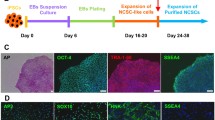
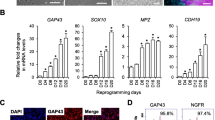
Abstract
Tissue engineering of Schwann cells (SCs) can serve a number of purposes, such as in vitro SC-related disease modeling, treatment of peripheral nerve diseases or peripheral nerve injury, and, potentially, treatment of CNS diseases. SCs can be generated from autologous stem cells in vitro by recapitulating the various stages of in vivo neural crest formation and SC differentiation. In this review, we survey the cellular and molecular mechanisms underlying these in vivo processes. We then focus on the current in vitro strategies for generating SCs from two sources of pluripotent stem cells, namely embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs). Different methods for SC engineering from ESCs and iPSCs are reviewed and suggestions are proposed for optimizing the existing protocols. Potential safety issues regarding the clinical application of iPSC-derived SCs are discussed as well. Lastly, we will address future aspects of SC engineering.




Similar content being viewed by others
References
Jessen, K. R., & Mirsky, R. (2005). The origin and development of glial cells in peripheral nerves. Nature Reviews Neuroscience, 6, 671–682.
Kamholz, J., Menichella, D., Jani, A., et al. (2000). Charcot-Marie-Tooth disease type 1: molecular pathogenesis to gene therapy. Brain, 123(Pt 2), 222–233.
Stadtfeld, M., & Hochedlinger, K. (2010). Induced pluripotency: history, mechanisms, and applications. Genes and Development, 24, 2239–2263.
Eser, F., Bodur, H., & Atan, Ç. (2009). Etiological factors of traumatic peripheral nerve injuries. Neurology India, 57(4), 434–437.
Novak, C. B., Anastakis, D. J., Beaton, D. E., Mackinnon, S. E., & Katz, J. (2011). Biomedical and psychosocial factors associated with disability after peripheral nerve injury. Journal of Bone and Joint Surgery, 93(10), 929–936.
Fawcett, J., & Keynes, R. J. (1990). Peripheral nerve regeneration. Annual Review of Neuroscience, 13, 43–60.
Terenghi, G. (1999). Peripheral nerve regeneration and neurotrophic factors. Journal of Anatomy, 194(Pt 1), 1–14.
Frostick, S. P., Yin, Q., & Kemp, G. J. (1998). Schwann cells, neurotrophic factors, and peripheral nerve regeneration. Microsurgery, 18(7), 397–405.
Jessen, K., & Mirsky, R. (2002). Signals that determine Schwann cell identity. Journal of Anatomy, 200(4), 367–376.
Hadlock, T., Elisseeff, J., Langer, R., Vacanti, J., & Cheney, M. (1998). A tissue-engineered conduit for peripheral nerve repair. Archives of Otolaryngology Head and Neck Surgery, 124(10), 1081–1086.
Fansa, H., Keilhoff, G., Wolf, G., & Schneider, W. (2001). Tissue engineering of peripheral nerves: a comparison of venous and acellular muscle grafts with cultured Schwann cells. Plastic and Reconstructive Surgery, 107(2), 485–494. discussion 495–6.
Frerichs, O., Fansa, H., Schicht, C., Wolf, G., Schneider, W., & Keilhoff, G. (2002). Reconstruction of peripheral nerves using acellular nerve grafts with implanted cultured Schwann cells. Microsurgery, 22(7), 311–315.
Rodríguez, F. J., Verdú, E., Ceballos, D., & Navarro, X. (2000). Nerve guides seeded with autologous Schwann cells improve nerve regeneration. Experimental Neurology, 161(2), 571–584.
Schlosshauer, B., Müller, E., Schröder, B., Planck, H., & Müller, H. (2003). Rat Schwann cells in bioresorbable nerve guides to promote and accelerate axonal regeneration. Brain Research, 963(1), 321–326.
Evans, G. R., Brandt, K., Katz, S., et al. (2002). Bioactive poly (L-lactic acid) conduits seeded with Schwann cells for peripheral nerve regeneration. Biomaterials, 23(3), 841–848.
Fansa, H., & Keilhoff, G. (2004). Comparison of different biogenic matrices seeded with cultured Schwann cells for bridging peripheral nerve defects. Neurological Research, 26(2), 167–173.
Keirstead, H. S., Nistor, G., Bernal, G., et al. (2005). Human embryonic stem cell-derived oligodendrocyte progenitor cell transplants remyelinate and restore locomotion after spinal cord injury. Journal of Neuroscience, 25(19), 4694–4705.
Totoiu, M. O., Nistor, G. I., Lane, T. E., & Keirstead, H. S. (2004). Remyelination, axonal sparing, and locomotor recovery following transplantation of glial-committed progenitor cells into the MHV model of multiple sclerosis. Experimental Neurology, 187(2), 254–265.
Blakemore, W. (1977). Remyelination of CNS axons by Schwann cells transplanted from the sciatic nerve. Nature, 266, 68–69.
Duncan, I., Aguayo, A., Bunge, R., & Wood, P. (1981). Transplantation of rat Schwann cells grown in tissue culture into the mouse spinal cord. Journal of Neurological Sciences, 49(2), 241–252.
Itoyama, Y., Webster, H. D., Richardson, E. P., & Trapp, B. D. (1983). Schwann cell remyelination of demyelinated axons in spinal cord multiple sclerosis lesions. Annals of Neurology, 14(3), 339–346.
Baron-Van Evercooren, A., Gansmuller, A., Duhamel, E., Pascal, F., & Gumpel, M. (1992). Repair of a myelin lesion by Schwann cells transplanted in the adult mouse spinal cord. Journal of Neuroimmunology, 40(2), 235–242.
Blakemore, W., Olby, N., & Franklin, R. (1995). The use of transplanted glial cells to reconstruct glial environments in the CNS. Brain Pathology, 5(4), 443–450.
Honmou, O., Felts, P. A., Waxman, S. G., & Kocsis, J. D. (1996). Restoration of normal conduction properties in demyelinated spinal cord axons in the adult rat by transplantation of exogenous Schwann cells. Journal of Neuroscience, 16(10), 3199–3208.
Iwashita, Y., Fawcett, J. W., Crang, A., Franklin, R. J., & Blakemore, W. F. (2000). Schwann cells transplanted into normal and X-irradiated adult white matter do not migrate extensively and show poor long-term survival. Experimental Neurology, 164(2), 292–302.
Dezawa, M., Takahashi, I., Esaki, M., Takano, M., & Sawada, H. (2001). Sciatic nerve regeneration in rats induced by transplantation of in vitro differentiated bone-marrow stromal cells. European Journal of Neuroscience, 14(11), 1771–1776.
Shimizu, S., Kitada, M., Ishikawa, H., Itokazu, Y., Wakao, S., & Dezawa, M. (2007). Peripheral nerve regeneration by the in vitro differentiated-human bone marrow stromal cells with Schwann cell property. Biochemical and Biophysical Research Communications, 359(4), 915–920.
Wakao, S., Hayashi, T., Kitada, M., et al. (2010). Long-term observation of auto-cell transplantation in non-human primate reveals safety and efficiency of bone marrow stromal cell-derived Schwann cells in peripheral nerve regeneration. Experimental Neurology, 223(2), 537–547.
Mimura, T., Dezawa, M., Kanno, H., Sawada, H., & Yamamoto, I. (2004). Peripheral nerve regeneration by transplantation of bone marrow stromal cell-derived Schwann cells in adult rats. Journal of Neurosurgery, 101(5), 806–812.
Tohill, M., Mantovani, C., Wiberg, M., & Terenghi, G. (2004). Rat bone marrow mesenchymal stem cells express glial markers and stimulate nerve regeneration. Neuroscience Letters, 362(3), 200–203.
Tohill, M. P., Mann, D. J., Mantovani, C. M., Wiberg, M., & Terenghi, G. (2004). Green fluorescent protein is a stable morphological marker for Schwann cell transplants in bioengineered nerve conduits. Tissue Engineering, 10(9–10), 1359–1367.
Caddick, J., Kingham, P. J., Gardiner, N. J., Wiberg, M., & Terenghi, G. (2006). Phenotypic and functional characteristics of mesenchymal stem cells differentiated along a Schwann cell lineage. Glia, 54(8), 840–849.
Brohlin, M., Mahay, D., Novikov, L. N., et al. (2009). Characterisation of human mesenchymal stem cells following differentiation into Schwann cell-like cells. Neuroscience Research, 64(1), 41–49.
Kingham, P. J., Kalbermatten, D. F., Mahay, D., Armstrong, S. J., Wiberg, M., & Terenghi, G. (2007). Adipose-derived stem cells differentiate into a Schwann cell phenotype and promote neurite outgrowth in vitro. Experimental Neurology, 207(2), 267–274.
Xu, Y., Liu, L., Li, Y., et al. (2008). Myelin-forming ability of Schwann cell-like cells induced from rat adipose-derived stem cells in vitro. Brain Research, 1239, 49–55.
Jiang, L., Zhu, J. K., Liu, X. L., Xiang, P., Hu, J., & Yu, W. H. (2008). Differentiation of rat adipose tissue-derived stem cells into Schwann-like cells in vitro. Neuroreport, 19(10), 1015–1019.
Radtke, C., Schmitz, B., Spies, M., Kocsis, J., & Vogt, P. (2009). Peripheral glial cell differentiation from neurospheres derived from adipose mesenchymal stem cells. International Journal of Developmental Neuroscience, 27(8), 817–823.
Chi, G. F., Kim, M., Kim, D., Jiang, M. H., & Son, Y. (2010). Schwann cells differentiated from spheroid-forming cells of rat subcutaneous fat tissue myelinate axons in the spinal cord injury. Experimental Neurology, 222(2), 304–317.
Fernandes, K. J., McKenzie, I. A., Mill, P., et al. (2004). A dermal niche for multipotent adult skin-derived precursor cells. Nature Cell Biology, 6(11), 1082–1093.
Toma, J. G., McKenzie, I. A., Bagli, D., & Miller, F. D. (2005). Isolation and characterization of multipotent skin-derived precursors from human skin. Stem Cells, 23(6), 727–737.
McKenzie, I. A., Biernaskie, J., Toma, J. G., Midha, R., & Miller, F. D. (2006). Skin-derived precursors generate myelinating Schwann cells for the injured and dysmyelinated nervous system. Journal of Neuroscience, 26(24), 6651–6660.
Sieber-Blum, M., Grim, M., Hu, Y., & Szeder, V. (2004). Pluripotent neural crest stem cells in the adult hair follicle. Developmental Dynamics, 231(2), 258–269.
Amoh, Y., Li, L., Katsuoka, K., Penman, S., & Hoffman, R. M. (2005). Multipotent nestin-positive, keratin-negative hair-follicle bulge stem cells can form neurons. Proceedings of the National academy of Sciences of the United States of America, 102(15), 5530–5534.
Krause, M. P., Dworski, S., Feinberg, K., et al. (2014). Direct genesis of functional rodent and human Schwann cells from skin mesenchymal precursors. Stem Cell Reports, 3(1), 85–100.
Amoh, Y., Li, L., Katsuoka, K., & Hoffman, R. M. (2009). Multipotent nestin-expressing hair follicle stem cells. Journal of Dermatology, 36(1), 1–9.
Biernaskie, J. A., McKenzie, I. A., Toma, J. G., & Miller, F. D. (2007). Isolation of skin-derived precursors (SKPs) and differentiation and enrichment of their Schwann cell progeny. Nature Protocols, 1(6), 2803–2812.
Biernaskie, J., Sparling, J. S., Liu, J., et al. (2007). Skin-derived precursors generate myelinating Schwann cells that promote remyelination and functional recovery after contusion spinal cord injury. Journal of Neuroscience, 27(36), 9545–9559.
Heine, W., Conant, K., Griffin, J. W., & Höke, A. (2004). Transplanted neural stem cells promote axonal regeneration through chronically denervated peripheral nerves. Experimental Neurology, 189(2), 231–240.
Murakami, T., Fujimoto, Y., Yasunaga, Y., et al. (2003). Transplanted neuronal progenitor cells in a peripheral nerve gap promote nerve repair. Brain Research, 974(1), 17–24.
Evans, M. J., & Kaufman, M. H. (1981). Establishment in culture of pluripotential cells from mouse embryos. Nature, 292(5819), 154–156.
Martin, G. R. (1981). Isolation of a pluripotent cell line from early mouse embryos cultured in medium conditioned by teratocarcinoma stem cells. Proceedings of the National Academy of Sciences, 78(12), 7634–7638.
Itskovitz-Eldor, J., Schuldiner, M., Karsenti, D., et al. (2000). Differentiation of human embryonic stem cells into embryoid bodies comprising the three embryonic germ layers. Molecular Medicine, 6(2), 88–95.
Biswas, A., & Hutchins, R. (2007). Embryonic stem cells. Stem Cells and Development, 16(2), 213–222.
Thomson, J. A., Itskovitz-Eldor, J., Shapiro, S. S., et al. (1998). Embryonic stem cell lines derived from human blastocysts. Science, 282(5391), 1145–1147.
Takahashi, K., & Yamanaka, S. (2006). Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell, 126(4), 663–676.
Takahashi, K., Tanabe, K., Ohnuki, M., et al. (2007). Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell, 131(5), 861–872.
Maherali, N., Sridharan, R., Xie, W., et al. (2007). Directly reprogrammed fibroblasts show global epigenetic remodeling and widespread tissue contribution. Cell Stem Cell, 1(1), 55–70.
Okita, K., Ichisaka, T., & Yamanaka, S. (2007). Generation of germline-competent induced pluripotent stem cells. Nature, 448(7151), 313–317.
Wernig, M., Zhao, J., Pruszak, J., et al. (2008). Neurons derived from reprogrammed fibroblasts functionally integrate into the fetal brain and improve symptoms of rats with Parkinson’s disease. Proceedings of the National Academy of Sciences, 105(15), 5856–5861.
Hanna, J., Wernig, M., Markoulaki, S., et al. (2007). Treatment of sickle cell anemia mouse model with iPS cells generated from autologous skin. Science, 318(5858), 1920–1923.
Du, Z., & Zhang, S. (2004). Neural differentiation from embryonic stem cells: which way? Stem Cells and Development, 13(4), 372–381.
Villanueva, S., Glavic, A., Ruiz, P., & Mayor, R. (2002). Posteriorization by FGF, Wnt, and retinoic acid is required for neural crest induction. Developmental Biology, 241(2), 289–301.
Sauka-Spengler, T., & Bronner-Fraser, M. (2008). A gene regulatory network orchestrates neural crest formation. Nature Reviews Molecular Cell Biology, 9(7), 557–568.
Kalluri, R., & Weinberg, R. A. (2009). The basics of epithelial-mesenchymal transition. Journal of Clinical Investigation, 119(6), 1420–1428.
Bronner, M. E., & LeDouarin, N. M. (2012). Development and evolution of the neural crest: an overview. Developmental Biology, 366(1), 2–9.
Thiery, J. P. (2002). Epithelial–mesenchymal transitions in tumour progression. Nature Reviews Cancer, 2(6), 442–454.
Peinado, H., Portillo, F., & Cano, A. (2004). Transcriptional regulation of cadherins during development and carcinogenesis. International Journal of Developmental Biology, 48(5–6), 365–375.
Batlle, E., Sancho, E., Francí, C., et al. (2000). The transcription factor snail is a repressor of E-cadherin gene expression in epithelial tumour cells. Nature Cell Biology, 2(2), 84–89.
Cano, A., Pérez-Moreno, M. A., Rodrigo, I., et al. (2000). The transcription factor snail controls epithelial–mesenchymal transitions by repressing E-cadherin expression. Nature Cell Biology, 2(2), 76–83.
Hajra, K. M., Chen, D. Y., & Fearon, E. R. (2002). The SLUG zinc-finger protein represses E-cadherin in breast cancer. Cancer Research, 62(6), 1613–1618.
Nieto, M. A. (2002). The snail superfamily of zinc-finger transcription factors. Nature Reviews Molecular Cell Biology, 3(3), 155–166.
De Craene, B., Gilbert, B., Stove, C., Bruyneel, E., van Roy, F., & Berx, G. (2005). The transcription factor snail induces tumor cell invasion through modulation of the epithelial cell differentiation program. Cancer Research, 65(14), 6237–6244.
Balzac, F., Avolio, M., Degani, S., et al. (2005). E-cadherin endocytosis regulates the activity of Rap1: a traffic light GTPase at the crossroads between cadherin and integrin function. Journal of Cell Science, 118(20), 4765–4783.
Thiery, J. P., & Sleeman, J. P. (2006). Complex networks orchestrate epithelial–mesenchymal transitions. Nature Reviews Molecular Cell Biology, 7(2), 131–142.
Kalcheim, C., & Burstyn-Cohen, T. (2005). Early stages of neural crest ontogeny: formation and regulation of cell delamination. International Journal of Developmental Biology, 49(2–3), 105–116.
Jessen, K. R., & Mirsky, R. (2005). The origin and development of glial cells in peripheral nerves. Nature Reviews Neuroscience, 6(9), 671–682.
Mirsky, R., Woodhoo, A., Parkinson, D. B., Arthur‐Farraj, P., Bhaskaran, A., & Jessen, K. R. (2008). Novel signals controlling embryonic Schwann cell development, myelination and dedifferentiation. Journal of the Peripheral Nervous System, 13(2), 122–135.
Pereira, J. A., Lebrun-Julien, F., & Suter, U. (2012). Molecular mechanisms regulating myelination in the peripheral nervous system. Trends in Neurosciences, 35(2), 123–134.
Stolt, C. C., & Wegner, M. (2010). SoxE function in vertebrate nervous system development. International Journal of Biochemistry and Cell Biology, 42(3), 437–440.
Britsch, S., Goerich, D. E., Riethmacher, D., et al. (2001). The transcription factor Sox10 is a key regulator of peripheral glial development. Genes and Development, 15(1), 66–78.
Schreiner, S., Cossais, F., Fischer, K., et al. (2007). Hypomorphic Sox10 alleles reveal novel protein functions and unravel developmental differences in glial lineages. Development, 134(18), 3271–3281.
Inoue, K., Khajavi, M., Ohyama, T., et al. (2004). Molecular mechanism for distinct neurological phenotypes conveyed by allelic truncating mutations. Nature Genetics, 36(4), 361–369.
Finzsch, M., Schreiner, S., Kichko, T., et al. (2010). Sox10 is required for Schwann cell identity and progression beyond the immature Schwann cell stage. Journal of Cell Biology, 189(4), 701–712.
Topilko, P., Schneider-Maunoury, S., Levi, G., et al. (1994). Krox-20 controls myelination in the peripheral nervous system. Nature, 371, 396–399.
Nagarajan, R., Svaren, J., Le, N., Araki, T., Watson, M., & Milbrandt, J. (2001). EGR2 mutations in inherited neuropathies dominant-negatively inhibit myelin gene expression. Neuron, 30(2), 355–368.
Ghislain, J., & Charnay, P. (2006). Control of myelination in Schwann cells: a Krox20 cis-regulatory element integrates Oct6, Brn2 and Sox10 activities. EMBO Reports, 7(1), 52–58.
Warner, L. E., Mancias, P., Butler, I. J., et al. (1998). Mutations in the early growth response 2 (EGR2) gene are associated with hereditary myelinopathies. Nature Genetics, 18(4), 382–384.
Timmerman, V., De Jonghe, P., Ceuterick, C., et al. (1999). Novel missense mutation in the early growth response 2 gene associated with Dejerine-Sottas syndrome phenotype. Neurology, 52(9), 1827–1832.
Boerkoel, C. F., Takashima, H., Bacino, C. A., Daentl, D., & Lupski, J. R. (2001). EGR2 mutation R359W causes a spectrum of Dejerine-Sottas neuropathy. Neurogenetics, 3(3), 153–157.
Jaegle, M., Ghazvini, M., Mandemakers, W., et al. (2003). The POU proteins Brn-2 and Oct-6 share important functions in Schwann cell development. Genes and Development, 17(11), 1380–1391.
Ghislain, J., Desmarquet-Trin-Dinh, C., Jaegle, M., Meijer, D., Charnay, P., & Frain, M. (2002). Characterisation of cis-acting sequences reveals a biphasic, axon-dependent regulation of Krox20 during Schwann cell development. Development, 129(1), 155–166.
Kao, S. C., Wu, H., Xie, J., et al. (2009). Calcineurin/NFAT signaling is required for neuregulin-regulated Schwann cell differentiation. Science, 323(5914), 651–654.
Jagalur, N. B., Ghazvini, M., Mandemakers, W., et al. (2011). Functional dissection of the Oct6 Schwann cell enhancer reveals an essential role for dimeric Sox10 binding. Journal of Neuroscience, 31(23), 8585–8594.
Zorick, T. S., Syroid, D. E., Brown, A., Gridley, T., & Lemke, G. (1999). Krox-20 controls SCIP expression, cell cycle exit and susceptibility to apoptosis in developing myelinating Schwann cells. Development, 126(7), 1397–1406.
Peirano, R. I., & Wegner, M. (2000). The glial transcription factor Sox10 binds to DNA both as monomer and dimer with different functional consequences. Nucleic Acids Research, 28(16), 3047–3055.
Slutsky, S. G., Kamaraju, A. K., Levy, A. M., Chebath, J., & Revel, M. (2003). Activation of myelin genes during transdifferentiation from melanoma to glial cell phenotype. Journal of Biological Chemistry, 278(11), 8960–8968.
Bondurand, N., Girard, M., Pingault, V., Lemort, N., Dubourg, O., & Goossens, M. (2001). Human Connexin 32, a gap junction protein altered in the X-linked form of Charcot-Marie-Tooth disease, is directly regulated by the transcription factor SOX10. Human Molecular Genetics, 10(24), 2783–2795.
Meyer, D., Yamaai, T., Garratt, A., et al. (1997). Isoform-specific expression and function of neuregulin. Development, 124(18), 3575–3586.
Marchionni, M. A., Goodearl, A. D., Chen, M. S., et al. (1993). Glial growth factors are alternatively spliced erbB2 ligands expressed in the nervous system. Nature, 362, 312–318.
Shah, N. M., Marchionni, M. A., Isaacs, I., Stroobant, P., & Anderson, D. J. (1994). Glial growth factor restricts mammalian neural crest stem cells to a glial fate. Cell, 77(3), 349–360.
Dong, Z., Brennan, A., Liu, N., et al. (1995). Neu differentiation factor is a neuron-glia signal and regulates survival, proliferation, and maturation of rat Schwann cell precursors. Neuron, 15(3), 585–596.
Jessen, K., & Mirsky, R. (1997). Embryonic Schwann cell development: the biology of Schwann cell precursors and early Schwann cells. Journal of Anatomy, 191(4), 501–505.
Nave, K., & Salzer, J. L. (2006). Axonal regulation of myelination by neuregulin 1. Current Opinion in Neurobiology, 16(5), 492–500.
Monje, P. V., Bartlett Bunge, M., & Wood, P. M. (2006). Cyclic AMP synergistically enhances neuregulin-dependent ERK and Akt activation and cell cycle progression in Schwann cells. Glia, 53(6), 649–659.
Limpert, A. S., & Carter, B. D. (2010). Axonal neuregulin 1 type III activates NF-kappaB in Schwann cells during myelin formation. Journal of Biological Chemistry, 285(22), 16614–16622.
Nickols, J. C., Valentine, W., Kanwal, S., & Carter, B. D. (2003). Activation of the transcription factor NF-κB in Schwann cells is required for peripheral myelin formation. Nature Neuroscience, 6(2), 161–167.
Arthur‐Farraj, P., Wanek, K., Hantke, J., et al. (2011). Mouse schwann cells need both NRG1 and cyclic AMP to myelinate. Glia, 59(5), 720–733.
Svaren, J., & Meijer, D. (2008). The molecular machinery of myelin gene transcription in Schwann cells. Glia, 56(14), 1541–1551.
Fregien, N. L., White, L. A., Bunge, M. B., & Wood, P. M. (2005). Forskolin increases neuregulin receptors in human Schwann cells without increasing receptor mRNA. Glia, 49(1), 24–35.
Sheean, M. E., McShane, E., Cheret, C., et al. (2014). Activation of MAPK overrides the termination of myelin growth and replaces Nrg1/ErbB3 signals during Schwann cell development and myelination. Genes and Development, 28(3), 290–303.
Cornbrooks, C. J., Carey, D. J., McDonald, J. A., Timpl, R., & Bunge, R. P. (1983). In vivo and in vitro observations on laminin production by Schwann cells. Proceedings of the National academy of Sciences of the United States of America, 80(12), 3850–3854.
Patton, B. L., Miner, J. H., Chiu, A. Y., & Sanes, J. R. (1997). Distribution and function of laminins in the neuromuscular system of developing, adult, and mutant mice. Journal of Cell Biology, 139(6), 1507–1521.
Chen, Z., Yu, W., & Strickland, S. (2007). Peripheral regeneration. Annual Review of Neuroscience, 30, 209–233.
Yu, W. M., Feltri, M. L., Wrabetz, L., Strickland, S., & Chen, Z. L. (2005). Schwann cell-specific ablation of laminin gamma1 causes apoptosis and prevents proliferation. Journal of Neuroscience, 25(18), 4463–4472.
Hossain, S., de la Cruz‐Morcillo, M., Sanchez‐Prieto, R., & Almazan, G. (2012). Mitogen-activated protein kinase p38 regulates krox-20 to direct schwann cell differentiation and peripheral myelination. Glia, 60(7), 1130–1144.
Erceg, S., Ronaghi, M., & Stojković, M. (2009). Human embryonic stem cell differentiation toward regional specific neural precursors. Stem Cells, 27(1), 78–87.
Denham, M., & Dottori, M. (2009). Signals involved in neural differentiation of human embryonic stem cells. Neurosignals, 17(4), 234–241.
Lee, G., Kim, H., Elkabetz, Y., et al. (2007). Isolation and directed differentiation of neural crest stem cells derived from human embryonic stem cells. Natural Biotechnology, 25(12).
Monsoro-Burq, A. H., Fletcher, R. B., & Harland, R. M. (2003). Neural crest induction by paraxial mesoderm in Xenopus embryos requires FGF signals. Development, 130(14), 3111–3124.
Kawaguchi, J., Nichols, J., Gierl, M. S., Faial, T., & Smith, A. (2010). Isolation and propagation of enteric neural crest progenitor cells from mouse embryonic stem cells and embryos. Development, 137(5), 693–704.
ten Berge, D., Koole, W., Fuerer, C., Fish, M., Eroglu, E., & Nusse, R. (2008). Wnt signaling mediates self-organization and axis formation in embryoid bodies. Cell Stem Cell, 3(5), 508–518.
Jessell, T. M. (2000). Neuronal specification in the spinal cord: inductive signals and transcriptional codes. Nature Reviews Genetics, 1(1), 20–29.
Mizuseki, K., Sakamoto, T., Watanabe, K., et al. (2003). Generation of neural crest-derived peripheral neurons and floor plate cells from mouse and primate embryonic stem cells. Proceedings of the National academy of Sciences of the United States of America, 100(10), 5828–5833.
Okawa, T., Kamiya, H., Himeno, T., et al. (2013). Transplantation of neural crest-like cells derived from induced pluripotent stem cells improves diabetic polyneuropathy in mice. Cell Transplantation, 22(10), 1767–1783.
Haastert, K., Mauritz, C., Chaturvedi, S., & Grothe, C. (2007). Human and rat adult Schwann cell cultures: fast and efficient enrichment and highly effective non-viral transfection protocol. Nature Protocols, 2(1), 99–104.
Kalcheim, C., & Gendreau, M. (1988). Brain-derived neurotrophic factor stimulates survival and neuronal differentiation in cultured avian neural crest. Developmental Brain Research, 41(1), 79–86.
Yu, D., Lee, K., Lee, J., et al. (2004). Changes of gene expression profiles during neuronal differentiation of central nervous system precursors treated with ascorbic acid. Journal of Neuroscience Research, 78(1), 29–37.
Eldridge, C. F., Bunge, M. B., Bunge, R. P., & Wood, P. M. (1987). Differentiation of axon-related Schwann cells in vitro. I. Ascorbic acid regulates basal lamina assembly and myelin formation. Journal of Cell Biology, 105(2), 1023–1034.
Pomp, O., Brokhman, I., Ben-Dor, I., Reubinoff, B., & Goldstein, R. S. (2005). Generation of peripheral sensory and sympathetic neurons and neural crest cells from human embryonic stem cells. Stem Cells, 23(7), 923–930.
Motohashi, T., Aoki, H., Chiba, K., Yoshimura, N., & Kunisada, T. (2007). Multipotent cell fate of neural crest-like cells derived from embryonic stem cells. Stem Cells, 25(2), 402–410.
Rathjen, J., Haines, B. P., Hudson, K. M., Nesci, A., Dunn, S., & Rathjen, P. D. (2002). Directed differentiation of pluripotent cells to neural lineages: homogeneous formation and differentiation of a neurectoderm population. Development, 129(11), 2649–2661.
Zhou, Y., & Snead, M. L. (2008). Derivation of cranial neural crest-like cells from human embryonic stem cells. Biochemical and Biophysical Research Communications, 376(3), 542–547.
Jiang, X., Gwye, Y., McKeown, S. J., Bronner-Fraser, M., Lutzko, C., & Lawlor, E. R. (2009). Isolation and characterization of neural crest stem cells derived from in vitro-differentiated human embryonic stem cells. Stem Cells and Development, 18(7), 1059–1070.
Lee, G., Chambers, S. M., Tomishima, M. J., & Studer, L. (2010). Derivation of neural crest cells from human pluripotent stem cells. Nature Protocols, 5(4), 688–701.
Ziegler, L., Grigoryan, S., Yang, I. H., Thakor, N. V., & Goldstein, R. S. (2011). Efficient generation of Schwann cells from human embryonic stem cell-derived neurospheres. Stem Cell Reviews and Reports, 7(2), 394–403.
Cui, L., Jiang, J., Wei, L., et al. (2008). Transplantation of embryonic stem cells improves nerve repair and functional recovery after severe sciatic nerve axotomy in rats. Stem Cells, 26(5), 1356–1365.
Chin, M. H., Mason, M. J., Xie, W., et al. (2009). Induced pluripotent stem cells and embryonic stem cells are distinguished by gene expression signatures. Cell Stem Cell, 5(1), 111–123.
Doi, A., Park, I., Wen, B., et al. (2009). Differential methylation of tissue-and cancer-specific CpG island shores distinguishes human induced pluripotent stem cells, embryonic stem cells and fibroblasts. Nature Genetics, 41(12), 1350–1353.
Liu, Q., Spusta, S. C., Mi, R., et al. (2012). Human neural crest stem cells derived from human ESCs and induced pluripotent stem cells: induction, maintenance, and differentiation into functional schwann cells. Stem Cells Translational Medicine, 1(4), 266–278.
Menendez, L., Kulik, M. J., Page, A. T., et al. (2013). Directed differentiation of human pluripotent cells to neural crest stem cells. Nature Protocols, 8(1), 203–212.
Wang, A., Tang, Z., Park, I., et al. (2011). Induced pluripotent stem cells for neural tissue engineering. Biomaterials, 32(22), 5023–5032.
Uemura, T., Takamatsu, K., Ikeda, M., et al. (2012). Transplantation of induced pluripotent stem cell-derived neurospheres for peripheral nerve repair. Biochemical and Biophysical Research Communications, 419(1), 130–135.
Ikeda, M., Uemura, T., Takamatsu, K., et al. (2013). Acceleration of peripheral nerve regeneration using nerve conduits in combination with induced pluripotent stem cell technology and a basic fibroblast growth factor drug delivery system. Journal of Biomedical Materials Research Part A.
Pomp, O., Brokhman, I., Ziegler, L., et al. (2008). PA6-induced human embryonic stem cell-derived neurospheres: a new source of human peripheral sensory neurons and neural crest cells. Brain Research, 1230, 50–60.
Nakagawa, M., Koyanagi, M., Tanabe, K., et al. (2008). Generation of induced pluripotent stem cells without Myc from mouse and human fibroblasts. Nature Biotechnology, 26(1), 101–106.
Melino, G. (2011). p63 is a suppressor of tumorigenesis and metastasis interacting with mutant p53. Cell Death and Differentiation, 18(9), 1487–1499.
Koyanagi, M., Takahashi, J., Arakawa, Y., et al. (2008). Inhibition of the Rho/ROCK pathway reduces apoptosis during transplantation of embryonic stem cell-derived neural precursors. Journal of Neuroscience Research, 86(2), 270–280.
Huangfu, D., Osafune, K., Maehr, R., et al. (2008). Induction of pluripotent stem cells from primary human fibroblasts with only Oct4 and Sox2. Nature Biotechnology, 26(11), 1269–1275.
Wernig, M., Meissner, A., Foreman, R., et al. (2007). In vitro reprogramming of fibroblasts into a pluripotent ES-cell-like state. Nature, 448(7151), 318–324.
Stadtfeld, M., Nagaya, M., Utikal, J., Weir, G., & Hochedlinger, K. (2008). Induced pluripotent stem cells generated without viral integration. Science, 322(5903), 945–949.
Okita, K., Nakagawa, M., Hyenjong, H., Ichisaka, T., & Yamanaka, S. (2008). Generation of mouse induced pluripotent stem cells without viral vectors. Science, 322(5903), 949–953.
Zhou, H., Wu, S., Joo, J. Y., et al. (2009). Generation of induced pluripotent stem cells using recombinant proteins. Cell Stem Cell, 4(5), 381–384.
Kim, D., Kim, C., Moon, J., et al. (2009). Generation of human induced pluripotent stem cells by direct delivery of reprogramming proteins. Cell Stem Cell, 4(6), 472.
Warren, L., Manos, P. D., Ahfeldt, T., et al. (2010). Highly efficient reprogramming to pluripotency and directed differentiation of human cells with synthetic modified mRNA. Cell Stem Cell, 7(5), 618–630.
Desponts, C., Ding, S. (2010). Using small molecules to improve generation of induced pluripotent stem cells from somatic cells. Cellular Programming and Reprogramming, 207–218.
Woltjen, K., Michael, I. P., Mohseni, P., et al. (2009). PiggyBac transposition reprograms fibroblasts to induced pluripotent stem cells. Nature, 458(7239), 766–770.
Kaji, K., Norrby, K., Paca, A., Mileikovsky, M., Mohseni, P., & Woltjen, K. (2009). Virus-free induction of pluripotency and subsequent excision of reprogramming factors. Nature, 458(7239), 771–775.
Jia, F., Wilson, K. D., Sun, N., et al. (2010). A nonviral minicircle vector for deriving human iPS cells. Nature Methods, 7(3), 197–199.
Yu, J., Hu, K., Smuga-Otto, K., et al. (2009). Human induced pluripotent stem cells free of vector and transgene sequences. Science, 324(5928), 797–801.
Chou, B., Mali, P., Huang, X., et al. (2011). Efficient human iPS cell derivation by a non-integrating plasmid from blood cells with unique epigenetic and gene expression signatures. Cell Research, 21(3), 518–529.
Ban, H., Nishishita, N., Fusaki, N., et al. (2011). Efficient generation of transgene-free human induced pluripotent stem cells (iPSCs) by temperature-sensitive Sendai virus vectors. Proceedings of the National Academy of Sciences, 108(34), 14234–14239.
Li, J., Christophersen, N. S., Hall, V., Soulet, D., & Brundin, P. (2008). Critical issues of clinical human embryonic stem cell therapy for brain repair. Trends in Neurosciences, 31(3), 146–153.
Yamanaka, S., Tanabe, K. (2009). Method for producing induced pluripotent stem cells, US Patent App. 13/059,188.
Kawasaki, H., Mizuseki, K., Nishikawa, S., et al. (2000). Induction of midbrain dopaminergic neurons from ES cells by stromal cell–derived inducing activity. Neuron, 28(1), 31–40.
Chambers, S. M., Qi, Y., Mica, Y., et al. (2012). Combined small-molecule inhibition accelerates developmental timing and converts human pluripotent stem cells into nociceptors. Nature Biotechnology, 30(7), 715–720.
Kreitzer, F. R., Salomonis, N., Sheehan, A., et al. (2013). A robust method to derive functional neural crest cells from human pluripotent stem cells. American Journal of Stem Cells, 2(2), 119.
Vierbuchen, T., Ostermeier, A., Pang, Z. P., Kokubu, Y., Südhof, T. C., & Wernig, M. (2010). Direct conversion of fibroblasts to functional neurons by defined factors. Nature, 463(7284), 1035–1041.
Yang, N., Zuchero, J. B., Ahlenius, H., et al. (2013). Generation of oligodendroglial cells by direct lineage conversion. Nature Biotechnology, 31(5), 434–439.
Najm, F. J., Lager, A. M., Zaremba, A., et al. (2013). Transcription factor-mediated reprogramming of fibroblasts to expandable, myelinogenic oligodendrocyte progenitor cells. Nature Biotechnology, 31(5), 426–433.
Parkinson, D. B., Bhaskaran, A., Droggiti, A., et al. (2004). Krox-20 inhibits Jun-NH2-terminal kinase/c-Jun to control Schwann cell proliferation and death. Journal of Cell Biology, 164(3), 385–394.
Ryu, E. J., Wang, J. Y., Le, N., et al. (2007). Misexpression of Pou3f1 results in peripheral nerve hypomyelination and axonal loss. Journal of Neuroscience, 27(43), 11552–11559.
Conflict of Interest
The authors declare no potential conflict of interests
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ma, MS., Boddeke, E. & Copray, S. Pluripotent Stem Cells for Schwann Cell Engineering. Stem Cell Rev and Rep 11, 205–218 (2015). https://doi.org/10.1007/s12015-014-9577-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12015-014-9577-1