Abstract
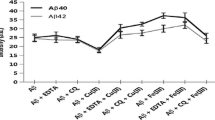
As reported by some authors, clioquinol (CQ), a 8-hydroxyquinoline derivative, has produced very encouraging results in the treatment of Alzheimer’s disease (AD). Its biological effects are most likely ascribed to complexation of specific metal ions, such as copper (II) and zinc (II), critically associated with β-amyloid (Aβ) aggregation/fibrillogenesis and degeneration processes in the brain. The present study was aimed at assessing the in vitro effects of CQ on the aggregation/fibrillogenesis properties of human Aβ either alone or complexed with Cu2+ and Zn2+. Surprisingly, our data indicated that CQ promoted rather than inhibited the formation of Aβ fibrillar aggregates when added metal ions were present. To understand whether the latter effects were related to the peptide amino acid sequence, we also investigated the aggregational profile of rat Aβ, which differs from the human homologous for three amino acidic substitutions. Such a sequence alteration drastically reduced the tendency of the peptide to undergo spontaneous aggregation/fibrillization. In the presence of CQ and metals, however, also rat Aβ showed a strong propensity to generate fibrillar aggregates. In agreement with the pro-aggregation effects observed in solution, studies with neuroblastoma cells demonstrated an impairment of cell functioning only in the presence of CQ + Aβ–metals. Based on the present findings, the literature data on the potential effectiveness of CQ-based chelation therapy in AD should be re-interpreted.








Similar content being viewed by others
References
Armstrong, R. A., Winsper, S. J., & Blair, J. A. (1995). Hypothesis: Is Alzheimer’s disease a metal/induced immune disorder. Neurodegeneration, 4, 107–111. doi:10.1006/neur.1995.0013.
Atwood, C. S., Moir, R. D., Huang, X., Scarpa, R. C., Bacarra, N. M., Romano, D. M., et al. (1998). Dramatic aggregation of Alzheimer A beta by Cu(II) is induced by conditions representing physiological acidosis. Journal of Biological Chemistry, 273, 12817–12826. doi:10.1074/jbc.273.21.12817.
Atwood, C. S., Scarpa, R. C., Huang, X., Moir, R. D., Jones, W. D., Fairlie, D. P., et al. (2000). Characterization of copper interactions with Alzheimer amyloid beta peptides: Identifications of an attomolar-affinity copper binding site on amyloid beta 1–42. Journal of Neurochemistry, 75, 1219–1233. doi:10.1046/j.1471-4159.2000.0751219.x.
Awasthi, A., Matsunaga, Y., & Yamada, T. (2005). Amyloid-beta causes apoptosis of neuronal cells via caspase cascade, which can be prevented by amyloid-beta-derived short peptides. Experimental Neurology, 196, 282–289. doi:10.1016/j.expneurol.2005.08.001.
Benvenisti-Zarom, L., Chen, J., & Regan, R. (2005). The oxidative neurotoxicity of clioquinol. Neuropharmacology, 49, 687–694.
Bocharova, O. V., Breydo, L., Salnikov, V. V., & Baskakov, I. (2005). Copper (II) inhibits in vitro conversion of prion protein into amyloid fibrils. Biochemistry, 44, 6776–6787. doi:10.1021/bi050251q.
Boyd-Kimball, D., Sultana, R., Mohmmad-Abdul, H., & Butterfield, A. (2004). Rodent Abeta(1–42) exhibits oxidative stress properties similar to those of human Abeta(1–42): Implications for proposed mechanisms of toxicity. Journal of Alzheimers Disease, 6, 515–525.
Bush, A. I. (2000). Metals and neuroscience. Current Opinion in Chemical Biology, 4, 184–191. doi:10.1016/S1367-5931(99)00073-3.
Bush, A. I. (2002). Metal complexing agents as therapies for Alzheimer’s disease. Neurobiology of Aging, 23, 1031–1038. doi:10.1016/S0197-4580(02)00120-3.
Bush, A. I. (2003). Copper, zinc and the metallobiology of Alzheimer’s disease. Alzheimer Disease and Associated Disorders, 17, 147–150. doi:10.1097/00002093-200307000-00005.
Bush, A. I., & Masters, C. L. (2001). Clioquinol’s return. Science, 292, 2251–2252. doi:10.1126/science.292.5525.2251.
Bush, A. I., & Tanzi, R. E. (2002). The galvanization of b-amyloid in Alzheimer’s disease. Proceedings of the National Academy of Sciences of the United States of America, 99, 7317–7319. doi:10.1073/pnas.122249699.
Bush, A. I., Masters, C. L., & Tanzi, R. E. (2003). Copper, Beta amyloid, and Alzheimer’s disease: Tapping a sensitive connection. Proceedings of the National Academy of Sciences of the United Sciences of America, 100, 11193–11194. doi:10.1073/pnas.2135061100.
Bush, A. I., Pettingell, W. H., Multhaup, G., dParadis, M., Vonsattel, J. P., Gusella, J. F., et al. (1994). Rapid induction of Alzheimer A-beta amyloid formation by zinc. Science, 265, 1464–1467. doi:10.1126/science.8073293.
Butterfield, D. A., & Boyd-Kimball, D. (2005). The critical role of methionine 35 in Alzheimer’s amyloid β-peptide(1–42)-induced oxidative stress and neurotoxicity. Biochimica et Biophysica Acta, 1073, 149–156.
Cherny, R. A., Atwood, C. S., Xilinas, M. E., Gray, D. N., Jones, W. D., Mclean, C. A., et al. (2001). Treatment with a copper-zinc chelator markedly and rapidly inhibits beta-amyloid accumulation in Alzheimer’s disease transgenic mice. Neuron, 30, 665–676. doi:10.1016/S0896-6273(01)00317-8.
Cherny, R. A., Legg, T. J., McLean, C. A., Fairlie, D. P., Huang, X., Atwood, C. S., et al. (1999). Aqueous dissolution of Alzheimer’s disease Aβ amyloids deposits by biometal depletion. Journal of Biological Chemistry, 274, 23223–23228. doi:10.1074/jbc.274.33.23223.
Cuajungco, M. P., Faget, K. Y., Huang, X., Tanzi, R. E., & Bush, A. I. (2000). Metal chelation as a potential therapy for Alzheimer’s disease. Annals of the New York Academy of Sciences of USA, 920, 292–304.
Dahlgren, K. N., Manelli, A. M., Stine, W. B., Jr., Baker, L. K., Krafft, G. A., & Ladu, M. J. (2002). Oligomeric and fibrillar species of amyloid-beta peptides differentially affect neuronal viability. Journal of Biological Chemistry, 277, 32046–32053. doi:10.1074/jbc.M201750200.
Daniel, K. G., Chen, D., Orlu, S., Cui, Q. C., Miller, F. R., & Dou, Q. P. (2005). Clioquinol and pyrrolidine dithiocarbamate complex with copper to form proteasome inhibitors and apoptosis inducers in human breast cancer cells. Breast Cancer Research, 7, 897–908. doi:10.1186/bcr1322.
Deshpande, A., Mina, E., Glabe, C., & Busciglio, J. (2006). Different conformations of amyloid β induce neurotoxicity by distinct mechanism in human cortical neurons. Journal of Neurosciences, 26, 6011–6018. doi:10.1523/JNEUROSCI.1189-06.2006.
Di Varia, M., Bazzicalupi, C., Oriolo, P., Messori, L., Bruni, B., & Zatta, P. (2004). Clioquinol, a drug for Alzheimer’s disease specifically interfering with brain metal metabolism: Structural characterization of its Zn(II) and copper(II) complexes. Inorganic Chemistry, 43, 3795–3797. doi:10.1021/ic0494051.
Ding, W. Q., Liu, B., Vaught, J. L., Palmiter, R. D., & Lind, S. E. (2006). Clioquinol and docosahexaenoic acid act synergistically to kill tumor cells. Molecular Cancer Therapies, 5, 1864–1872. doi:10.1158/1535-7163.MCT-06-0067.
Domingo, J. L. (2006). Aluminium and other metals in Alzheimer’s disease: A review of potential therapy with chelating agents. Journal of Alzheimers Diseases, 10, 331–341.
Drago, D., Bettella, M., Bolognin, S., Cendron, L., Scancar, J., Milacic, R., et al. (2007). Potential pathogenic role of b-Amyloid-aluminum complex in Alzheimer’s disease. International Journal of Biochemistry and Cell Biology, 40, 731–746. doi:10.1016/j.biocel.2007.10.014.
Drouet, B., Pincon-Raymond, M., Chambaz, J., & Pillot, T. (2000). Molecular basis of Alzheimer’s disease. Cellular and Molecular Life Sciences, 57, 705–715. doi:10.1007/s000180050035.
Ferrada, E., Aranciba, V., Loeb, B., Norambuena, E., Olea-azar, E., & Huidobro-toto, J. P. (2007). Stechiometry and conditional stability constants of Cu (II) or Zn (II) clioquinol complexes; implications for Alzheimer’s and Hungtington’s disease therapy. Neurotoxicology, 28, 445–449. doi:10.1016/j.neuro.2007.02.004.
Filiz, G., Caragounis, A., Bica, L., Du, T., Masters, C. L., Crouch, P. J., & White, A. R. (2008). Clioquinol inhibits peroxide-mediated toxicity through up-regulation of phosphoinositol-3-kinase and inhibition of p53 activity. International Journal of Biochemistry & Cell Biology, 40(5), 1030–1042.
Gaeta, A., & Hider, R. C. (2005). The crucial role of metal ions in neurodegeneration: The basis for a promising therapeutic strategy. British Journal of Pharmacology, 146, 1041–1059. doi:10.1038/sj.bjp.0706416.
Glabe, C. G. (2005). Amyloid accumulation and pathogensis of Alzheimer’s disease: Significance of monomeric, oligomeric and fibrillar Abeta. Sub-Cellular Biochemistry, 38, 167–177.
Hardy, J. A., & Higgins, G. A. (1992). Alzheimer’s disease: The amyloid cascade hypothesis. Science, 256, 184–185. doi:10.1126/science.1566067.
House, E., Collingwod, J., Khan, A., Korchazkina, O., Berthon, G., & Exley, C. (2004). Aluminium, iron, zinc and copper influence the in vitro formation of Aβ42 in a manner which may have consequences for metal chelation therapy in Alzheimer disease. Journal of Alzheimers Diseases, 6, 291–301.
Hsiao, K., Chapman, P., Nilsen, S., Eckman, C., Harigaya, Y., Younkin, S., et al. (1996). Correlative memory deficits, Aβ elevation, and amyloid plaques in transgenic mice. Science, 274, 99–102. doi:10.1126/science.274.5284.99.
Jenagaratnam, L., McShane, R. (2006). Clioquinol for the treatment of Alzheimer’s disease. Cochrane Database of Systematic Reviews, Issue 1 Art N: CD00538.
Kaur, D., Yantiri, F., Rajagopalan, S., Kumar, J., Mo, J. Q., Boonplueang, R., et al. (2003). Genetic or pharmacological iron chelation prevents MPTP-induced neurotoxicity in vivo: A novel therapy for Parkinson’s disease. Neuron, 37, 899–909. doi:10.1016/S0896-6273(03)00126-0.
Lesné, S., Koh, M. T., Kotilinek, L., Kayed, R., Glabe, C. G., Yang, A., et al. (2006). A specific amyloid-β protein assembly in the brain impairs memory. Nature, 440, 352–357. doi:10.1038/nature04533.
LeVine, H., 3rd. (1993). Thioflavine T interaction with syntethic Alzheimer’s disease beta-amyloid peptides: Detection of amyloid aggregation in solution. Protein Science, 2, 404–410.
Lovell, M. A., Robertson, J. D., Teesdale, W. J., Campbell, J. L., & Markersbery, W. R. (1998). Copper, iron and zinc in Alzheimer’s disease senile plaques. Journal of the Neurological Sciences, 158, 47–52. doi:10.1016/S0022-510X(98)00092-6.
Masuda, T., Hida, H., Kanda, Y., Aihara, N., Ohta, K., Yamada, K., et al. (2007). Oral administration of metal chelator ameliorates motor dysfunction after a small hemorrhage near the internal capsule in rat. Journal of Neuroscience Research, 1112, 1–15.
Maynard, C. J., Bush, A. I., Masters, C. L., Cappai, R., & Li, Q. X. (2005). Metals and amyloid-beta in Alzheimer’s disease. International Journal of Experimental Pathology, 86, 147–159. doi:10.1111/j.0959-9673.2005.00434.x.
McLaurin, J., & Chakrabartty, A. (1997). Characterization of the interactions of Alzheimer beta-amyloid peptides with phospholipid membranes. European Journal of Biochemistry, 245, 355–363. doi:10.1111/j.1432-1033.1997.t01-2-00355.x.
Miura, T., Suzuki, K., Kohata, N., & Takeuchi, H. (2000). Metal binding modes of Alzheimer’s amyloid beta-peptide in insoluble aggregates and soluble complexes. Biochemistry, 39, 7024–7031. doi:10.1021/bi0002479.
Nguyen, T., Hamby, A., & Massa, S. M. (2005). Clioquinol down-regulates mutant huntingtin expression in vitro and mitigates pathology in a Huntington’s disease mouse model. Proceedings of the National Academy of Sciences of the United States of America, 102, 11840–11845. doi:10.1073/pnas.0502177102.
Perry, G., Nunomura, A., Hirai, K., Zhu, X., Perez, M., Avila, J., et al. (2002). Is oxidative damage the fundamental pathogenic mechanism of Alzheimer’s disease and other neurodegenerative diseases? Free Radic. Biology and Medicine, 22, 1475–1479. doi:10.1016/S0891-5849(02)01113-9.
Raman, B., Ban, T., Yamaguchi, K., Sakai, M., Kawai, T., Naiki, H., et al. (2005). Metal ion-dependent effect of clioquinol on the fibril growth of an amyloid beta-peptide. Journal of Biological Chemistry, 280, 16157–16162. doi:10.1074/jbc.M500309200.
Ricchelli, F., Buggio, R., Drago, D., Salmona, M., Forloni, G., Negro, A., et al. (2006). Aggregation/fibrillogenesis of recombinant human prion protein and Gerstmann-Sträussler-Scheinker disease peptides in the presence of metal ions. Biochemistry, 45, 6724–6732. doi:10.1021/bi0601454.
Ricchelli, F., Drago, D., Filippi, B., Tognon, G., & Zatta, P. (2005). Aluminum-triggered structural modifications and aggregation of beta-amyloids. Cellular and Molecular Life Sciences, 62, 1724–1733. doi:10.1007/s00018-005-5141-0.
Ritchie, C. W., Bush, A. I., Mackinnon, A., Macfarlane, S., Mastwyk, M., MacGregor, L., et al. (2003). Metal-protein attenuation with clioquinol targeting Abeta amyloid deposition and toxicity in Alzheimer disease: A pilot phase 2 clinical trial. Archives of Neurology, 60, 1685–1691. doi:10.1001/archneur.60.12.1685.
Sampson, E., Jenagaratnam, L., & McShane, R. (2008). Metal protein attenuating compounds for the treatment of Alzheimer’s disease. Cochrane Database of Systematic Reviews, 23, CD005380.
Shäfer, S., Pajonk, F. G., Multhaup, G., & Bayer, T. A. (2007). Copper and clioquinol treatment in young APP transgenic and wild-type transgenic mice: Effects on the life expectancy, body weight, and metal-ion levels. Journal of Molecular Medicine, 85, 405–413. doi:10.1007/s00109-006-0140-7.
Shearman, M. S., Hawtin, S. R., & Tailor, V. J. (1995). The intracellular component of cellular 3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide (MTT) reduction is specifically inhibited by beta-amyloid peptides. Journal of Neurochemistry, 65, 218–227.
Talaga, P., & Queré, L. (2002). The plasma membrane: A target and hurdle for the development of anti-Abeta drugs? Current Drug Targets. CNS and Neurological Disorders, 6, 567–574. doi:10.2174/1568007023338897.
Todorich, B. M., & Connor, J. R. (2004). Redox metals in Alzheimer’s disease. Annals of the New York Academy of Sciences, 1012, 171–178. doi:10.1196/annals.1306.014.
Treiber, C., Simons, A., Strauss, M., Hafner, M., Cappai, R., Bayer, T. A., et al. (2004). Clioquinol mediates copper uptake and counteracts copper efflux activities of the amyloid precursor protein of Alzheimer’s disease. Journal of Biological Chemistry, 279, 51958–51964. doi:10.1074/jbc.M407410200.
Walsh, D. M., Klyubin, I., Shankart, G. M., Townsend, M., Fadeeva, J. V., Betts, V., et al. (2005). The role of cell-derived oligomers of Abeta in Alzheimer’s disease and avenues for therapeutic intervention. Biochemical Society Transactions, 33, 1087–1090. doi:10.1042/BST20051087.
Wang, S. S., Becerra-Artega, A., & Good, T. A. (2002). Development of a novel diffusion-based method to estimate the size of the aggregated Aβ species responsible for neurotoxicity. Biotechnology and Bioengineering, 80, 50–59. doi:10.1002/bit.10347.
White, A. R., Du, T., Laughton, K. M., Volitakis, I., Sharples, R. A., Xilinas, M. E., et al. (2006). Degradation of the Alzheimer disease amyloid beta-peptide by metal-dependent up-regulation of metalloprotease activity. Journal of Biological Chemistry, 281, 17670–17680. doi:10.1074/jbc.M602487200.
Yanker, B. A. (1996). Mechanisms of neuronal degeneration in Alzheimer’s disease. Neuron, 16, 921–932. doi:10.1016/S0896-6273(00)80115-4.
Yassin, M. S., Ekblom, J., Xilinias, M., Gottfries, C. G., & Orleand, L. (2000). Changes in uptake of vitamin B12 and trace metals in brains of mice treated with clioquinol. Journal of Neurological Sciences, 173, 40–44. doi:10.1016/S0022-510X(99)00297-X.
Yatin, S. M., Varadarajan, S., Link, C. D., & Butterfield, D. A. (1999). In vitro and in vivo oxidative stress associated with Alzheimer’s amyloid β-peptide (1–42). Neurobiology of Aging, 20, 325–330. doi:10.1016/S0197-4580(99)00056-1.
Zatta, P. (Ed.). (2003). Metal ions and neurodegenerative disorders (pp. 1–511). Singapore, London: World Scientific.
Acknowledgements
This work was supported by a grant from CNR/MIUR (FIRB no. RBNE03PX83). We gratefully acknowledge Prof. Luigi Messori and Dr. Chiara Gabbiani for helpful discussions.
Author information
Authors and Affiliations
Corresponding author
Additional information
An erratum to this article can be found at http://dx.doi.org/10.1007/s12017-008-8050-1
Rights and permissions
About this article
Cite this article
Bolognin, S., Zatta, P., Drago, D. et al. Mutual Stimulation of Beta-Amyloid Fibrillogenesis by Clioquinol and Divalent Metals. Neuromol Med 10, 322–332 (2008). https://doi.org/10.1007/s12017-008-8046-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-008-8046-x