease. Neurological Sciences, 36(9), 1581–1587. https://doi.org/10.1007/s10072-015-2201-6
Rodriguez, M. C., Obeso, J. A., & Olanow, C. W. (1998). Subthalamic nucleus-mediated excitotoxicity in Parkinson’s disease: A target for neuroprotection. Annals of Neurology, 44(3 Suppl 1), S175-188. https://doi.org/10.1002/ana.410440726
Rui, W., Li, S., Xiao, H., Xiao, M., & Shi, J. (2020). Baicalein Attenuates Neuroinflammation by Inhibiting NLRP3/caspase-1/GSDMD Pathway in MPTP Induced Mice Model of Parkinson’s Disease. International Journal of Neuropsychopharmacology. https://doi.org/10.1093/ijnp/pyaa060
Schapira, A. H., McDermott, M. P., Barone, P., Comella, C. L., Albrecht, S., Hsu, H. H., et al. (2013). Pramipexole in patients with early Parkinson’s disease (PROUD): A randomised delayed-start trial. Lancet Neurology, 12(8), 747–755. https://doi.org/10.1016/s1474-4422(13)70117-0
Schenkman, M., Moore, C. G., Kohrt, W. M., Hall, D. A., Delitto, A., Comella, C. L., et al. (2018). Effect of high-intensity treadmill exercise on motor symptoms in patients with de novo parkinson disease: A Phase 2 randomized clinical trial. JAMA Neurology, 75(2), 219–226. https://doi.org/10.1001/jamaneurol.2017.3517
Schwarzschild, M. A., Schwid, S. R., Marek, K., Watts, A., Lang, A. E., Oakes, D., et al. (2008). Serum urate as a predictor of clinical and radiographic progression in Parkinson disease. Archives of Neurology, 65(6), 716–723. https://doi.org/10.1001/archneur.2008.65.6.nct70003
Schwarzschild, M. A., Ascherio, A., Beal, M. F., Cudkowicz, M. E., Curhan, G. C., Hare, J. M., et al. (2014). Inosine to increase serum and cerebrospinal fluid urate in Parkinson disease: A randomized clinical trial. JAMA Neurology, 71(2), 141–150. https://doi.org/10.1001/jamaneurol.2013.5528
Seet, R. C., Lee, C. Y., Lim, E. C., Tan, J. J., Quek, A. M., Chong, W. L., et al. (2010). Oxidative damage in Parkinson disease: Measurement using accurate biomarkers. Free Radical Biology & Medicine, 48(4), 560–566. https://doi.org/10.1016/j.freeradbiomed.2009.11.026
Shoulson, I. (1992). An interim report of the effect of selegiline (L-deprenyl) on the progression of disability in early Parkinson’s disease. The Parkinson Study Group. Eur Neurol, 32(Suppl 1), 46–53. https://doi.org/10.1159/000116869
Shoulson, I. (1998). DATATOP: a decade of neuroprotective inquiry. Parkinson Study Group. Deprenyl And Tocopherol Antioxidative Therapy Of Parkinsonism. Annals of Neurology, 44(3), S160-166.
Shoulson, I., Penney, J., McDermott, M., Schwid, S., Kayson, E., Chase, T., et al. (2001). A randomized, controlled trial of remacemide for motor fluctuations in Parkinson’s disease. Neurology, 56(4), 455–462. https://doi.org/10.1212/wnl.56.4.455
Silveira, C. R. A., MacKinley, J., Coleman, K., Li, Z., Finger, E., Bartha, R., et al. (2019). Ambroxol as a novel disease-modifying treatment for Parkinson’s disease dementia: Protocol for a single-centre, randomized, double-blind, placebo-controlled trial. BMC Neurology, 19(1), 20. https://doi.org/10.1186/s12883-019-1252-3
Smith, Y., Bevan, M. D., Shink, E., & Bolam, J. P. (1998). Microcircuitry of the direct and indirect pathways of the basal ganglia. Neuroscience, 86(2), 353–387. https://doi.org/10.1016/s0306-4522(98)00004-9
Snow, B. J., Rolfe, F. L., Lockhart, M. M., Frampton, C. M., O’Sullivan, J. D., Fung, V., et al. (2010). A double-blind, placebo-controlled study to assess the mitochondria-targeted antioxidant MitoQ as a disease-modifying therapy in Parkinson’s disease. Movement Disorders, 25(11), 1670–1674. https://doi.org/10.1002/mds.23148
Srikanth, M., Chandrasaharan, K., Zhao, X., Chayaburakul, K., Ong, W. Y., & Herr, D. R. (2018). Metabolism of Docosahexaenoic Acid (DHA) Induces Pyroptosis in BV-2 Microglial Cells. Neuromolecular Medicine, 20(4), 504–514. https://doi.org/10.1007/s12017-018-8511-0
Stocchi, F., Rascol, O., Destee, A., Hattori, N., Hauser, R. A., Lang, A. E., et al. (2013). AFQ056 in Parkinson patients with levodopa-induced dyskinesia: 13-week, randomized, dose-finding study. Movement Disorders, 28(13), 1838–1846. https://doi.org/10.1002/mds.25561
Storch, A., Jost, W. H., Vieregge, P., Spiegel, J., Greulich, W., Durner, J., et al. (2007). Randomized, double-blind, placebo-controlled trial on symptomatic effects of coenzyme Q(10) in Parkinson disease. Archives of Neurology, 64(7), 938–944. https://doi.org/10.1001/archneur.64.7.nct60005
Stubendorff, K., Larsson, V., Ballard, C., Minthon, L., Aarsland, D., & Londos, E. (2014). Treatment effect of memantine on survival in dementia with Lewy bodies and Parkinson’s disease with dementia: A prospective study. British Medical Journal Open, 4(7), e005158. https://doi.org/10.1136/bmjopen-2014-005158
Tabi, T., Vecsei, L., Youdim, M. B., Riederer, P., & Szoko, E. (2020). Selegiline: A molecule with innovative potential. Journal of Neural Transmission (vienna), 127(5), 831–842. https://doi.org/10.1007/s00702-019-02082-0
Tison, F., Keywood, C., Wakefield, M., Durif, F., Corvol, J. C., Eggert, K., et al. (2016). A Phase 2A Trial of the Novel mGluR5-Negative allosteric modulator dipraglurant for levodopa-induced dyskinesia in parkinson’s disease. Movement Disorders, 31(9), 1373–1380. https://doi.org/10.1002/mds.26659
van der Kolk, N. M., de Vries, N. M., Kessels, R. P. C., Joosten, H., Zwinderman, A. H., Post, B., et al. (2019). Effectiveness of home-based and remotely supervised aerobic exercise in Parkinson’s disease: A double-blind, randomised controlled trial. Lancet Neurology, 18(11), 998–1008. https://doi.org/10.1016/S1474-4422(19)30285-6
Verdoorn, T. A., Burnashev, N., Monyer, H., Seeburg, P. H., & Sakmann, B. (1991). Structural determinants of ion flow through recombinant glutamate receptor channels. Science, 252(5013), 1715–1718. https://doi.org/10.1126/science.1710829
Verschuur, C. V. M., Suwijn, S. R., Boel, J. A., Post, B., Bloem, B. R., van Hilten, J. J., et al. (2019). Randomized Delayed-Start Trial of Levodopa in Parkinson’s Disease. New England Journal of Medicine, 380(4), 315–324. https://doi.org/10.1056/NEJMoa1809983
Volpe, D., Signorini, M., Marchetto, A., Lynch, T., & Morris, M. E. (2013). A comparison of Irish set dancing and exercises for people with Parkinson’s disease: A phase II feasibility study. BMC Geriatrics, 13(1), 54. https://doi.org/10.1186/1471-2318-13-54
Vranesic, I., Ofner, S., Flor, P. J., Bilbe, G., Bouhelal, R., Enz, A., et al. (2014). AFQ056/mavoglurant, a novel clinically effective mGluR5 antagonist: Identification, SAR and pharmacological characterization. Bioorganic & Medicinal Chemistry, 22(21), 5790–5803. https://doi.org/10.1016/j.bmc.2014.09.033
Whone, A. L., Watts, R. L., Stoessl, A. J., Davis, M., Reske, S., Nahmias, C., et al. (2003). Slower progression of Parkinson’s disease with ropinirole versus levodopa: The REAL-PET study. Annals of Neurology, 54(1), 93–101. https://doi.org/10.1002/ana.10609
Yoritaka, A., Kawajiri, S., Yamamoto, Y., Nakahara, T., Ando, M., Hashimoto, K., et al. (2015). Randomized, double-blind, placebo-controlled pilot trial of reduced coenzyme Q10 for Parkinson’s disease. Parkinsonism & Related Disorders, 21(8), 911–916. https://doi.org/10.1016/j.parkreldis.2015.05.022
Yuan, Y., Rangarajan, P., Kan, E. M., Wu, Y., Wu, C., & Ling, E. A. (2015). Scutellarin regulates the Notch pathway and affects the migration and morphological transformation of activated microglia in experimentally induced cerebral ischemia in rats and in activated BV-2 microglia. Journal of Neuroinflammation, 12, 11. https://doi.org/10.1186/s12974-014-0226-z
Zarmouh, N. O., Messeha, S. S., Elshami, F. M., & Soliman, K. F. (2016). Natural Products Screening for the Identification of Selective Monoamine Oxidase-B Inhibitors. European Journal of Medicinal Plants, 15(1), 1–16. https://doi.org/10.9734/EJMP/2016/26453
Acknowledgements
This work was supported by grants from the Ministry of Education (NUHSRO/2019/051/T1/Seed-Mar/04) and the National Medical Research Council of Singapore (HLCA21Jan-0019) and A*STAR Singapore.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interests to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ong, WY., Leow, D.MK., Herr, D.R. et al. What Do Randomized Controlled Trials Inform Us About Potential Disease-Modifying Strategies for Parkinson’s Disease?. Neuromol Med 25, 1–13 (2023). https://doi.org/10.1007/s12017-022-08718-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-022-08718-x