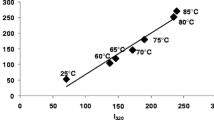
Abstract
Riboflavin (RF) is a vitamin that only exists in plants and microorganisms and must be procured externally by humans. On the other hand, there are two major allergic factors in cow’s milk, including β-lactoglobulin (βLG) and β-casein (βCN), while their allergic properties can be eliminated by binding to micronutrients. In this regard, we examined the binding process of RF to βLG and βCN in the binary and ternary systems by different spectroscopies such as zeta potential, electric conductivity, and molecular modeling. According to the result of the fluorescence spectrum regarding the interaction of RF with βLG and βCN in binary and ternary systems, an increase in RF concentration declined the fluorescence intensity of three systems and also caused the quenching of proteins. Static quenching plays a pivotal role in the formation of stable interactions. The obtained thermodynamic parameters by Van’t Hoff equation ascertained the predominance of hydrogen bonds and van der Waals interaction in all the systems. Considering how the negative value of ΔH0 resulted in the negative value of ΔG0, the systems were assumed to be enthalpy driven. The outcomes of circular dichroism (CD) disclosed that the attachment of RF to the targets of systems increased their a-helix content, which particularly included the binding of RF to βLG that led to the conversion of β-sheet to α-helix content. As indicated by the results of zeta potential, the low concentration of RF contained the dominance of hydrophobic forces in the interactions, whereas the enlargement of this concentration prevailed electrostatic forces. Moreover, conductometry measurements showed an extension in the rate of ionizable groups due to the addition of RF to the systems, which may increase the probability of an interaction between RF, βCN, and βLG in binary and ternary systems. In consistency with the outcomes of molecular dynamics simulation, the data of molecular docking approved the capability of RF in forming strong and stable interactions with βCN and βLG.















Similar content being viewed by others
References
Haug, A., Høstmark, A. T., & Harstad, O. M. (2007). Bovine milk in human nutrition–a review. Lipids in Health and Disease, 6(1), 1–16.
Korhonen, H. J. (2011) Bioactive milk proteins, peptides and lipids and other functional components derived from milk and bovine colostrum. Functional foods: Elsevier, 471–511.
Lin, T., Meletharayil, G., Kapoor, R., & Abbaspourrad, A. (2021). Bioactives in bovine milk: chemistry, technology, and applications. Nutrition Reviews, 79, 48–69.
Santos, D. I., Saraiva, J. M. A., Vicente, A. A., Moldão-Martins, M. (2019) Methods for determining bioavailability and bioaccessibility of bioactive compounds and nutrients. Innovative thermal and non-thermal processing, bioaccessibility and bioavailability of nutrients and bioactive compounds: Elsevier, 23–54.
Høst, A. (1994). Cow’s milk protein allergy and intolerance in infancy some clinical, epidemiological and immunological aspects. Pediatric Allergy and Immunology, 5(S6), 5–36.
Subramani, I. G., Perumal, V., Gopinath, S. C., Mohamed, N. M., Ovinis, M., & Sze, L. L. (2021). 1, 1’-Carbonyldiimidazole-copper nanoflower enhanced collapsible laser scribed graphene engraved microgap capacitive aptasensor for the detection of milk allergen. Scientific Reports, 11(1), 1–12.
Zepeda-Ortega, B., Goh, A., Xepapadaki, P., Sprikkelman, A., Nicolaou, N., & Hernandez, R. E. H., et al. (2021). Strategies and future opportunities for the prevention, diagnosis, and management of cow milk allergy. Frontiers in Immunology, 12, 1877.
Marcone, S., Belton, O., & Fitzgerald, D. J. (2017). Milk‐derived bioactive peptides and their health promoting effects: a potential role in atherosclerosis. British Journal of Clinical Pharmacology, 83(1), 152–162.
Wong, D. W., Camirand, W. M., Pavlath, A. E., Parris, N., & Friedman, M. (1996). Structures and functionalities of milk proteins. Critical Reviews in Food Science & Nutrition. 36(8), 807–844.
Truswell, A. (2005). The A2 milk case: a critical review. European Journal of Clinical Nutrition, 59(5), 623–631.
Mayer, H. K., Lenz, K., & Halbauer, E.-M. (2021). “A2 milk” authentication using isoelectric focusing and different PCR techniques. Food Research International, 147, 110523.
McLachlan, C. (2001). β-casein A1, ischaemic heart disease mortality, and other illnesses. Medical Hypotheses, 56(2), 262–272.
Thakur, N., Chauhan, G., Mishra, B., Mendiratta, S., Pattanaik, A., & Singh, T. U., et al. (2020). Comparative evaluation of feeding effects of A1 and A2 cow milk derived casein hydrolysates in diabetic model of rats. Journal of Functional Foods, 75, 104272.
Fiocchi, A., Bognanni, A., Brożek, J., Ebisawa, M., Schünemann, H., & Ansotegui, I. J., et al. (2022). World Allergy Organization (WAO) diagnosis and rationale for action against cow’s milk allergy (dracma) guidelines update–I–Plan and definitions. World Allergy Organisation Journal. 15(1), 100609.
Hufnagl, K., Ghosh, D., Wagner, S., Fiocchi, A., Dahdah, L., & Bianchini, R., et al. (2018). Retinoic acid prevents immunogenicity of milk lipocalin Bos d 5 through binding to its immunodominant T-cell epitope. Scientific Reports, 8(1), 1–12.
Afify, S. M., Pali-Schöll, I., Hufnagl, K., Hofstetter, G., El-Bassuoni, M. A.-R., & Roth-Walter, F., et al. (2021). Bovine beta-lactoglobulin cross-protects against pollen allergies in an innate manner in BALB/c mice: Potential model for the farm effect. Frontiers in Immunology, 12, 176.
Roth-Walter, F., Pacios, L. F., Gomez-Casado, C., Hofstetter, G., Roth, G. A., & Singer, J., et al. (2014). The major cow milk allergen Bos d 5 manipulates T-helper cells depending on its load with siderophore-bound iron. PloS ONE, 9(8), e104803.
Loss, G., Apprich, S., Waser, M., Kneifel, W., Genuneit, J., & Büchele, G., et al. (2011). The protective effect of farm milk consumption on childhood asthma and atopy: the GABRIELA study. Journal of Allergy and Clinical Immunology, 128(4), 766–73.e4.
Roth-Walter, F., Afify, S. M., Pacios, L. F., Blokhuis, B. R., Redegeld, F., & Regner, A., et al. (2021). Cow’s milk protein β-lactoglobulin confers resilience against allergy by targeting complexed iron into immune cells. Journal of Allergy and Clinical Immunology, 147(1), 321–34.e4.
Considine, T., Flanagan, J., Loveday, S.M., Ellis, A. (2020) Interaction between milk proteins and micronutrients. Milk proteins: Elsevier, 537–571.
Plantone, D., Pardini, M., & Rinaldi, G. (2021). Riboflavin in neurological diseases: a narrative review. Clinical Drug Investigation, 41(6), 513–527.
You, J., Pan, X., Yang, C., Du, Y., Osire, T., & Yang, T., et al. (2021). Microbial production of riboflavin: biotechnological advances and perspectives. Metabolic Engineering, 68, 46–58.
Zhao, G., Dong, F., Lao, X., & Zheng, H. (2021). Strategies to increase the production of biosynthetic riboflavin. Molecular Biotechnology, 63(10), 909–918.
Lei, J., Xin, C., Xiao, W., Chen, W., & Song, Z. (2021). The promise of endogenous and exogenous riboflavin in anti-infection. Virulence., 12(1), 2314–2326.
Murakami, K., Miyake, Y., Sasaki, S., Tanaka, K., Fukushima, W., & Kiyohara, C., et al. (2010). Dietary intake of folate, vitamin B6, vitamin B12 and riboflavin and risk of Parkinson’s disease: a case–control study in Japan. British Journal of Nutrition, 104(5), 757–764.
Li, D., Zhu, J., Jin, J., & Yao, X. (2007). Studies on the binding of nevadensin to human serum albumin by molecular spectroscopy and modeling. Journal of Molecular Structure, 846(1-3), 34–41.
Zhao, R., Qin, X., & Zhong, J. (2021). Interaction between curcumin and β-Casein: multi-spectroscopic and molecular dynamics simulation methods. Molecules., 26(16), 5092.
Al-Shabib, N. A., Khan, J. M., Malik, A., Alsenaidy, M. A., Rehman, M. T., & AlAjmi, M. F., et al. (2018). Molecular insight into binding behavior of polyphenol (rutin) with beta lactoglobulin: spectroscopic, molecular docking and MD simulation studies. Journal of Molecular Liquids, 269, 511–520.
Moeiniafshari, A.-A., Zarrabi, A., & Bordbar, A.-K. (2015). Exploring the interaction of naringenin with bovine beta-casein nanoparticles using spectroscopy. Food Hydrocolloids, 51, 1–6.
Hou, H.-N., Qi, Z.-D., OuYang, Y.-W., Liao, F.-L., Zhang, Y., & Liu, Y. (2008). Studies on interaction between Vitamin B12 and human serum albumin. Journal of Pharmaceutical and Biomedical Analysis, 47(1), 134–139.
Li, Y., He, W., Liu, J., Sheng, F., Hu, Z., & Chen, X. (2005). Binding of the bioactive component jatrorrhizine to human serum albumin. Biochimica et Biophysica Acta (BBA)-General Subjects, 1722(1), 15–21.
Aprodu, I., Ursache, F.-M., Turturică, M., Râpeanu, G., & Stănciuc, N. (2017). Thermal stability of the complex formed between carotenoids from sea buckthorn (Hippophae rhamnoides L.) and bovine β-lactoglobulin. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 173, 562–571.
Jones, G. A., & Bradshaw, D. S. (2019). Resonance energy transfer: from fundamental theory to recent applications. Frontiers in Physics, 7, 100.
Yang, G., Liu, Y., Teng, J., & Zhao, C.-X. (2021). FRET ratiometric nanoprobes for nanoparticle monitoring. Biosensors, 11(12), 505.
Pimsin, N., Kongsanan, N., Keawprom, C., Sricharoen, P., Nuengmatcha, P., & Oh, W.-C., et al. (2021). Ultratrace detection of nickel (II) ions in water samples using dimethylglyoxime-doped GQDs as the induced metal complex nanoparticles by a resonance light scattering sensor. ACS Omega, 6(23), 14796–14805.
Chaturvedi, S. K., Ahmad, E., Khan, J. M., Alam, P., Ishtikhar, M., & Khan, R. H. (2015). Elucidating the interaction of limonene with bovine serum albumin: a multi-technique approach. Molecular BioSystems, 11(1), 307–316.
Zhang, H.-X., Huang, X., & Zhang, M. (2008). Thermodynamic studies on the interaction of dioxopromethazine to β-cyclodextrin and bovine serum albumin. Journal of Fluorescence, 18(3), 753–760.
Jones, C. (2021). Wavelength calibration uncertainty in protein circular dichroism data bank spectra. Applied Spectroscopy, 75(9), 1207–1211.
Ruzza, P., Honisch, C., Hussain, R., & Siligardi, G. (2021). Free radical generation in Far-UV synchrotron radiation circular dichroism assays—protein and buffer composition contribution. International Journal of Molecular Sciences, 22(21), 11325.
Ferraris, S., Cazzola, M., Peretti, V., Stella, B., & Spriano, S. (2018). Zeta potential measurements on solid surfaces for in vitro biomaterials testing: surface charge, reactivity upon contact with fluids and protein absorption. Frontiers in Bioengineering and Biotechnology, 6, 60.
Chen, V. B., Arendall, W. B., Headd, J. J., Keedy, D. A., Immormino, R. M., & Kapral, G. J., et al. (2010). MolProbity: all-atom structure validation for macromolecular crystallography. Acta Crystallographica Section D: Biological Crystallography, 66(1), 12–21.
Mu, G., Zhang, Z., Wang, J., Jiang, S., Wang, H., & Xu, Y., et al. (2021). Antigenicity and safety evaluation of lactiplantibacillus plantarum 7-2 screened to reduce α-Casein antigen. Foods, 11(1), 88.
Acknowledgements
The financial support of the Research Council of the Mashhad Branch, Islamic Azad University, is gratefully acknowledged.
Funding
The financial support of the Research Council of the Mashhad Branch, Islamic Azad University is appreciated and acknowledged.
Author information
Authors and Affiliations
Contributions
F.S. wrote the main manuscript text. S.M., Z.A., F.S., F.R., and S.T. conducted an investigated and amassed the data; Z.A., M.S., and J.C. revised the manuscript. J.C. supervised and conceptualised the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Samandar, F., Malek-Mohammadi, S., Aram, Z. et al. New Perspective on the Interaction Behavior Between Riboflavin and β Lactoglobulin-β Casein Complex by Biophysical Techniques. Cell Biochem Biophys 82, 175–191 (2024). https://doi.org/10.1007/s12013-023-01197-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-023-01197-2