Abstract
Objective
Assess the safety and efficacy of upcoming stem cell treatments and analyze their effects on the cognitive and behavioral impairments in patients diagnosed with autism.
Methods
We included controlled and noncontrolled, randomized and non-randomized trials evaluating stem cell therapy as a treatment in patients with autism spectrum disorder compared to placebo or without comparator. Data Sources: Scopus, Web of Science, MEDLINE and EMBASE. Risk of bias was assessed using Cochrane’s Risk of Bias tool and the NIH’s Quality Assessment Tool for Studies With No Control Group.
Results
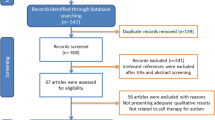
Eleven trials including 461 patients proved eligible. ABC scale meta-analysis showed a mean raw of -11.97 in the intervention groups (95 % CI -91.45 to 67.52, p < 0.01). CARS scale reported a mean raw of -9.08 (95 % CI -15.43 to -2.73, p < 0.01). VABS scale was reported by their domains: communication domain reported a mean raw of 2.69 (95 % CI 1.30 to 4.08, p = 0.92); daily living domain, 1.99 (95 % CI 0.83 to 3.15, p = 0.51); motor domain, 1.06 (95 % CI -0.37 to 2.48, p = 0.20); socialization domain, 3.09 (95 % CI 1.71 to 4.48, p = 0.61); adaptive behavior domain, 2.10 (95 % CI 1.04 to 3.16, p = 0.36). Furthermore, the most common side effects reported included fever, hyperactivity, vomit, headache, and aggressiveness; no serious adverse events were reported.
Conclusions
The body of evidence suggests that stem cell therapy significantly improves scales in patients with autism spectrum disorder, hence, future studies should help us have more confidence in the results. We found no serious adverse events related to the stem cell therapy.
Graphical Abstract








Similar content being viewed by others
Data Availability
Not applicable.
Code Availability
Not applicable.
Abbreviations
- ASDs:
-
Autism spectrum disorders
- MSCs:
-
mesenchymal stem cells
- BMMNC:
-
bone marrow mononuclear cell
- CBMNC:
-
cord blood mononuclear cell
- CRD42021225190:
-
PROSPERO
References
Theoharides, T. C., Kempuraj, D., & Redwood, L. (2009). Autism: an emerging “neuroimmune disorder” in search of therapy. In Expert Opinion on Pharmacotherapy (Vol. 10, Issue 13, pp. 2127–2143). https://doi.org/10.1517/14656560903107789
American Psychiatric Association. (2013). Diagnostic and Statistical Manual of Mental Disorders (DSM-5®). American Psychiatric Pub
Siniscalco, D., Kannan, S., Semprún-Hernández, N., Eshraghi, A. A., Brigida, A. L., & Antonucci, N. (2018). Stem cell therapy in autism: recent insights. In Stem Cells and Cloning: Advances and Applications (Vol. 11, pp. 55–67). https://doi.org/10.2147/sccaa.s155410
de Magistris, L., de Magistris, L., Picardi, A., Siniscalco, D., Riccio, M. P., Sapone, A. … Bravaccio, C. (2013). Antibodies against food antigens in patients with autistic spectrum disorders. In BioMed Research International (Vol. 2013, pp. 1–11). https://doi.org/10.1155/2013/729349
Sun, J. M., Dawson, G., Franz, L., Howard, J., McLaughlin, C., Kistler, B. … Kurtzberg, J. (2020). Infusion of human umbilical cord tissue mesenchymal stromal cells in children with autism spectrum disorder. In STEM CELLS Translational Medicine (Vol. 9, Issue 10, pp. 1137–1146). https://doi.org/10.1002/sctm.19-0434
Payakachat, N., Tilford, M., Kovacs, J., & Kuhlthau, K. (2012). Autism spectrum disorders: a review of measures for clinical, health services and cost–effectiveness applications. Expert Review of Pharmacoeconomics & Outcomes Research, 12(Issue 4), 485–503. https://doi.org/10.1586/erp.12.29
Ellison-Wright, Z., & Boardman, C. (2015). Diagnosis and management of ASD in children and adolescents. Progress in Neurology and Psychiatry, 19(6), 28–32
Nevels, R. M., Dehon, E. E., Alexander, K., & Gontkovsky, S. T. (2010). Psychopharmacology of aggression in children and adolescents with primary neuropsychiatric disorders: a review of current and potentially promising treatment options. Experimental and Clinical Psychopharmacology, 18(2), 184–201
Carpenter, K. L. H., Major, S., Tallman, C., Chen, L. W., Franz, L., Sun, J. … Dawson, G. (2019). White matter tract changes associated with clinical improvement in an open-label trial assessing autologous umbilical cord blood for treatment of young children with autism. In STEM CELLS Translational Medicine (Vol. 8, Issue 2, pp. 138–147). https://doi.org/10.1002/sctm.18-0251
Boncoraglio, G. B., Ranieri, M., Bersano, A., Parati, E. A., & Del Giovane, C. (2019). Stem cell transplantation for ischemic stroke. Cochrane Database of Systematic Reviews, 5, CD007231
Lv, Y. T., Zhang, Y., Liu, M., Qiuwaxi, J. N. T., Ashwood, P., Cho, S. C. … Hu, X. (2013). Transplantation of human cord blood mononuclear cells and umbilical cord-derived mesenchymal stem cells in autism. Journal of Translational Medicine, 11, 196
Nguyen, L. T., Nguyen, A. T., Vu, C. D., Ngo, D. V., & Bui, A. V. (2017). Outcomes of autologous bone marrow mononuclear cells for cerebral palsy: an open label uncontrolled clinical trial. BMC Pediatrics, 17(1), 104
Spejo, A. B., Carvalho, J. L., Goes, A. M., & Oliveira, A. L. R. (2013). Neuroprotective effects of mesenchymal stem cells on spinal motoneurons following ventral root axotomy: synapse stability and axonal regeneration. Neuroscience, 250, 715–732
Ziv, Y., Finkelstein, A., Geffen, Y., Kipnis, J., Smirnov, I., Shpilman, S. … Yoles, E. (2007). A novel immune-based therapy for stroke induces neuroprotection and supports neurogenesis. Stroke; a Journal of Cerebral Circulation, 38(2 Suppl), 774–782
Shen, L. H., Li, Y., Chen, J., Zacharek, A., Gao, Q., Kapke, A. … Chopp, M. (2007). Therapeutic benefit of bone marrow stromal cells administered 1 month after stroke. Journal of Cerebral Blood Flow and Metabolism: Official Journal of the International Society of Cerebral Blood Flow and Metabolism, 27(1), 6–13
McTighe, S. M., Neal, S. J., Lin, Q., Hughes, Z. A., & Smith, D. G. (2013). The BTBR mouse model of autism spectrum disorders has learning and attentional impairments and alterations in acetylcholine and kynurenic acid in prefrontal cortex. PLoS One, 8(4), e62189
Segal-Gavish, H., Karvat, G., Barak, N., Barzilay, R., Ganz, J., Edry, L., & Kimchi, T. (2016). Mesenchymal stem cell transplantation promotes neurogenesis and ameliorates autism related behaviors in BTBR Mice. Autism Research: Official Journal of the International Society for Autism Research, 9(1), 17–32
Perets, N., Segal-Gavish, H., Gothelf, Y., Barzilay, R., Barhum, Y., Abramov, N. … Offen, D. (2017). Long term beneficial effect of neurotrophic factors-secreting mesenchymal stem cells transplantation in the BTBR mouse model of autism. Behavioural Brain Research, 331, 254–260
Ha, S., Park, H., Mahmood, U., Ra, J. C., Suh, Y. H., & Chang, K. A. (2017). Human adipose-derived stem cells ameliorate repetitive behavior, social deficit and anxiety in a VPA-induced autism mouse model. In Behavioural Brain Research (Vol. 317, pp. 479–484). https://doi.org/10.1016/j.bbr.2016.10.004
Price, J. (2020). Cell therapy approaches to autism: a review of clinical trial data. Molecular Autism, 11(1), 37
Sharma, A., Gokulchandran, N., Sane, H., Nagrajan, A., Paranjape, A., Kulkarni, P. … Badhe, P. (2013). Autologous bone marrow mononuclear cell therapy for autism: an open label proof of concept study. Stem Cells International, 2013, 623875
Bansal, H., Verma, P., Agrawal, A., Leon, J., Sundell, I. B., & Koka, P. S. (2016). A short study report on bone marrow aspirate concentrate cell therapy in ten South Asian Indian patients with autism. Journal of Stem Cells, 11(1), 25–36
Dawson, G., Sun, J. M., Davlantis, K. S., Murias, M., Franz, L., Troy, J., & Kurtzberg, J. (2017). Autologous cord blood infusions are safe and feasible in young children with autism spectrum disorder: results of a single-center phase i open-label Trial. Stem Cells Translational Medicine, 6(5), 1332–1339
Higgins, J. P. T., & Thomas, J. (2019). Cochrane Handbook for Systematic Reviews of Interventions. Wiley
Balduzzi, S., Rücker, G., & Schwarzer, G. (2019). How to perform a meta-analysis with R: A practical tutorial. Evidence-Based Mental Health, 22(4), 153–160. https://doi.org/10.1136/ebmental-2019-300117
Riordan, N. H., Hincapié, M. L., Morales, I., Fernández, G., Allen, N., Leu, C., & Novarro, N. (2019). Allogeneic human umbilical cord mesenchymal stem cells for the treatment of autism spectrum disorder in children: safety profile and effect on cytokine levels. Stem Cells Translational Medicine, 8(10), 1008–1016
Chez, M., Lepage, C., Parise, C., Dang-Chu, A., Hankins, A., & Carroll, M. (2018). Safety and observations from a placebo-controlled, crossover study to assess use of autologous umbilical cord blood stem cells to improve symptoms in children with autism. Stem Cells Translational Medicine, 7(4), 333–341
Dawson, G., Sun, J. M., Baker, J., Carpenter, K., Compton, S., Deaver, M., Franz,L., Heilbron, N., Herold, B., Horrigan, J., Howard, J., Kosinski, A., Major, S., Murias,M., Page, K., Prasad, V. K., Sabatos-DeVito, M., Sanfilippo, F., Sikich, L., … Kurtzberg,J. (2020). A Phase II Randomized Clinical Trial of the Safety and Efficacy of Intravenous Umbilical Cord Blood Infusion for Treatment of Children with Autism Spectrum Disorder. The Journal of Pediatrics, 222, 164–173.e5
Murias, M., Major, S., Compton, S., Buttinger, J., Sun, J. M., Kurtzberg, J., & Dawson, G. (2018). Electrophysiological biomarkers predict clinical improvement in an open-label trial assessing efficacy of autologous umbilical cord blood for treatment of autism. In STEM CELLS Translational Medicine (Vol. 7, Issue 11, pp. 783–791). https://doi.org/10.1002/sctm.18-0090
Nguyen Thanh, L., Nguyen, H. P., Ngo, M. D., Bui, V. A., Dam, P. T. M., Bui, H. T. P. … Heke, M. (2021). Outcomes of bone marrow mononuclear cell transplantation combined with interventional education for autism spectrum disorder. Stem Cells Translational Medicine, 10(1), 14–26
Bradstreet, J. J., Sych, N., Antonucci, N., Klunnik, M., Ivankova, O., Matyashchuk, I. … Siniscalco, D. (2014). Efficacy of fetal stem cell transplantation in autism spectrum disorders: an open-labeled pilot study. Cell Transplantation, 23(Suppl 1), S105–S112
Acknowledgements
We want to thank Marahí Ortiz-Castillo for the design and execution of the graphical abstract.
Author information
Authors and Affiliations
Contributions
All authors were involved, in the conceiving of the research idea and elaboration of the research protocol. LVM supervised the project fulfillment. EGL designed the search strategy and performed the literature search. GGM, MSF, AJBG, AGM screened studies for eligibility. GGM, MSF, AJBG, AGM assessed the risk of bias. GGM, MSF, AJBG, AGM performed data extraction. MSF performed the statistical analysis. MOC, DARS, MSF, AJBG, AGM contributed in the design, interpretation of data and drafting of the work. All authors were involved in the elaboration and approval of the final version of the manuscript. LVM is guarantor. The corresponding author attests that all listed authors meet authorship criteria and that no others meeting the criteria have been omitted.
Corresponding author
Ethics declarations
Conflicts of Interest/Competing Interest
The authors have no conflicts of interest to declare that are relevant to the content to this article.
Ethics Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 14.6 KB)
Rights and permissions
About this article
Cite this article
Villarreal-Martínez, L., González-Martínez, G., Sáenz-Flores, M. et al. Stem Cell Therapy in the Treatment of Patients With Autism Spectrum Disorder: a Systematic Review and Meta-analysis. Stem Cell Rev and Rep 18, 155–164 (2022). https://doi.org/10.1007/s12015-021-10257-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12015-021-10257-0