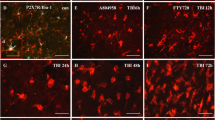
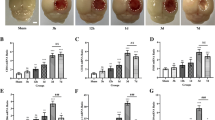
Abstract
The tripeptide glycine–proline–glutamate analogue NNZ-2566 (Neuren Pharmaceuticals) demonstrates neuroprotective efficacy in models of traumatic brain injury. In penetrating ballistic-like brain injury (PBBI), it significantly decreases injury-induced upregulation of inflammatory cytokines including TNF-α, IFN-γ, and IL-6. However, the mechanism by which NNZ-2566 acts has yet to be determined. The activating transcription factor-3 (ATF3) is known to repress expression of these inflammatory cytokines and was increased at the mRNA and protein level 24-h post-PBBI. This study investigated whether 12 h of NNZ-2566 treatment following PBBI alters atf3 expression. PBBI alone significantly increased atf3 mRNA levels by 13-fold at 12 h and these levels were increased by an additional fourfold with NNZ-2566 treatment. To confirm that changes in mRNA translated to changes in protein expression, ATF3 expression levels were determined in vivo in microglia/macrophages, T cells, natural killer cells (NKCs), astrocytes, and neurons. PBBI alone significantly increased ATF3 in microglia/macrophages (820 %), NKCs (58 %), and astrocytes (51 %), but decreased levels in T cells (48 %). NNZ-2566 treatment further increased ATF3 protein expression in microglia/macrophages (102 %), NKCs (308 %), and astrocytes (13 %), while reversing ATF3 decreases in T cells. Finally, PBBI increased ATF3 levels by 55 % in neurons and NNZ-2566 treatment further increased these levels an additional 33 %. Since increased ATF3 may be an innate protective mechanism to limit inflammation following injury, these results demonstrating that the anti-inflammatory and neuroprotective drug NNZ-2566 increase both mRNA and protein levels of ATF3 in multiple cell types provide a cellular mechanism for NNZ-2566 modulation of neuroinflammation following PBBI.








Similar content being viewed by others
References
Amiry-Moghaddam, M., Otsuka, T., Hurn, P. D., Traystman, R. J., Haug, F. M., Froehner, S. C., et al. (2003). An alpha-syntrophin-dependent pool of AQP4 in astroglial end-feet confers bidirectional water flow between blood and brain. Proceedings of the National Academy of Sciences of the United States of America, 100(4), 2106–2111.
Bickerdike, M. J., Thomas, G. B., Batchelor, D. C., Sirimanne, E. S., Leong, W., Lin, H., et al. (2009). NNZ-2566: A Gly-Pro-Glu analogue with neuroprotective efficacy in a rat model of acute focal stroke. Journal of the Neurological Sciences, 278(1–2), 85–90.
Bienvenu, T. C., Busti, D., Magill, P. J., Ferraguti, F., & Capogna, M. (2012). Cell-type-specific recruitment of amygdala interneurons to hippocampal theta rhythm and noxious stimuli in vivo. [Research Support, Non-U.S. Gov’t]. Neuron, 74(6), 1059–1074.
Byram, S. C., Serpe, C. J., Pruett, S. B., Sanders, V. M., & Jones, K. J. (2003). Natural killer cells do not mediate facial motoneuron survival after facial nerve transection. Brain, Behavior, and Immunity, 17(6), 417–425.
Cartagena, C. M., Ahmed, F., Burns, M. P., Pajoohesh-Ganji, A., Pak, D. T., Faden, A. I., et al. (2008). Cortical injury increases cholesterol 24S hydroxylase (Cyp46) levels in the rat brain. Journal of Neurotrauma, 25(9), 1087–1098.
Chen, B. P., Liang, G., Whelan, J., & Hai, T. (1994). ATF3 and ATF3 delta Zip. Transcriptional repression versus activation by alternatively spliced isoforms. Journal of Biological Chemistry, 269(22), 15819–15826.
Chen, Y., & Swanson, R. A. (2003). Astrocytes and brain injury. Journal of Cerebral Blood Flow and Metabolism, 23(2), 137–149.
Dong, Y., & Benveniste, E. N. (2001). Immune function of astrocytes. Glia, 36(2), 180–190.
Efficacy and Safety Study of Intravenous Progesterone in Patients with Severe Traumatic Brain Injury (SyNAPSe). (2010). BHR Pharma, LLC. Accessed July 9, 2012 from http://www.clinicaltrials.gov/ct2/show/NCT01143064?term=nct01143064&rank=1.
Erythropoietin in Traumatic Brain Injury (EPO-TBI). (2009). Australian and New Zealand Intensive Care Research Centre. Accessed July 9, 2012 from http://www.clinicaltrials.gov/ct2/show/NCT00987454?term=nct00987454&rank=1.
Fauriat, C., Long, E. O., Ljunggren, H. G., & Bryceson, Y. T. (2010). Regulation of human NK-cell cytokine and chemokine production by target cell recognition. Blood, 115(11), 2167–2176.
Francis, J. S., Dragunow, M., & During, M. J. (2004). Over expression of ATF-3 protects rat hippocampal neurons from in vivo injection of kainic acid. Brain Research. Molecular Brain Research, 124(2), 199–203.
Gilchrist, M., Thorsson, V., Li, B., Rust, A. G., Korb, M., Roach, J. C., et al. (2006). Systems biology approaches identify ATF3 as a negative regulator of Toll-like receptor 4. Nature, 441(7090), 173–178.
Hammarberg, H., Lidman, O., Lundberg, C., Eltayeb, S. Y., Gielen, A. W., Muhallab, S., et al. (2000). Neuroprotection by encephalomyelitis: Rescue of mechanically injured neurons and neurotrophin production by CNS-infiltrating T and natural killer cells. Journal of Neuroscience, 20(14), 5283–5291.
Harty, J. T., Tvinnereim, A. R., & White, D. W. (2000). CD8+ T cell effector mechanisms in resistance to infection. Annual Review of Immunology, 18, 275–308.
Israelsson, C., Bengtsson, H., Kylberg, A., Kullander, K., Lewen, A., Hillered, L., et al. (2008). Distinct cellular patterns of upregulated chemokine expression supporting a prominent inflammatory role in traumatic brain injury. Journal of Neurotrauma, 25(8), 959–974.
Lau, L. T., & Yu, A. C. (2001). Astrocytes produce and release interleukin-1, interleukin-6, tumor necrosis factor alpha and interferon-gamma following traumatic and metabolic injury. Journal of Neurotrauma, 18(3), 351–359.
Litvak, V., Ramsey, S. A., Rust, A. G., Zak, D. E., Kennedy, K. A., Lampano, A. E., et al. (2009). Function of C/EBPdelta in a regulatory circuit that discriminates between transient and persistent TLR4-induced signals. Nature Immunology, 10(4), 437–443.
Lu, X. C., Chen, R. W., Yao, C., Wei, H., Yang, X., Liao, Z., et al. (2009a). NNZ-2566, a glypromate analog, improves functional recovery and attenuates apoptosis and inflammation in a rat model of penetrating ballistic-type brain injury. Journal of Neurotrauma, 26(1), 141–154.
Lu, X. C., Hartings, J. A., Si, Y., Balbir, A., Cao, Y., & Tortella, F. C. (2011). Electrocortical pathology in a rat model of penetrating ballistic-like brain injury. Journal of Neurotrauma, 28(1), 71–83.
Lu, X. C., Si, Y., Williams, A. J., Hartings, J. A., Gryder, D., & Tortella, F. C. (2009b). NNZ-2566, a glypromate analog, attenuates brain ischemia-induced non-convulsive seizures in rats. Journal of Cerebral Blood Flow and Metabolism, 29(12), 1924–1932.
Massi, L., Lagler, M., Hartwich, K., Borhegyi, Z., Somogyi, P., & Klausberger, T. (2012). Temporal dynamics of parvalbumin-expressing axo-axonic and basket cells in the rat medial prefrontal cortex in vivo. [Research Support, Non-U.S. Gov’t]. Journal of Neuroscience, 32(46), 16496–16502.
Matsumoto, Y., Kohyama, K., Aikawa, Y., Shin, T., Kawazoe, Y., Suzuki, Y., et al. (1998). Role of natural killer cells and TCR gamma delta T cells in acute autoimmune encephalomyelitis. European Journal of Immunology, 28(5), 1681–1688.
Mazzeo, A. T., Kunene, N. K., Gilman, C. B., Hamm, R. J., Hafez, N., & Bullock, M. R. (2006). Severe human traumatic brain injury, but not cyclosporin a treatment, depresses activated T lymphocytes early after injury. Journal of Neurotrauma, 23(6), 962–975.
Myer, D. J., Gurkoff, G. G., Lee, S. M., Hovda, D. A., & Sofroniew, M. V. (2006). Essential protective roles of reactive astrocytes in traumatic brain injury. Brain, 129(Pt 10), 2761–2772.
Natale, J. E., Ahmed, F., Cernak, I., Stoica, B., & Faden, A. I. (2003). Gene expression profile changes are commonly modulated across models and species after traumatic brain injury. Journal of Neurotrauma, 20(10), 907–927.
Perussia, B. (1996). The cytokine profile of resting and activated NK cells. Methods, 9(2), 370–378.
Raivich, G., Jones, L. L., Kloss, C. U., Werner, A., Neumann, H., & Kreutzberg, G. W. (1998). Immune surveillance in the injured nervous system: T-lymphocytes invade the axotomized mouse facial motor nucleus and aggregate around sites of neuronal degeneration. Journal of Neuroscience, 18(15), 5804–5816.
Rosenberger, C. M., Clark, A. E., Treuting, P. M., Johnson, C. D., & Aderem, A. (2008). ATF3 regulates MCMV infection in mice by modulating IFN-gamma expression in natural killer cells. Proceedings of the National Academy of Sciences of the United States of America, 105(7), 2544–2549.
Saura, J., Curatolo, L., Williams, C. E., Gatti, S., Benatti, L., Peeters, C., et al. (1999). Neuroprotective effects of Gly-Pro-Glu, the N-terminal tripeptide of IGF-1, in the hippocampus in vitro. Neuroreport, 10(1), 161–164.
Schousboe, A., & Waagepetersen, H. S. (2005). Role of astrocytes in glutamate homeostasis: Implications for excitotoxicity. Neurotoxicity Research, 8(3–4), 221–225.
Schroeter, M., & Jander, S. (2005). T-cell cytokines in injury-induced neural damage and repair. Neuromolecular Medicine, 7(3), 183–195.
Seijffers, R., Allchorne, A. J., & Woolf, C. J. (2006). The transcription factor ATF-3 promotes neurite outgrowth. Molecular and Cellular Neuroscience, 32(1–2), 143–154.
Stoica, B., Byrnes, K., & Faden, A. I. (2009). Multifunctional drug treatment in neurotrauma. Neurotherapeutics, 6(1), 14–27.
Stow, J. L., Low, P. C., Offenhauser, C., & Sangermani, D. (2009). Cytokine secretion in macrophages and other cells: Pathways and mediators. Immunobiology, 214(7), 601–612.
Study of NNZ-2566 in Patients with Traumatic Brain Injury (INTREPID2566). (2008). Neuren Pharmaceuticals. Accessed July 9, 2012 from http://www.clinicaltrials.gov/ct2/show/NCT00805818?term=NNZ&rank=4.
Tambuyzer, B. R., Ponsaerts, P., & Nouwen, E. J. (2009). Microglia: Gatekeepers of central nervous system immunology. Journal of Leukocyte Biology, 85(3), 352–370.
Thompson, M. R., Xu, D., & Williams, B. R. (2009). ATF3 transcription factor and its emerging roles in immunity and cancer. Journal of Molecular Medicine (Berlin, Germany), 87(11), 1053–1060.
Vianney-Rodrigues, P., Iancu, O. D., & Welsh, J. P. (2011). Gamma oscillations in the auditory cortex of awake rats. [Research Support, N.I.H., Extramural Research Support, Non-U.S. Gov’t]. European Journal of Neuroscience, 33(1), 119–129.
Wei, H. H., Lu, X. C., Shear, D. A., Waghray, A., Yao, C., Tortella, F. C., et al. (2009). NNZ-2566 treatment inhibits neuroinflammation and pro-inflammatory cytokine expression induced by experimental penetrating ballistic-like brain injury in rats. Journal of Neuroinflammation, 6, 19.
Whitmore, M. M., Iparraguirre, A., Kubelka, L., Weninger, W., Hai, T., & Williams, B. R. (2007). Negative regulation of TLR-signaling pathways by activating transcription factor-3. Journal of Immunology, 179(6), 3622–3630.
Williams, A. J., Hartings, J. A., Lu, X. C., Rolli, M. L., Dave, J. R., & Tortella, F. C. (2005). Characterization of a new rat model of penetrating ballistic brain injury. Journal of Neurotrauma, 22(2), 313–331.
Williams, A. J., Hartings, J. A., Lu, X. C., Rolli, M. L., & Tortella, F. C. (2006a). Penetrating ballistic-like brain injury in the rat: Differential time courses of hemorrhage, cell death, inflammation, and remote degeneration. Journal of Neurotrauma, 23(12), 1828–1846.
Williams, A. J., Ling, G. S., & Tortella, F. C. (2006b). Severity level and injury track determine outcome following a penetrating ballistic-like brain injury in the rat. Neuroscience Letters, 408(3), 183–188.
Williams, A. J., Wei, H. H., Dave, J. R., & Tortella, F. C. (2007). Acute and delayed neuroinflammatory response following experimental penetrating ballistic brain injury in the rat. Journal of Neuroinflammation, 4, 17.
Zhang, S. J., Buchthal, B., Lau, D., Hayer, S., Dick, O., Schwaninger, M., et al. (2011). A signaling cascade of nuclear calcium-CREB-ATF3 activated by synaptic NMDA receptors defines a gene repression module that protects against extrasynaptic NMDA receptor-induced neuronal cell death and ischemic brain damage. Journal of Neuroscience, 31(13), 4978–4990.
Acknowledgments
We would like to thank Matthew Bombard and Weihong Yang for their excellent assistance with surgical procedures. These studies were supported in part by a cooperative research and development agreement with Neuren Pharmaceuticals Ltd. (W81XWH-05-0074). This material has been reviewed by the Walter Reed Army Institute of Research. There is no objection to its presentation and/or publication. The opinions or assertions contained herein are the private views of the authors and are not to be construed as official, or reflecting true views of the Department of the Army or the Department of Defense.
Conflict of interest
No competing financial interests exist.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
12017_2013_8236_MOESM1_ESM.tif
Supplimental Figure 1. Hematoxylin and eosin staining of sham (upper panel) and PBBI (lower panel) coronal sections 24-h post-injury. (Bar = 2 mm). (TIFF 27772 kb)
12017_2013_8236_MOESM2_ESM.tif
Supplimental Figure 2. Confocal Microscopy of ATF3 and OX42 protein expression in Microglia 24-h post-injury. (Bar = 50 μm). (TIFF 4491 kb)
12017_2013_8236_MOESM3_ESM.tif
Supplimental Figure 3. Confocal Microscopy of ATF3 and CD3 protein expression in T Cells 24-h post-injury. (Bar = 50 μm). (TIFF 5362 kb)
12017_2013_8236_MOESM4_ESM.tif
Supplimental Figure 4. Confocal Microscopy of ATF3 and NKC protein expression in Natural Killer Cells 24-h post-injury. (Bar = 50 μm). (TIFF 3768 kb)
12017_2013_8236_MOESM5_ESM.tif
Supplimental Figure 5. Confocal Microscopy of ATF3 and GFAP protein expression in Astrocytes 24-h post-injury. (Bar = 50 μm). (TIFF 4294 kb)
12017_2013_8236_MOESM6_ESM.tif
Supplimental Figure 6. Confocal Microscopy of ATF3 and NeuN protein expression in Neurons 24-h post-injury. (Bar = 50 μm). (TIFF 1827 kb)
Rights and permissions
About this article
Cite this article
Cartagena, C.M., Phillips, K.L., Williams, G.L. et al. Mechanism of Action for NNZ-2566 Anti-inflammatory Effects Following PBBI Involves Upregulation of Immunomodulator ATF3. Neuromol Med 15, 504–514 (2013). https://doi.org/10.1007/s12017-013-8236-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-013-8236-z