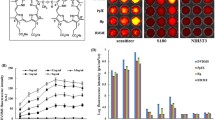
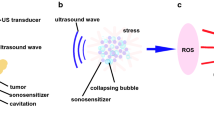
Abstract
The effects of ultrasonic parameters and treatment conditions on the in vitro cellular experiments of sonodynamic therapy (SDT) have not been fully studied. Exploring the factors that affect the efficacy of SDT can provide a reference for screening effective sonosensitizers in vitro. The aim of this work is to investigate the factors that affected the SDT effects in cancer cells. Cancer cells in culture plates were exposed to ultrasound and sonosensitizers. The intracellular drug concentration was measured by using flow cytometry and the cell viability was determined by MTT assay. The SDT effects of cancer cells treated with different ultrasonic parameters under the same sonosensitizer concentration were different. The ultrasonic parameters, intracellular drug concentration, drug treatment time, cell amount, and cell status could affect the sonodynamic therapeutic effects. It is necessary to select appropriate ultrasound conditions and optimize the cellular status to make the results of the in vitro cellular experiments more reliable.










Similar content being viewed by others
References
Xu, J., Xia, X., Wang, X., Xu, C., Wang, P., Xiang, J., et al. (2010). Sonodynamic action of pyropheophorbide—A methyl ester in liver cancer cells. Journal of Ultrasound in Medicine, 29(7), 1031–1037
Suehiro, S., Ohnishi, T., Yamashita, D., Kohno, S., Inoue, A., Nishikawa, M., et al. (2018). Enhancement of antitumor activity by using 5-ALA-mediated sonodynamic therapy to induce apoptosis in malignant gliomas: Significance of high-intensity focused ultrasound on 5-ALA-SDT in a mouse glioma model. Journal of Neurosurgery, 129(6), 1416–1428
Osaki, T., Uto, Y., Ishizuka, M., Tanaka, T., Yamanaka, N. & Kurahashi, T. et al.(2017). Artesunate enhances the cytotoxicity of 5-aminolevulinic acid-based sonodynamic therapy against mouse mammary tumor cells in vitro. Molecules, 22, 4
Shen, J., Cao, S., Sun, X., Pan, B., Cao, J., Che, D., et al. (2018). Sinoporphyrin sodium-mediated sonodynamic therapy inhibits RIP3 expression and induces apoptosis in the H446 small cell lung cancer cell line. Cellular Physiology and Biochemistry, 51(6), 2938–2954
Rosenthal, I., Sostaric, J. Z., & Riesz, P. (2004). Sonodynamic therapy—A review of the synergistic effects of drugs and ultrasound. Ultrasonics Sonochemistry, 11(6), 349–363
Wan, G. Y., Liu, Y., Chen, B. W., Liu, Y. Y., Wang, Y. S., & Zhang, N. (2016). Recent advances of sonodynamic therapy in cancer treatment. Cancer Biology & Medicine, 13(3), 325–338
Snehota, M., Vachutka, J., Ter Haar, G., Dolezal, L., & Kolarova, H. (2020). Therapeutic ultrasound experiments in vitro: Review of factors influencing outcomes and reproducibility. Ultrasonics, 107, 106167
Geng, C., Zhang, Y., Hidru, T. H., Zhi, L., Tao, M., Zou, L., et al. (2018). Sonodynamic therapy: A potential treatment for atherosclerosis. Life Sciences, 207, 304–313
Ninomiya, K., Noda, K., Ogino, C., Kuroda, S. I., & Shimizu, N. (2014). Enhanced OH radical generation by dual-frequency ultrasound with TiO2 nanoparticles: Its application to targeted sonodynamic therapy. Ultrasonics Sonochemistry, 21(1), 289–294
Secomski, W., Bilmin, K., Kujawska, T., Nowicki, A., Grieb, P., & Lewin, P. A. (2017). In vitro ultrasound experiments: Standing wave and multiple reflections influence on the outcome. Ultrasonics, 77, 203–213
Miyoshi, N., Sostaric, J. Z., & Riesz, P. (2003). Correlation between sonochemistry of surfactant solutions and human leukemia cell killing by ultrasound and porphyrins. Free Radical Biology & Medicine, 34(6), 710–719
Zhu, B., Liu, Q., Wang, Y., Wang, X., Wang, P., Zhang, L., et al. (2010). Comparison of accumulation, subcellular location, and sonodynamic cytotoxicity between hematoporphyrin and protoporphyrin IX in L1210 cells. Chemotherapy, 56(5), 403–410
Liu, Y., Bai, H., Wang, H., Wang, X., Liu, Q., Zhang, K., et al. (2019). Comparison of hypocrellin B-mediated sonodynamic responsiveness between sensitive and multidrug-resistant human gastric cancer cell lines. Journal of Medical Ultrasonics, 46(1), 15–26
Zhang, P., Ren, Z., Chen, Z., Zhu, J., Liang, J., Liao, R., et al. (2018). Iron oxide nanoparticles as nanocarriers to improve chlorin e6-based sonosensitivity in sonodynamic therapy. Drug Design, Development and Therapy, 12, 4207–4216
Chang, N., Qin, D., Wu, P., Xu, S., Wang, S., & Wan, M. (2019). IR780 loaded perfluorohexane nanodroplets for efficient sonodynamic effect induced by short-pulsed focused ultrasound. Ultrasonics Sonochemistry, 53, 59–67
Prescott, M., Mitchell, J., Totti, S., Lee, J., Velliou, E., & Bussemaker, M. (2018). Sonodynamic therapy combined with novel anti-cancer agents, sanguinarine and ginger root extract: Synergistic increase in toxicity in the presence of PANC-1 cells in vitro. Ultrasonics Sonochemistry, 40(Pt B), 72–80
Varchi, G., Foglietta, F., Canaparo, R., Ballestri, M., Arena, F., Sotgiu, G., et al. (2015). Engineered porphyrin loaded core-shell nanoparticles for selective sonodynamic anticancer treatment. Nanomedicine, 10(23), 3483–3494
Osaki, T., Yokoe, I., Uto, Y., Ishizuka, M., Tanaka, T., Yamanaka, N., et al. (2016). Bleomycin enhances the efficacy of sonodynamic therapy using aluminum phthalocyanine disulfonate. Ultrasonics Sonochemistry, 28, 161–168
Chen, H. J., Zhou, X. B., Gao, Y., Zheng, B. Y., Tang, F. X., & Huang, J. D. (2014). Recent progress in development of new sonosensitizers for sonodynamic cancer therapy. Drug Discovery Today, 19(4), 502–509
Liu, Q., Li, X., Xiao, L., Wang, P., Wang, X., & Tang, W. (2008). Sonodynamically induced antitumor effect of hematoporphyrin on Hepatoma 22. Ultrasonics Sonochemistry, 15(6), 943–948
Yumita, N., Nishigaki, R., Umemura, K., & Umemura, S. I. (1989). Hematoporphyrin as a sensitizer of cell‐damaging effect of ultrasound. Japanese Journal of Cancer Research, 80(3), 219–222
Su, X., Wang, P., Wang, X., Guo, L., Li, S., & Liu, Q. (2013). Involvement of MAPK activation and ROS generation in human leukemia U937 cells undergoing apoptosis in response to sonodynamic therapy. International Journal of Radiation Biology, 89(11), 915–927
Zheng, R., Zhang, W., Wang, X., & Gao, H. (2010). The sonodynamic effects of Chlorin e6 on the proliferation of human lung adenocarcinoma cell SPCA-1. Zhongguo Fei Ai Za Zhi, 13(3), 201–205
Kimura, T., Sakamoto, T., Leveque, J.-M., Sohmiya, H., Fujita, M., Ikeda, S., et al. (1996). Standardization of ultrasonic power for sonochemical reaction. Ultrasonics Sonochemistry 3(3), S157–S161
O’Brien, W. D. (2007). Ultrasound—Biophysics mechanisms. Progress in Biophysics and Molecular Biology, 93(1–3), 212–255
Kasaai, M. R. (2013). Input power-mechanism relationship for ultrasonic irradiation food and polymer applications. Natural Science, 5(8), 14–22
Barati, A. H., Mokhtari-Dizaji, M., Mozdarani, H., Bathaie, Z., & Hassan, Z. M. (2007). Effect of exposure parameters on cavitation induced by low-level dual-frequency ultrasound. Ultrasonics Sonochemistry, 14(6), 783–789
Kuroki, M., Hachimine, K., Abe, H., Shibaguchi, H., Kuroki, M., Maekawa, S.-I, et al. (2007). Sonodynamic therapy of cancer using novel sonosensitizers. Anticancer Research, 27(6A), 3673–3678
Ter Haar, G. (2011). Ultrasonic imaging: Safety considerations. Interface Focus, 1(4), 686–697
Lin, X. H., Song, J. B., Chen, X. Y., & Yang, H. H. (2020). Ultrasound activated sensitizers and applications. Angewandte Chemie International Edition, 59, 2–24
Wang, P., Wang, X. B., Liu, Q. H., Tang, W., & Li, T. (2008). Enhancement of ultrasonically induced cytotoxic effect by hematoporphyrin in vitro. Chemotherapy, 54(5), 364–371
Hensel, K., Mienkina, M. P., & Schmitz, G. (2011). Analysis of ultrasound fields in cell culture wells for in vitro ultrasound therapy experiments. Ultrasound in Medicine and Biology, 37(12), 2105–2115
Poltawski, L., & Watson, T. (2007). Relative transmissivity of ultrasound coupling agents commonly used by therapists in the UK. Ultrasound in Medicine and Biology, 33(1), 120–128
Thanh Nguyen, T., Asakura, Y., Koda, S., & Yasuda, K. (2017). Dependence of cavitation, chemical effect, and mechanical effect thresholds on ultrasonic frequency. Ultrasonics Sonochemistry, 39, 301–306
Armour, E. P., & Corry, P. M. (1982). Cytotoxic effects of ultrasound in vitro dependence on gas content, frequency, radical scavengers, and attachment. Radiation Research, 89(2), 369–380
Pang, X., Xu, C. S., Jiang, Y., Xiao, Q. C., & Leung, A. W. (2016). Natural products in the discovery of novel sonosensitizers. Pharmacology & Therapeutics, 162, 144–151
Sugita, N., Kawabata, K., Sasaki, K., Sakata, I., & Umemura, S. (2007). Synthesis of amphiphilic derivatives of rose bengal and their tumor accumulation. Bioconjugate Chemistry, 18(3), 866–873
Yumita, N., & Umemura, S. (2003). Sonodynamic therapy with photofrin II on AH130 solid tumor. Pharmacokinetics, tissue distribution and sonodynamic antitumoral efficacy of photofrin II. Cancer Chemotherapy and Pharmacology, 51(2), 174–178
Yumita, N., Nishigaki, R., & Umemura, S. (2000). Sonodynamically induced antitumor effect of Photofrin II on colon 26 carcinoma. Journal of Cancer Research and Clinical Oncology, 126(10), 601–606
Redmond, R. W., & Kochevar, I. E. (2006). Spatially resolved cellular responses to singlet oxygen. Photochemistry and Photobiology, 82(5), 1178–1186
Ozben, T. (2007). Oxidative stress and apoptosis: Impact on cancer therapy. Journal of Pharmaceutical Sciences, 96(9), 2181–2196
Kim, Y. S., Rhim, H., Choi, M. J., Lim, H. K., & Choi, D. (2008). High-intensity focused ultrasound therapy: An overview for radiologists. Korean Journal of Radiology, 9(4), 291–302
VanBavel, E. (2007). Effects of shear stress on endothelial cells: Possible relevance for ultrasound applications. Progress in Biophysics and Molecular Biology, 93(1–3), 374–383
Lajoinie, G., De Cock, I., Coussios, C. C., Lentacker, I., Le Gac, S., Stride, E., et al. (2016). In vitro methods to study bubble-cell interactions: Fundamentals and therapeutic applications. Biomicrofluidics, 10(1), 011501
Guzman, H. R., McNamara, A. J., Nguyen, D. X., & Prausnitz, M. R. (2003). Bioeffects caused by changes in acoustic cavitation bubble density and cell concentration: A unified explanation based on cell-to-bubble ratio and blast radius. Ultrasound in Medicine and Biology, 29(8), 1211–1222
Brayman, A. A., Church, C. C., & Miller, M. W. (1996). Re-evaluation of the concept that high cell concentrations “protect” cells in vitro from ultrasonically induced lysis. Ultrasound in Medicine and Biology, 22(4), 497–514
Shankar, H., & Pagel, P. S. (2011). Potential adverse ultrasound-related biological effects: A critical review. Anesthesiology, 115(5), 1109–1124
Sviridov, A. P., Andreev, V. G., Ivanova, E. M., Osminkina, L. A., Tamarov, K. P. & & Timoshenko, V. Y. (2013). Porous silicon nanoparticles as sensitizers for ultrasonic hyperthermia. Applied Physics Letters, 103, 19
Acknowledgements
This work was supported by the National Natural Science Foundation of China (81871481 and 81571802), the Fujian Provincial Youth Top-notch Talent Support program, China and the Natural Science Foundation of Fujian Province (2022J01069).
Author information
Authors and Affiliations
Contributions
Y.Z.: writing—original draft, investigation, data curation. J.W.: investigation, validation. H.C.: resources. Y.G.: conceptualization, writing—review and editing, supervision, funding acquisition.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zheng, Y., Wang, J., Chen, H. et al. Exploring Different Ultrasonic Parameters and Treatment Conditions to Optimize In Vitro Sonodynamic Therapeutic Effects in Cancer Cells. Cell Biochem Biophys 82, 303–314 (2024). https://doi.org/10.1007/s12013-023-01189-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-023-01189-2