Abstract
Background & Aim
Search for an effective therapy for patients with hepatitis B virus related acute-on-chronic liver failure (HBV-ACLF) remains an important issue. This study investigated the efficacy of umbilical cord-derived mesenchymal stem cell (UC-MSC) transplantation in patients with HBV-ACLF.
Methods
45 consecutive entecavir-treated HBV-ACLF patients were prospectively studied. Among these patients, 11 received both plasma exchange (PE) and a single transplantation of UC-MSCs (group A), while 34 received only PE (group B). The primary endpoint was survival at 24 months.
Results
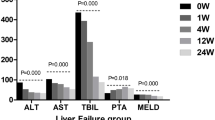
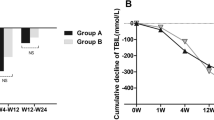
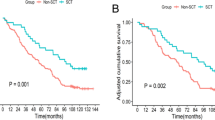
Compared with group B, levels of albumin, alanine aminotransferase, aspartate aminotransferase, total bilirubin, direct bilirubin, prothrombin time (PT), international normalized ratio (INR) and model for end-stage liver disease score in group A improved significantly at 4 weeks after transplantation (p < 0.05). Levels of albumin, PT and INR in group A were also markedly improved at 24 months (p < 0.05). Group A had significantly higher cumulative survival rate at 24 months (54.5 % v.s. 26.5 %, p = 0.015 by log rank test). Between the two groups, levels of creatinine, White blood cell, hemoglobin and platelet were similar. HBeAg loss and hepatocellular carcinoma incidence were similar at 24 months. Group assignment (relative risk: 2.926, 95%confidence interval: 1.043–8.203, p = 0.041) was an independent predictor for survival at 24 months. Success rate of UC-MSC transplantation was 100 % in group A. No severe adverse event was observed in any patient.
Conclusion
UC-MSC transplantation is safe and effective for HBV-ACLF patients treated with PE and entecavir. It further improves the hepatic function and survival.

Similar content being viewed by others
Abbreviations
- HBV-ACLF:
-
Hepatitis B virus related acute-on-chronic liver failure
- LT:
-
Liver transplantation
- ALS:
-
Artificial liver support systems
- PE:
-
Plasma exchange
- ETV:
-
Entecavir
- MSCs:
-
Mesenchymal stem cells
- BM-MSCs:
-
Bone marrow-derived MSCs
- UC-MSCs:
-
Umbilical cord-derived MSCs
- INR:
-
International normalized ratio
- PTA:
-
Prothrombin activity
- PLT:
-
Platelet
- HBsAg:
-
Hepatitis B surface antigen
- NAs:
-
Nucleos(t)ide analogs
- HCV:
-
Hepatitis C virus
- HDV:
-
Hepatitis D virus
- HIV:
-
Human immunodeficiency virus
- HCC:
-
Hepatocellular carcinoma
- AFP:
-
Alpha-fetoprotein
- CT:
-
Computed tomography
- USG:
-
Ultrasonography
- PT:
-
Prothrombin time
- CBC:
-
Complete blood count
- MELD:
-
Model for end-stage liver disease
- SD:
-
Standard deviation
- ALT:
-
Alanine amnotransferase
- AST:
-
Aspartate aminotransferase
- TBIL:
-
Total bilirubin
- DBIL:
-
Direct bilirubin
- Cr:
-
Creatinine
- WBC:
-
White blood cell
- HGB:
-
Hemoglobulin
References
Liang, X. F., Chen, Y. S., Wang, X. J., et al. (2005). A study on the sero-epidemiology of hepatitis B in Chinese population aged over 3-years old [in Chinese. Zhonghua Liu Xing Bing Xue Za Zhi, 26, 655–658.
Sarin, S. K., Kedarisetty, C. K., Abbas, Z., et al. (2014). APASL ACLF working party. Acute-on-chronic liver failure: consensus recommendations of the Asian Pacific association for the study of the liver (APASL) 2014. Hepatology International, 8(4), 453–471.
Murray, K. F., & Carithers, R. L. A. A. S. L. D. (2005). Practice guidelines: evaluation of the patient for liver transplantation. Hepatology, 41, 1407–1432.
Maiwall, R., Maras, J. S., Nayak, S. L., et al. (2014). Liver dialysis in acute-on-chronic liver failure: current and future perspectives. Hepatology International, 8(Suppl 2), 505–513.
Larsen, F. S., Schmidt, L. E., Bernsmeier, C., et al. (2015). High-volume plasma exchange in patients with acute liver failure: an open randomised controlled trial. Journal of Hepatology, 64(1), 69–78.
Yue-Meng, W., Yang, L. H., Yang, J. H., et al. (2016). The effect of plasma exchange on entecavir-treated chronic hepatitis B patients with hepatic de-compensation and acute-onchronic liver failure. Hepatology International, 10(3), 462–469.
Lee, K. D., Kuo, T. K., Whang-Peng, J., et al. (2004). In vitro hepatic differentiation of human mesenchymal stem cells. Hepatology, 40, 1275–1284.
Terai, S., Ishikawa, T., Omori, K., et al. (2006). Improved liver function in patients with liver cirrhosis after autologous bone marrow cell infusion therapy. Stem Cells, 24, 2292–2298.
Kharaziha, P., Hellström, P. M., Noorinayer, B., et al. (2009). Improvement of liver function in liver cirrhosis patients after autologous mesenchymal stem cell injection: a phase I-II clinical trial. European Journal of Gastroenterology & Hepatology, 21, 1199–1205.
Zhang, Z., Lin, H., Shi, M., et al. (2012). Human umbilical cord mesenchymal stem cells improve liver function and ascites in decompensated liver cirrhosis patients. Journal of Gastroenterology and Hepatology, 27(suppl 2), 112–120.
Shi, M., Zhang, Z., Xu, R., et al. (2012). Human mesenchymal stem cell transfusion is safe and improves liver function in acute-on-chronic liver failure patients. Stem Cells Translational Medicine, 1(10), 725–731.
Liang, P., Xie, D.-y., Lin, B.-L., et al. (2011). Autologous bone marrow mesenchymal stem cell transplantation in liver failure patients caused by hepatitis B: short-term and long-term outcomes. Hepatology, 54(3), 820–828.
Sarin, S. K., Kumar, A., Almeida, J. A., et al. (2009). Acute-onchronic liver failure: consensus recommendations of the Asian Pacific association for the study of the liver (APASL. Hepatology International, 3, 269–282.
Yue-Meng, W., Li, Y. H., Wu, H. M., et al. (2016) Telbivudine versus lamivudine and entecavir fortreatment-naïve decompensated hepatitis B virus-relatedcirrhosis. Clinical and Experimental Medicine 19 Apr 2016
LL, L., Liu, Y. J., Yang, S. G., et al. (2006). Isolation and characterization of human umbilical cord mesenchymal stem cells with hematopoiesis-supportive function and other potentials. Haematologica, 91, 1017–1026.
Kamath, P. S., & Kim, W. R. (2007). The model for end-stage liver disease (MELD). Hepatology, 45(3), 797–805.
Flink, H. J., Sprengers, D., Hansen, B. E., et al. (2005). HBV 99-01 study group. Flares in chronic hepatitis B patients induced by the host or the virus? Relation to treatment response during peg-interferon alpha-2b therapy. Gut, 54, 1604–1609.
Moreau, R., Jalan, R., Gines, P., et al. (2013). Acute-on-chronic liver failure is a distinct syndrome that develops in patients with acute decompensation of cirrhosis. Gastroenterology, 144(7), 1426–1437.
Xie, F., Yan, L., Lu, J., et al. (2013). Effects of nucleoside analogue on patients with chronic hepatitis B-associated liver failure: meta-analysis. PloS One, 8(1), e54773.
Chang, T. T., Gish, R. G., de Man, R., et al. (2006). A comparison of entecavir and lamivudine for HBeAg-positive chronic hepatitis B. The New England Journal of Medicine, 354, 1001–1010.
Lai, C. L., Shouval, D., Lok, A. S., et al. (2006). Entecavir versus lamivudine for patients with HBeAg negative chronic hepatitis B. The New England Journal of Medicine, 354, 1011–1020.
Tenney, D. J., Rose, R. E., Baldick, C. J., et al. (2009). Long-term monitoring shows hepatitis B virus resistance to entecavir in nucleoside-naive patients is rare through 5 years of therapy. Hepatology, 49, 1503–1514.
Lange, C. M., Bojunga, J., Hofmann, W. P., et al. (2009). Severe lactic acidosis during treatment of chronic hepatitis B with entecavir in patients with impaired liver function. Hepatology, 50, 2001–2006.
Liu, X. Y., Peng, F., Pan, Y. J., et al. (2015). Advanced therapeutic strategies for HBV-related acute-on-chronic liver failure. Hepatobiliary & Pancreatic Diseases International, 14(4), 354–360.
Zheng, Z., Li, X., Li, Z., et al. (2013). Artificial and bioartificial liver support systems for acute and acute-on-chronic hepatic failure: a meta-analysis and meta-regression. Experimental and Therapeutic Medicine, 6, 929–936.
Nakae, H., Yonekawa, T., Narita, K., et al. (2001). Are proinflammatory cytokine concentrations reduced by plasma exchange in patients with severe acute hepatic failure? Research Communications in Molecular Pathology and Pharmacology, 109, 65–72.
Nakamura, T., Ushiyama, C., Suzuki, S., et al. (2000). Effect of plasma exchange on serum tissue inhibitor of metalloproteinase 1 and cytokine concentrations in patients with fulminant hepatitis. Blood Purification, 18, 50–54.
Garg, V., Garg, H., Khan, A., et al. (2012). Granulocyte-colony stimulating factor mobilizes CD34+ cells and improves survival of patients with acute-on-chronic liver failure. Gastroenterology, 142, 505–512.
Xue, H. L., Zeng, W. Z., XL, W., et al. (2015). Clinical therapeutic effects of human umbilical cord-derived mesenchymal stem cells transplantation in the treatment of end-stage liver disease. Transplantation Proceedings, 47(2), 412–418.
Zhu, X., He, B., Zhou, X., et al. (2013). Effects of transplanted bone-marrow-derived mesenchymal stem cells in animal models of acute hepatitis. Cell and Tissue Research, 351, 477–486.
Shi, M., Liu, Z. W., & Wang, F. S. (2011). Immunomodulatory properties and therapeutic application of mesenchymal stem cells. Clinical and Experimental Immunology, 164, 1–8.
Aurich, I., Mueller, L. P., Aurich, H., et al. (2007). Functional integration of hepatocytes derived from human mesenchymal stem cells into mouse livers. Gut, 56, 405–415.
di Bonzo, L. V., Ferrero, I., Cravanzola, C., et al. (2008). Human mesenchymal stem cells as a two-edged sword in hepatic regenerative medicine: engraftment and hepatocyte differentiation versus profibrogenic potential. Gut, 57, 223–231.
Baertschiger RM, Serre-Beinier V, Morel P, et al. Fibrogenic potential of human multipotent mesenchymal stromal cells in injured liver. PloS One 2009; 4:e6657.
Aurich, H., Sgodda, M., Kaltwasser, P., et al. (2009). Hepatocyte differentiation of mesenchymal stem cells from human adipose tissue in vitro promotes hepatic integration in vivo. Gut, 58, 570–581.
Acknowledgments
We kindly thank Prof. Jin-Hui Yang for his theoretical support and for permitting us to carry out this study. We thank Mr. Hong-Wei Wang from Shenzhen Baike Cell Engineering Research Institute for providing human UC-MSCs and instructing the clinical application of UC-MSCs. We thank interventional radiologists Prof. Ying-Chun Li and Song-Wei Li for their technical support in performing transplantation of UC-MSCs via the hepatic artery.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflicts of interest.
Funding
None.
Rights and permissions
About this article
Cite this article
Li, YH., Xu, Y., Wu, HM. et al. Umbilical Cord-Derived Mesenchymal Stem Cell Transplantation in Hepatitis B Virus Related Acute-on-Chronic Liver Failure Treated with Plasma Exchange and Entecavir: a 24-Month Prospective Study. Stem Cell Rev and Rep 12, 645–653 (2016). https://doi.org/10.1007/s12015-016-9683-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12015-016-9683-3